The Periodic Table Development of the Periodic Table

The Periodic Table

Development of the Periodic Table • Dmitri Mendeleev- arranged the periodic table based on average atomic masses – Noticed a pattern in the elements when arranged by their atomic masses • Every 8 th element had similar chemical properties – However, after new elements were discovered, some elements weren’t in the correct spots

• Henry Mosely– Arranged the periodic table by increasing atomic number (# of protons) – Resulted in a clear pattern of the chemical properties of the elements. – Periodic Law- a periodic repetition of chemical and physical properties of elements

First thing first • Rows- periods • Columns- groups or families
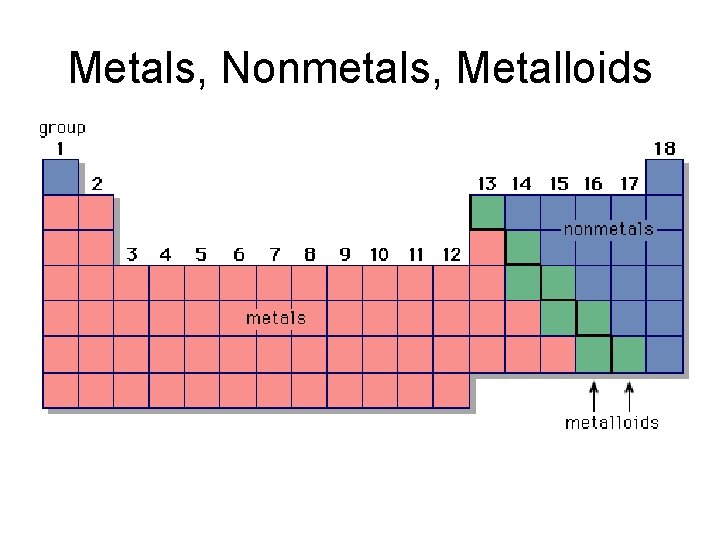
Metals, Nonmetals, Metalloids
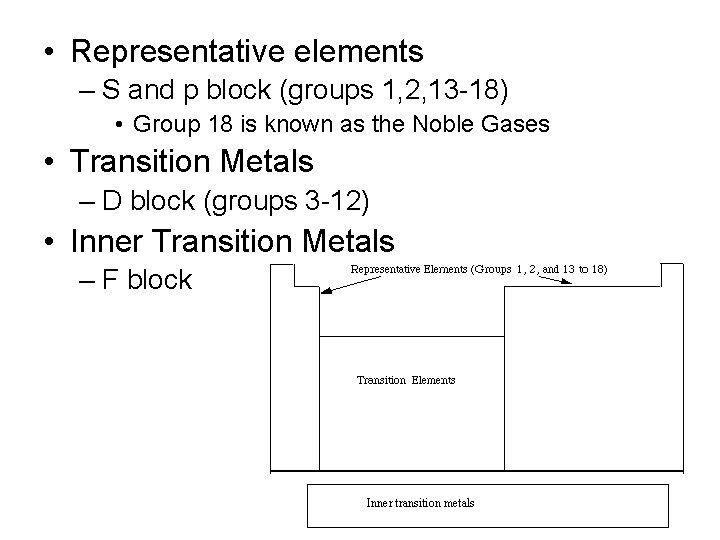
• Representative elements – S and p block (groups 1, 2, 13 -18) • Group 18 is known as the Noble Gases • Transition Metals – D block (groups 3 -12) • Inner Transition Metals – F block Inner transition metals

Valence Shell • The outermost (or highest) energy level containing electrons • All atoms want to have a full valence shell – Full= 8 electrons • except H and He (full = 2 electrons) – The Noble Gases have a full valence shell • Atoms will lose or gain electrons to achieve a full valence shell

How do we find valence electrons? 1. Write the electron configuration for that element. 2. Locate the highest energy level 3. Add up the number of electrons in each orbital of that energy level Example- Write the electron configuration for each element: 1. Ne 2. Ar 3. Kr
- Slides: 8