THE PERIODIC TABLE CHAPTER 7 ARRANGING THE ELEMENTS

THE PERIODIC TABLE CHAPTER 7

ARRANGING THE ELEMENTS Chapter 7 - Section 1

ORGANIZATION • If you walk into a market trying to find milk, where do you look for it? ? - the dairy section of course!! • Now imagine if the market or any other store was not organized by categories how long it would take you to locate things!! • Well… in the 1860’s scientists had the same problem – they have about 60 known elements but no way of organizing them

DISCOVERING A PATTERN • Dmitri Mendeleev first discovered a pattern to the elements in 1869 • He first organized the elements by similar properties physical & chemical properties like density, melting points, appearance & reactivity. • Mendeleev noticed: • fluorine & chlorine are both gases at room temp & they irritate the lungs • silver & copper are both shiny & tarnish in air • Li, Na, & K all react explosively with water
PERIODIC PROPERTIES OF THE ELEMENTS • Finally Mendeleev ranked the elements by increasing atomic mass – a pattern emerged! • He noticed there was a repeating pattern in a “periodic” way with increasing the atomic mass • Just like a calendar – the week repeats every 7 days • He used his table to predict that elements with certain properties would be discovered later.
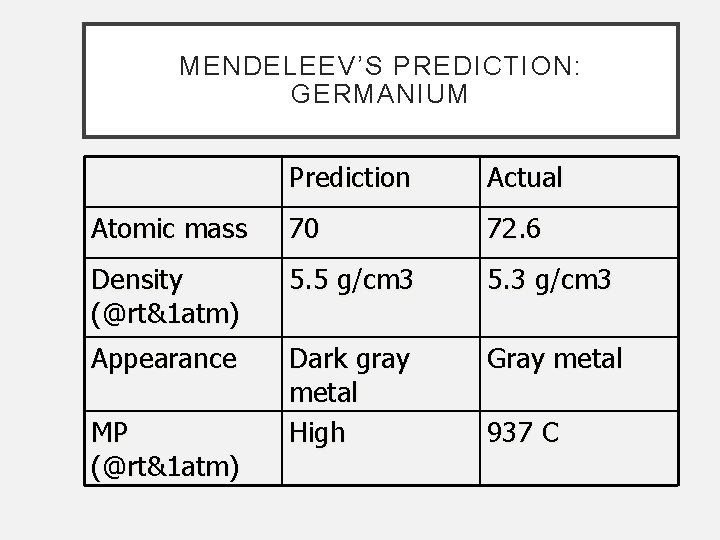
MENDELEEV’S PREDICTION: GERMANIUM Prediction Actual Atomic mass 70 72. 6 Density (@rt&1 atm) 5. 5 g/cm 3 5. 3 g/cm 3 Appearance Dark gray metal High Gray metal MP (@rt&1 atm) 937 C

CHANGING THE ARRANGEMENT • Henry Moseley determined the number of protons—the atomic number—in an atom. He then rearranged the periodic table by placing the elements in order of increasing atomic number. • He used the wavelengths of x rays emitted from heavier elements to determine how many protons they had in their nuclei.
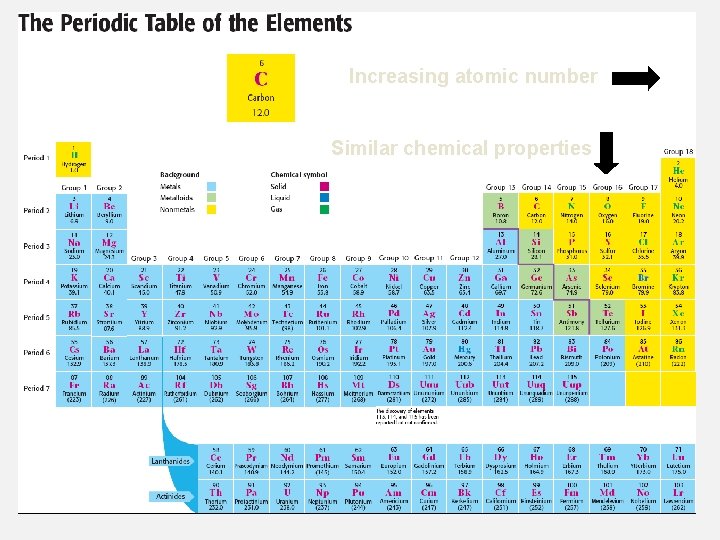
Increasing atomic number Similar chemical properties
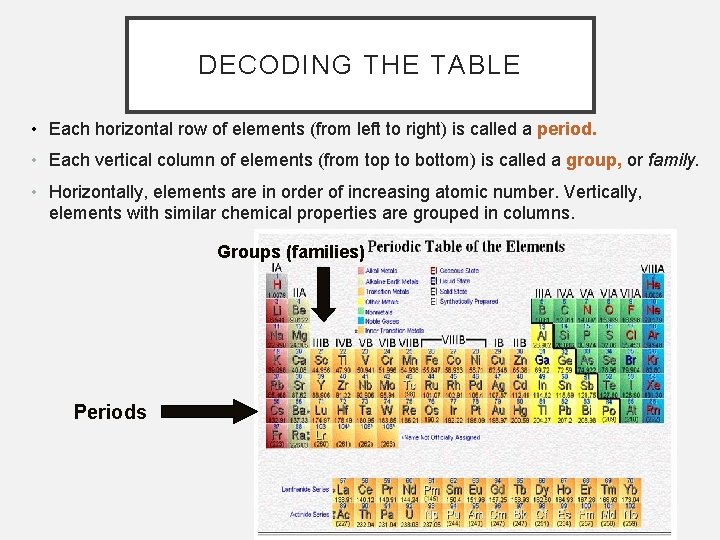
DECODING THE TABLE • Each horizontal row of elements (from left to right) is called a period. • Each vertical column of elements (from top to bottom) is called a group, or family. • Horizontally, elements are in order of increasing atomic number. Vertically, elements with similar chemical properties are grouped in columns. Groups (families) Periods

THE PERIODIC LAW • The periodic law states that the repeating chemical and physical properties of elements change periodically with the atomic numbers of the elements. • All of the elements follow the periodic law.

GROUPING THE ELEMENTS Chapter 7 Section 2

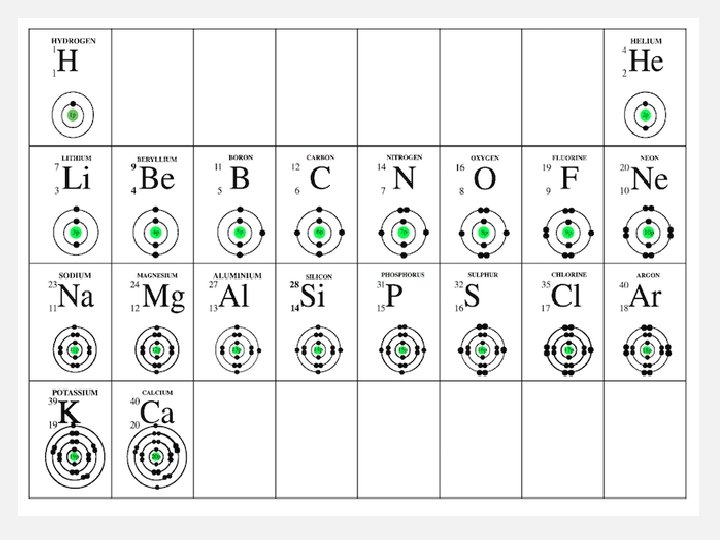
GROUPS/FAMILIES • The elements in a group have similar properties because the atoms of the elements have similar numbers of valence electrons (electrons in their outer energy level). • In order to have a complete set of electrons in their outer level, atoms will often take, give, or share electrons with other atoms. Elements whose atoms undergo such processes are called reactive.

REACTIVITY • As a group metals easily lost electrons to become positively charged ions (cations). • Both nonmetals and metalloids generally take or share electrons, making them negatively charged ions (anions).

GROUP 1: ALKALI METALS • Alkali metals are elements in Group 1 of the periodic table. • Most reactive metals - their atoms easily lose the one electron in the outer level of their atoms. • So reactive that in nature they are found only combined with other elements • Other characteristics: soft, shiny, silver color, low density

GROUP 2: ALKALINE-EARTH METALS • Alkaline-earth metals are elements in Group 2. They are less reactive than alkali metals are, but more reactive than other metals. • Atoms of alkaline-earth metals have two electrons in their outer level.
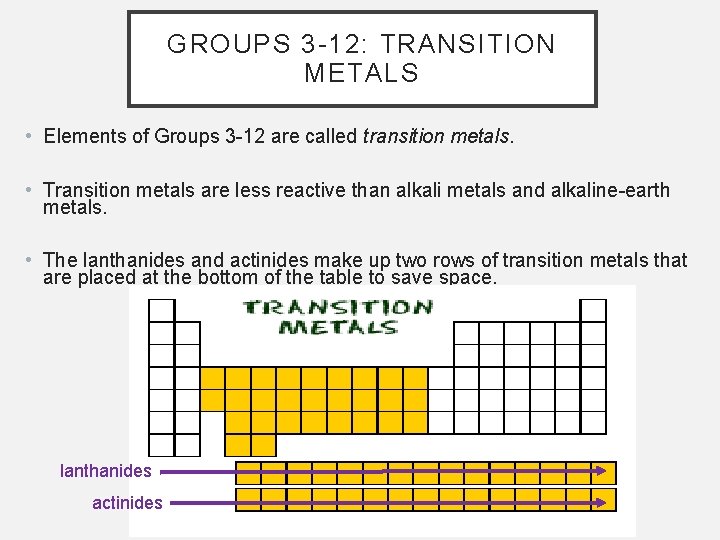
GROUPS 3 -12: TRANSITION METALS • Elements of Groups 3 -12 are called transition metals. • Transition metals are less reactive than alkali metals and alkaline-earth metals. • The lanthanides and actinides make up two rows of transition metals that are placed at the bottom of the table to save space. lanthanides actinides

GROUPS 3 -12: TRANSITION METALS, CONTINUED • Transition metals have 1 or 2 electrons in the outer level of their atoms. So, the properties of the transition metals vary. • But because these elements are metals, they share the properties of metals. • In general, they are also very similar to one another and can be confused.
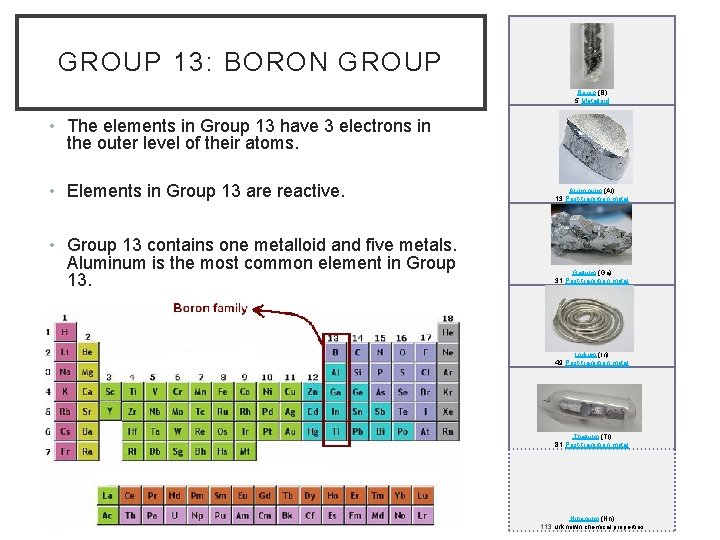
GROUP 13: BORON GROUP Boron (B) 5 Metalloid • The elements in Group 13 have 3 electrons in the outer level of their atoms. • Elements in Group 13 are reactive. • Group 13 contains one metalloid and five metals. Aluminum is the most common element in Group 13. Aluminium (Al) 13 Post-transition metal Gallium (Ga) 31 Post-transition metal Indium (In) 49 Post-transition metal Thallium (Tl) 81 Post-transition metal Nihonium (Nh) 113 unknown chemical properties

Carbon (C) 6 Polyatomic nonmetal Silicon (Si) 14 Metalloid GROUP 14: CARBON GROUP • The elements in Group 14 have 4 electrons in the outer level of their atoms. Their reactivity varies. • Group 14 contains metals, nonmetals, and metalloids. Germanium (Ge) 32 Metalloid • Diamond, graphite, and soot are all natural forms of the nonmetal carbon. Tin (Sn) 50 Post-transition metal Lead (Pb) 82 Post-transition metal Flerovium (Fl) 114 Unknown chemical properties

GROUP 15: NITROGEN GROUP Nitrogen (N) 7 Diatomic nonmetal • The elements in Group 15 have 5 electrons in the outer level of their atoms. Their reactivity varies. Phosphorus (P) 15 Polyatomic nonmetal • Group 15 contains metals, nonmetals, and metalloids. Arsenic (As) 33 Metalloid Antimony (Sb) 51 Metalloid Bismuth (Bi) 83 Post-transition metal Moscovium (Mc) 115 unknown chemical properties
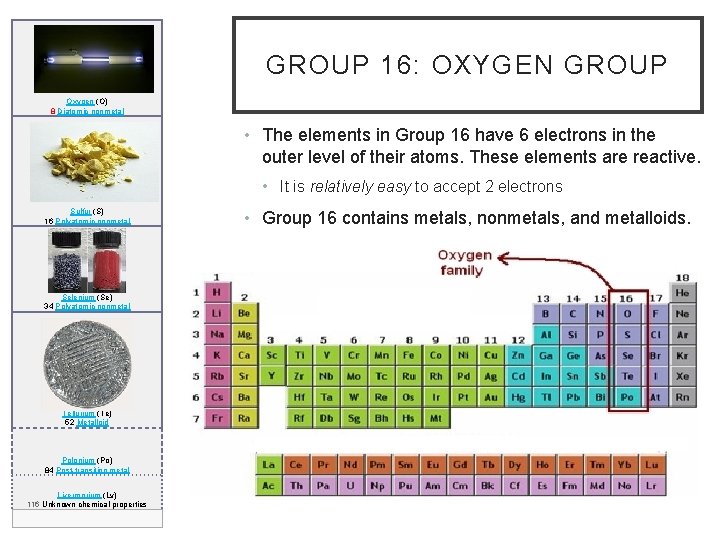
GROUP 16: OXYGEN GROUP Oxygen (O) 8 Diatomic nonmetal • The elements in Group 16 have 6 electrons in the outer level of their atoms. These elements are reactive. • It is relatively easy to accept 2 electrons Sulfur (S) 16 Polyatomic nonmetal Selenium (Se) 34 Polyatomic nonmetal Tellurium (Te) 52 Metalloid Polonium (Po) 84 Post-transition metal Livermorium (Lv) 116 Unknown chemical properties • Group 16 contains metals, nonmetals, and metalloids.
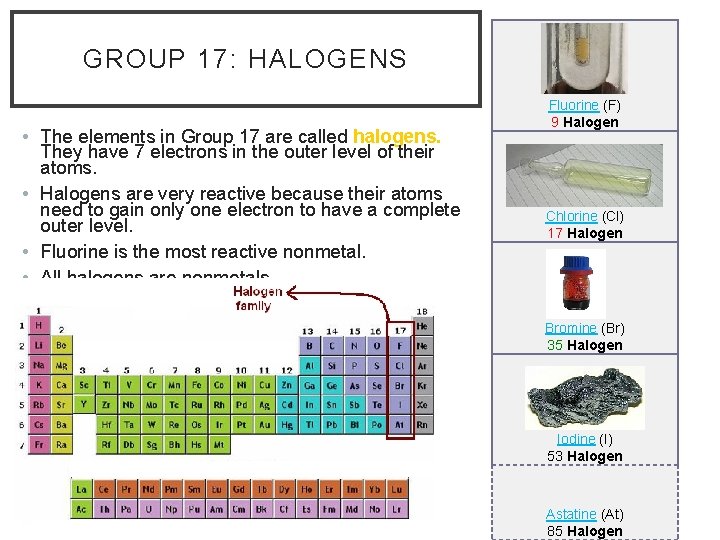
GROUP 17: HALOGENS • The elements in Group 17 are called halogens. They have 7 electrons in the outer level of their atoms. • Halogens are very reactive because their atoms need to gain only one electron to have a complete outer level. • Fluorine is the most reactive nonmetal. • All halogens are nonmetals. • Most halogens are toxic to humans. Fluorine (F) 9 Halogen Chlorine (Cl) 17 Halogen Bromine (Br) 35 Halogen Iodine (I) 53 Halogen Astatine (At) 85 Halogen

GROUP 18: NOBLE GASES Helium (He) 2 Neon (Ne) 10 Argon (Ar) 18 • The elements in Group 18 are called noble gases. Atoms of noble gases have a full set of electrons in their outer level. • Helium has 2 valence electrons, all others have 8 valence electrons • Noble gases are relatively unreactive. These elements were first called inert gases. • All noble gases are nonmetals. Krypton (Kr) 36 Xenon (Xe) 54 Radon (Rn) 86 Oganesson (Og) 118

HYDROGEN • The properties of hydrogen do not match the properties of any single group, so hydrogen is set apart in the table. • Hydrogen has one electron in the outer level of its atoms. Hydrogen is reactive. • Hydrogen is the most abundant element in the universe.
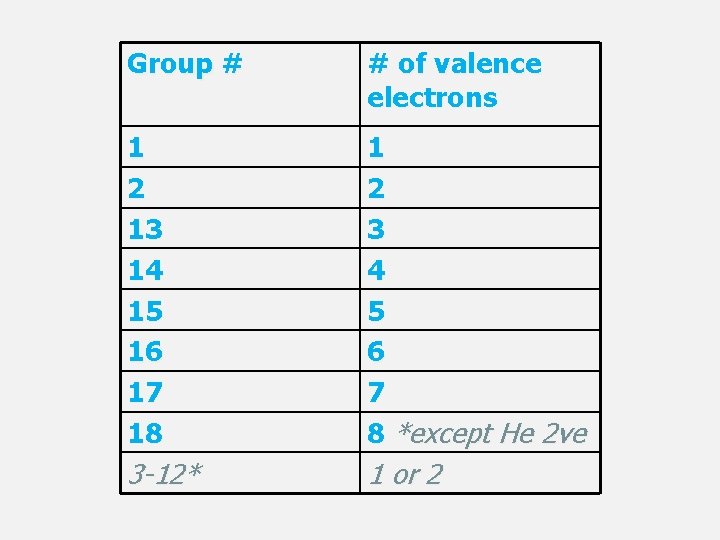
Group # # of valence electrons 1 2 13 14 15 16 17 18 1 2 3 4 5 6 7 8 *except He 2 ve 3 -12* 1 or 2
- Slides: 26