The Periodic Table Created by Mendeleev Lists all

The Periodic Table • Created by Mendeleev • Lists all elements known to humans. • Everything in the universe is made up of these ‘building blocks’.

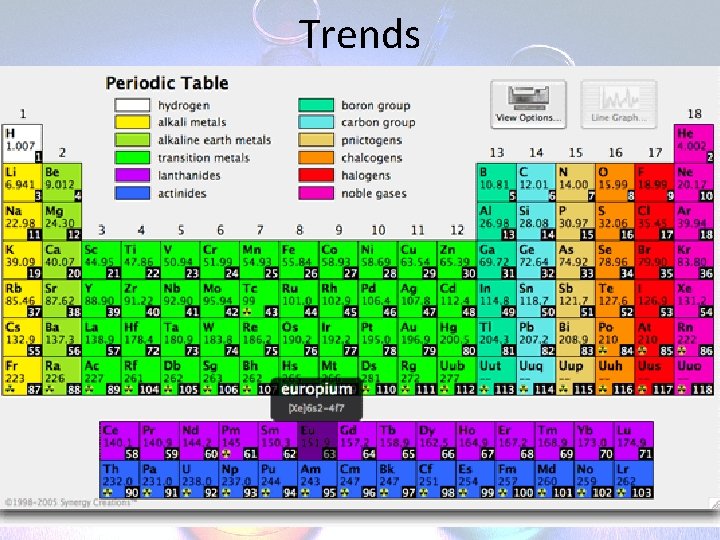
The Periodic Table • • • Metals on the left Non-Metals on the right Separated by the “staircase” Metalloids have properties of both Refer to back in text

The Periodic Table • Metals are – shiny, malleable, good conductors, and mostly solids (at room temp. ) – React with acid • Non-Metals – dull, brittle, insulators, can be solids liquids or gases – Do not react with acid

Trends in the Periodic Table – Rows (“Periods”) tells you how many orbits/shells that element has – Column (“Families”) Tells you how many valence electrons that element has note: transition metals are exception to the rule
Trends

Chemical Families • Alkali Metals – 1 st column (Li, Na, K, etc. ) – Shiny silvery metals, make soluble compounds • Alkaline Earth Metals – 2 nd column (Mg, Ca, Ba, etc. ) – Shiny silvery metals, make non-soluble compounds

Chemical Families • Halogens – 7 th column (F, Cl, Br, etc. ) – Poisonous and reactive. • Noble Gases – 8 th column (He, Ne, etc. ) – Tend not to form compounds. Not reactive.

Look at periodic table 1) How many electrons total do these elements have? a) Li b) N c) Cl 3 5 17 2) How many valence electrons do these elements have? a) O b) Be c) Al d) Ar 6 4 3 8 3) How many shells/orbits do these elements have? a) Mg b) He c) P d) Ne 3 1 3 2

Forming Compounds

Forming Compounds • Atoms are rarely in their ‘stable’ form – Only the noble gases. • Usually atoms obtain a full valence shell by bonding (gaining and loseing e-) • Atoms bond to other atoms to make Molecules (a. k. a. Compounds)
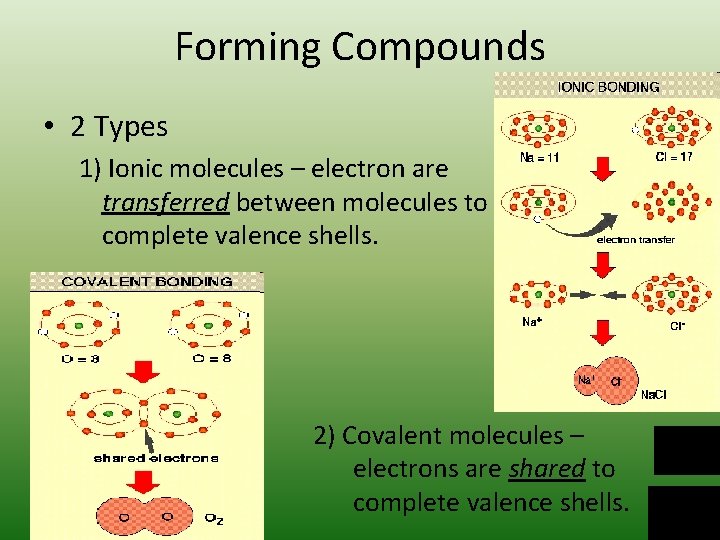
Forming Compounds • 2 Types 1) Ionic molecules – electron are transferred between molecules to complete valence shells. 2) Covalent molecules – electrons are shared to complete valence shells.
Ionic Bonding

Ionic Compounds • Between a metal and a non-metal – Ex. Na. Cl, Mg. Cl 2 , Al. F 3 • Electrons are transferred from the metal to the non-metal. • “Ions” (charged atoms) become formed. • Ions attract each other and are ‘bonded’ together forming IONIC BONDS.
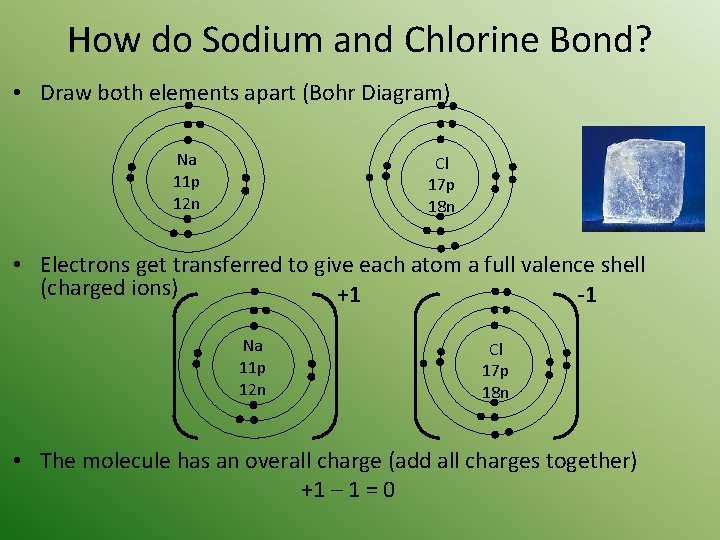
How do Sodium and Chlorine Bond? • Draw both elements apart (Bohr Diagram) Na 11 p 12 n Cl 17 p 18 n • Electrons get transferred to give each atom a full valence shell (charged ions) +1 -1 Na 11 p 12 n Cl 17 p 18 n • The molecule has an overall charge (add all charges together) +1 – 1 = 0
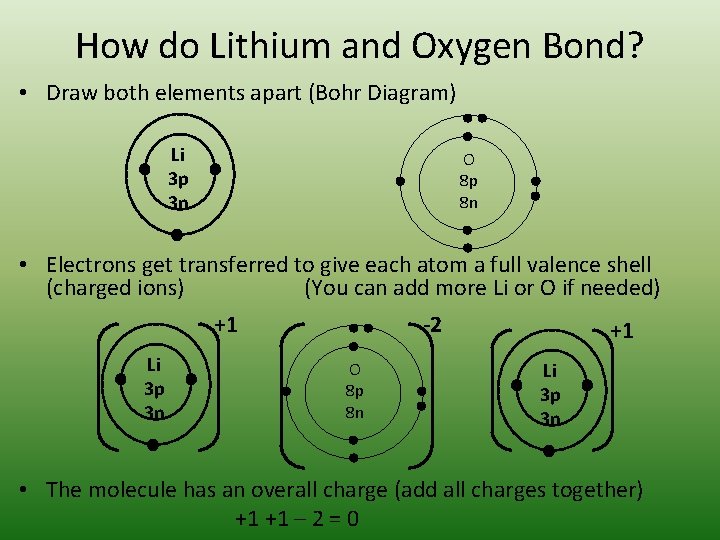
How do Lithium and Oxygen Bond? • Draw both elements apart (Bohr Diagram) Li 3 p 3 n O 8 p 8 n • Electrons get transferred to give each atom a full valence shell (charged ions) (You can add more Li or O if needed) +1 -2 +1 Li 3 p 3 n O 8 p 8 n Li 3 p 3 n • The molecule has an overall charge (add all charges together) +1 +1 – 2 = 0

How do Calcium and Chlorine Bond? • Draw both elements apart (Bohr Diagram) • Electrons get transferred to give each atom a full valence shell (charged ions) (You can add more Ca or Cl if needed) • The molecule has an overall charge (add all charges together)

Try Page 189 #1 -4
- Slides: 17