The Periodic Table Dimitri Mendeleev I Periods n

The Periodic Table Dimitri Mendeleev

I. Periods n The rows ( ) on the periodic table are called PERIODS. A. There are 7 periods, just as there are 7 energy levels. B. Each period has a similar econfiguration. *1 st period: outer e- occupies the 1 s orbital *2 nd period: 2 s and 2 p orbitals *3 rd period: 3 s and 3 p orbitals

II. Groups or Families n The columns ( ) are called GROUPS or FAMILIES. They usually have similar properties. n There are 18 groups or families.

III. Classification of Elements A. n n n n METALS Found to the left of the staircase Have luster (shiny) Excellent conductors of electricity Ductile (can be rolled into wires Malleable (can be flattened into sheets) React with water and oxygen in the air and will rust or corrode LOSE valence electrons

B. NONMETALS – – Have no luster Are not ductile Are not malleable Do not react with water and oxygen to rust or corrode – Poor/no conductivity – GAIN electrons

C. METALLOIDS – Have characteristics of both metals and nonmetals – Semi-conductors = o. k. at conducting electricity The elements that are classified as metalloids are: B, Si, Ge, As, Sb, Te, Po, At ***All elements touching the staircase, EXCEPT FOR aluminum, are metalloids.

IV. Main Group or Representative Elements (s- and p-blocks) n Groups 1 A, 2 A, 3 A – 8 A (13 -18) n A. All elements in each group or family have the same number of outer (valence) electrons. Ex: Group 3 A> all elements in that family have the same # of valence electrons, 3. n B. There are 8 groups in the “Representative Elements”. (We will list them below. )
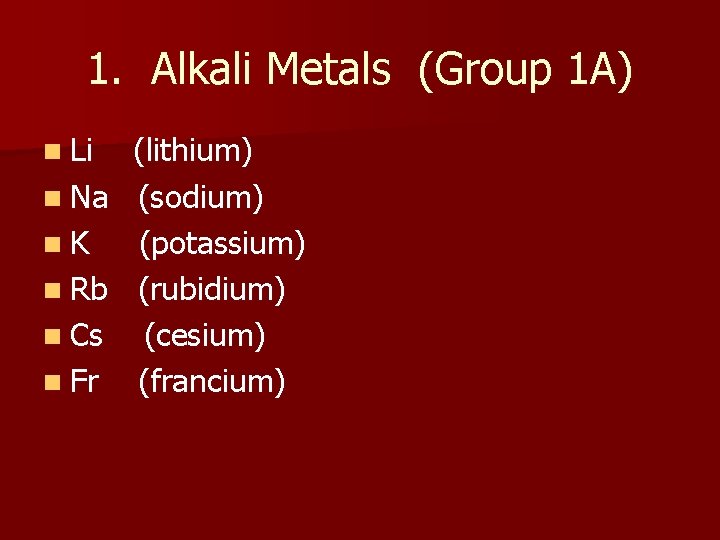
1. Alkali Metals (Group 1 A) n Li n Na n. K n Rb n Cs n Fr (lithium) (sodium) (potassium) (rubidium) (cesium) (francium)

n High reactivity—they are the MOST reactive metals n Explosive in water n Very soft metals n React with oxygen in the air n Have 1 valence electron n Lose 1 electron to become stable and then have a n +1 charge n Excellent conductors of electricity

2. Alkaline-Earth Metals (Group 2 A) n Be (beryllium) n Mg (magnesium) n Ca (calcium) n Sr (strontium) n Ba (barium) n Ra (radium)

n Reactive, but not as reactive as the Gr. 1 A n Harder and stronger than Gr. 1 A n Have 2 valence electrons n Lose 2 electrons and then have a n +2 charge

3. The Boron Family (Group 3 A) n. B n Al n Ga n In n Tl (boron – a metalloid) (aluminum) The rest are metals. (gallium) (indium) (thallium)

The METALS of this group (all of them except for boron) n Have 3 valence electrons n Usually lose 3 electrons n +3 charge

4. The Carbon Family (Group 4 A) n. C n Si n Ge n Sn n Pb (carbon – nonmetal) (silicon – metalloid) (germanium – metalloid) (tin – metal) (lead – metal)

n Have 4 valence electrons n Carbon, silicon, and germanium share electrons – do not gain or lose electrons n Tin and lead lose 2 or 4 electrons for a +2 or +4 charge.

5. The Nitrogen Family (Group 5 A) n. N n. P n As n Sb n Bi (nitrogen – nonmetal) (phosphorus – nonmetal) (arsenic – metalloid) (antimony – metalloid) (bismuth – metal)

n Have 5 valence electrons n GAIN 3 electrons and have a n -3 charge

6. The Oxygen Family (Group 6 A) n. O n. S n Se n Te n Po (oxygen – nonmetal) (sulfur – nonmetal) (selenium – nonmetal) (tellurium – metalloid) (polonium – metalloid)

n Have 6 valence electrons n GAIN 2 electrons to have a n -2 charge n Also called the chalcogens

7. Halogens (Group 7 A) n. F n Cl n Br n. I n At (fluorine -- nonmetal) (chlorine -- nonmetal) (bromine -- nonmetal) (iodine -- nonmetal) (astatine – metalloid)

n Highly reactive -- they are the most reactive nonmetals. n Usually combine with most metals to form salts (“Halogen” is Greek for ‘salt former’. ) n Have 7 valence electrons n GAIN 1 electron to have a n -1 charge

8. Noble Gases (Group 8 A) n He (helium) n Ne (neon) n Ar (argon) n Kr (krypton) n Xe (xenon) n Rn (radon) All are nonmetals, gases.

n Have 8 valence electrons n Do NOT react with other elements because they do not need any more electrons to be stable n Have a “ 0” charge

C. Transition Metals n Found between Gr. 2 A and Gr. 3 A (“D” block) n Valence electrons vary n They all LOSE electrons n Some common transition metals are copper, silver, gold, zinc,

D. Rare Earth Metals or Inner Transition Metals 1. Lanthanoid Series: (also called the Lanthanide series) -- atomic #58 – 71 n They are shiny, reactive metals that are often used to make alloys. n 2. Actanoid Series: (also called the Actinide series) -- atomic #90 – 103 n Have unstable arrangements or protons and neutrons n All are radioactive and most are man-made n

E. Hydrogen n Most common isotope has 1 proton and no neutrons n Most common element in the universe n Behaves like no other element n Has 1 valence electron n Loses the electron to have a n +1 charge
- Slides: 26