THE PERIODIC TABLE CHAPTER 6 Johann Wolfgang Dbereiner

THE PERIODIC TABLE CHAPTER 6

Johann Wolfgang Döbereiner (1780 – 1849) – German Chemist ØObserved that several elements could be organized into groups of threecalled “triads” Øelements of each triad shared similar properties

John Newlands (1837 – 1898) – English Chemist Øarranged table according to atomic mass Øobserved periodicity of the elements Ølaw of octaves: properties of elements repeat every eighth element

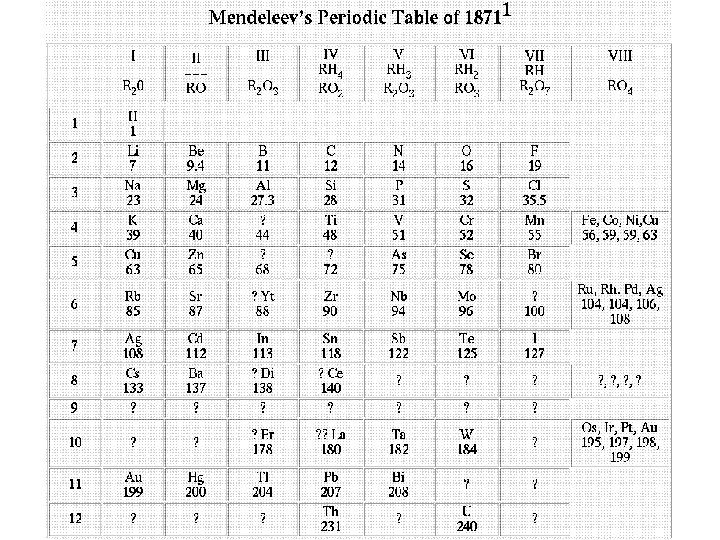
Dmitri Mendeleev (1834 – 1907) – Russian Chemist ØPublished first table that is the basis for today’s periodic table Øarranged elements by increasing atomic mass Øassigned elements with similar properties to the same columns Øpredicted existence of missing elements based on his table Øproblem: some elements did not fit according to their atomic mass and properties I/Te, Co/Ni, Ar/K

Henry Moseley (1887 – 1915) – English Physicist Ø developed idea of atomic number Øused x-rays to determine the atomic number of elements Øcorrected Mendeleev’s table by arranging elements according to atomic number, not atomic mass Øfounded modern periodic law: when elements are arranged in order of increasing atomic number, their physical and chemical properties show a periodic pattern

Glenn Seaborg (1912 – 1999) – American Chemist ØBorn in Ishpeming, Michigan (Upper Peninsula) ØWon the Nobel Prize in Chemistry in 1951 ØDeveloped the actinide concept, which led to the arrangement of the actinide series on the periodic table.

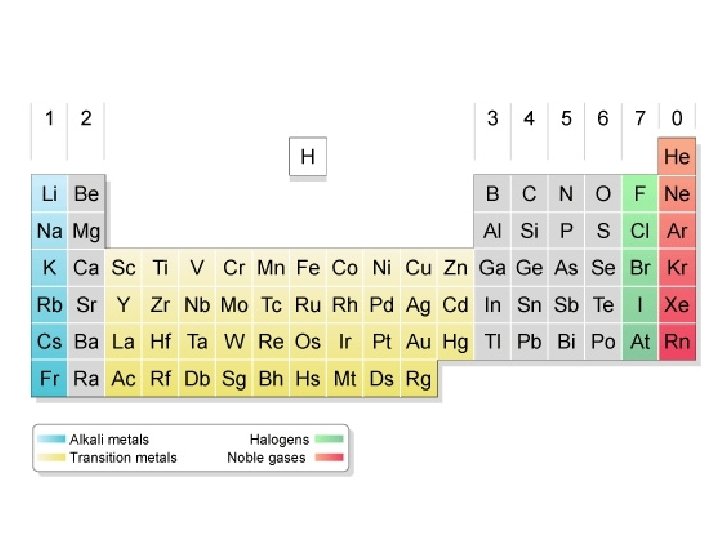
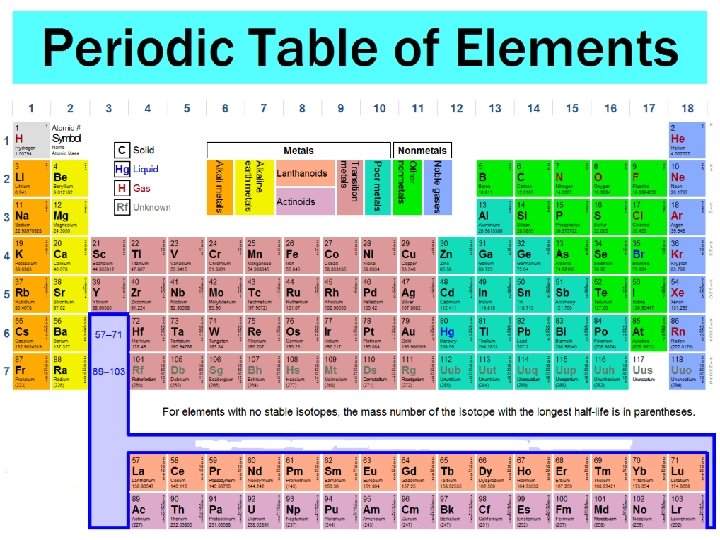
B. Reading the Table 1. Be familiar with the location of: a. Alkali metals b. Alkaline earth metals c. Metalloids d. Transition metals e. Main-group elements f. Halogens g. Noble gases h. s, p, d, f blocks i. Horizontal rows- period or series j. Vertical columns- group or family k. Most probable charge and outer shell configuration
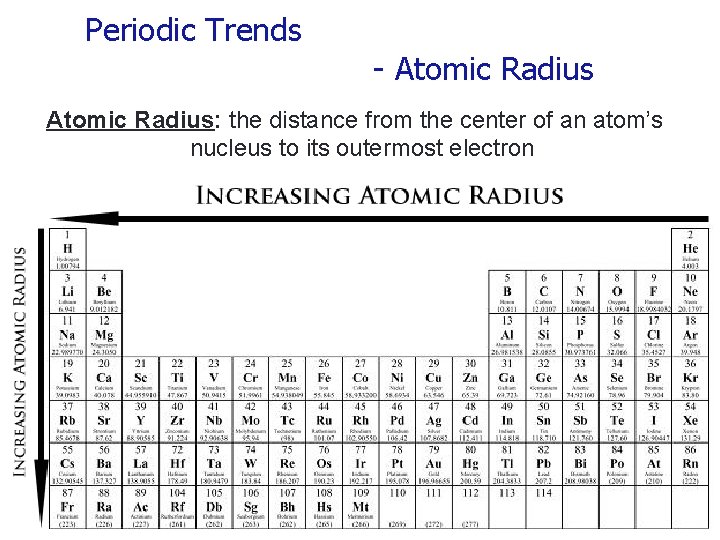
Periodic Trends - Atomic Radius: the distance from the center of an atom’s nucleus to its outermost electron

Periodic Trends NO CLEAR TREND If you remove an electron Øthe ionic radius decreases Øthe nucleus has to attract fewer electrons – Ionic Radius

Periodic Trends NO CLEAR TREND If you add an electron Øthe ionic radius increases Øthe nucleus has to attract more electrons Øfull outer shell results in increased repulsion among electrons – Ionic Radius

Periodic Trends – Ionization Energy – the energy required to remove an electron

Periodic Trends – Electronegativity – an atom’s ability to attract electrons in a chemical bond

Periodic Trends – Electron affinity – the energy change that occurs when an electron is acquired by a neutral atom
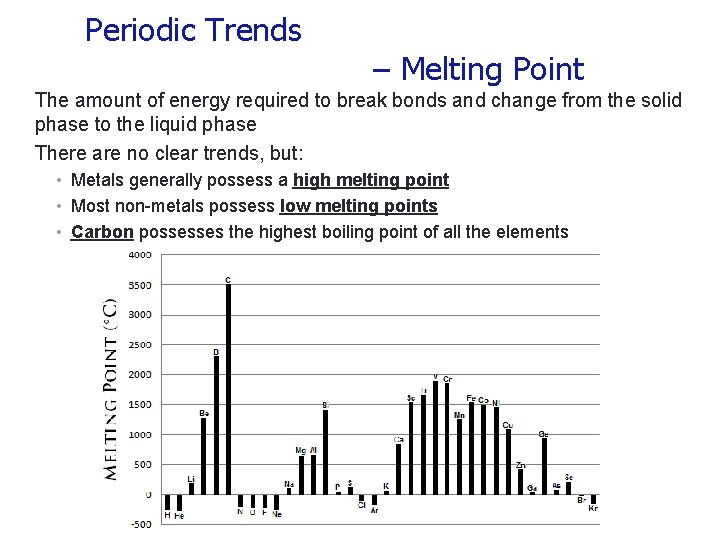
Periodic Trends – Melting Point The amount of energy required to break bonds and change from the solid phase to the liquid phase There are no clear trends, but: • Metals generally possess a high melting point • Most non-metals possess low melting points • Carbon possesses the highest boiling point of all the elements
- Slides: 17