The Periodic Table Dimitri Mendeleev Dimitri Mendeleevs Table

The Periodic Table

Dimitri Mendeleev

Dimitri Mendeleev’s Table l. He grouped the elements in columns by similar properties and in order of increasing atomic mass. He predicted the properties of several elements before they were discovered.

The Modern Periodic Table l Elements are still grouped by similar properties in the same column or group. But… l Now they are ordered by increasing atomic number instead of increasing atomic mass. l Thanks to Henry Moseley, who determined the atomic numbers!

Horizontal rows are called PERIODS. 1 There are 7 periods 2 3 4 5 6 7

Vertical columns are called groups or families. Elements in groups (or columns) exhibit similar properties. 1 2 13 14 15 16 17 3 4 5 6 7 8 9 10 11 12 18

Representative Elements l Exhibit a wide range of both physical and chemical properties l Groups 1 A through 8 A (or groups 1 -2, and 13 -18)
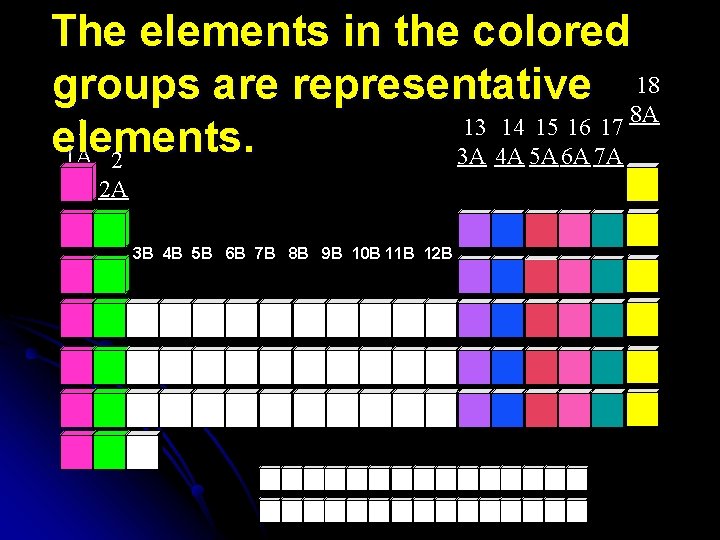
The elements in the colored groups are representative 18 8 A 1 13 14 15 16 17 elements. 1 A 2 3 A 4 A 5 A 6 A 7 A 2 A 3 B 4 B 5 B 6 B 7 B 8 B 9 B 10 B 11 B 12 B
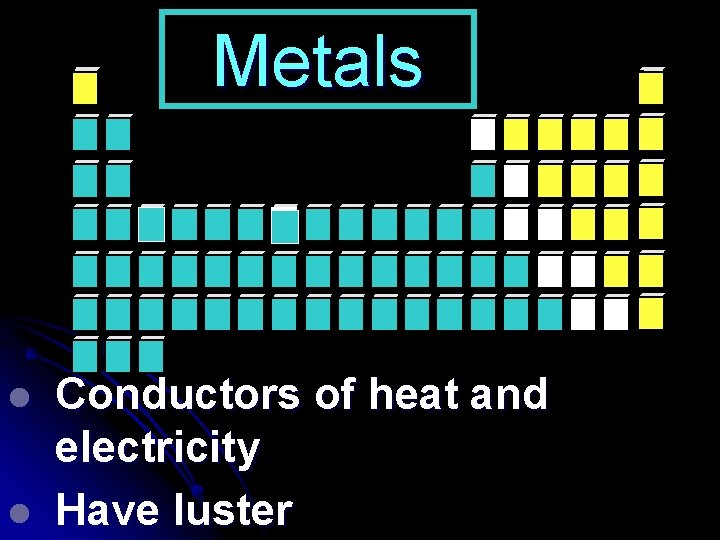
Metals l l Conductors of heat and electricity Have luster

• • Brittle as solids Can also be gases or liquids Non-conductors Non-lustrous Nonmetals

Semi-metals or Metalloids Properties of both metals and nonmetals B Al Si Ge As Sb Te Po At Bold Staircase separates metals on the left from nonmetals on the right.
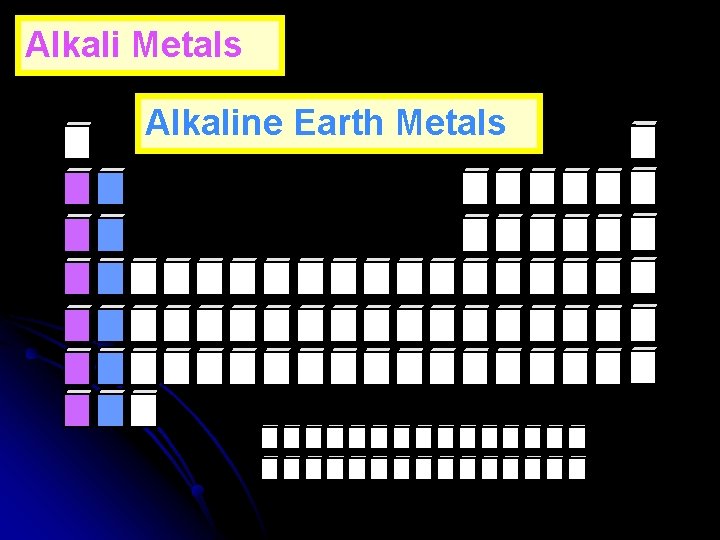
Alkali Metals Alkaline Earth Metals

Transition metals Groups 3 – 12, or the “B” Group Inner Transition Metals
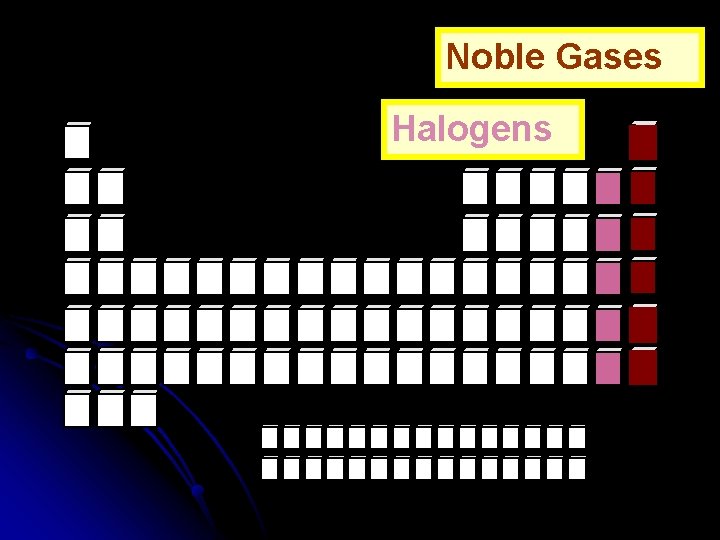
Noble Gases Halogens

Color Coding of Periodic Table Each element on our periodic table is color coded according to the physical state of the element when it’s in its natural state. l Red indicates a gas. l Blue indicates a liquid. l Black indicates a solid. l White (outlined) indicates elements that don’t occur naturally. l
- Slides: 15