The Periodic Table Ch 6 CVHS Chemistry History
The Periodic Table Ch 6 CVHS Chemistry

History of Periodic Table Development By 1860 scientists had discovered 60 elements & their atomic masses ◦ Gave each group w/ similar properties a name lithium, sodium, and potassium were known as the alkali metals 1829: Döbereiner developed triads ◦ Groups of 3 elements w/ similar properties organized by atomic mass vertically Density increased w/ atomic mass Dmitri Mendeleev ◦ Later organized elements horizontally by atomic mass & noticed that the properties of the elements followed a pattern across the rows. Periodicity intervals) (tendency to occur @ regular ◦ Mendeleev’s periodic table demonstrated the periodicity of the elements.

Mendeleev’s Periodic Table Organized by atomic mass, chemical & physical properties of elements. Correctly predicted properties of unknown elements & left blank spaces on his table. Was certain about the properties of elements determining where they would be placed moved elements away from where the atomic mass would predict. Te & I are not in order by atomic mass, they are ordered by physical & chemical properties (& in today’s table Atomic # as well)

Modern Periodic Table Organized by Atomic Number, not Atomic mass Atomic # = protons Atomic # increases as you move across Each row (except the first) begins with a metal and ends with a noble gas The properties of elements change in an orderly progression from left to right: periodicity The pattern in properties repeats after column 18

Periodic Law The statement that the physical and chemical properties of the elements repeat in a regular pattern when they are arranged in order of increasing atomic number is known as the periodic law Atomic radius decreases as you move across the table.
Atomic Structure & Periodic Table As atomic # increases across the periodic table so do the # of electrons The chemical behavior of the elements is determined by the number of electrons in their valence shell (s &

Atomic Structure & Periodic Table Groups or families ◦ Vertical columns, have same valence electrons Periods ◦ Horizontal rows, atomic # increases by one as you go across Valence Electrons ◦ The electrons in the outermost s & p sublevels. ◦ Available for reacting ◦ Elements take or release electrons to make their outermost shell stable.
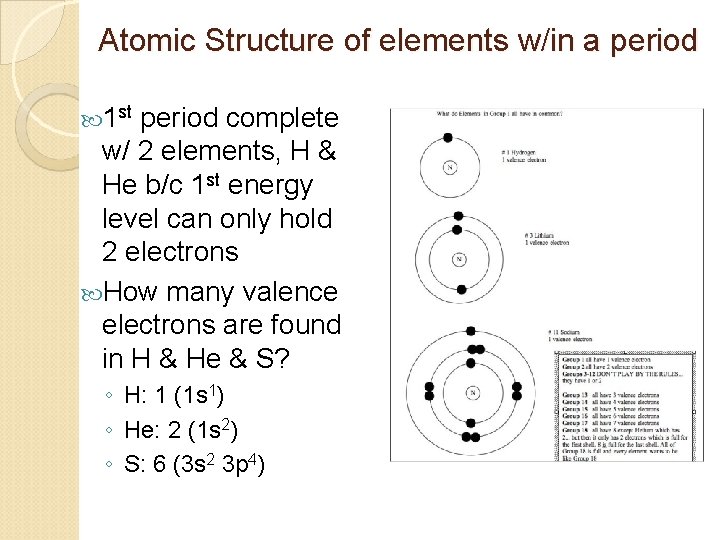
Atomic Structure of elements w/in a period 1 st period complete w/ 2 elements, H & He b/c 1 st energy level can only hold 2 electrons How many valence electrons are found in H & He & S? ◦ H: 1 (1 s 1) ◦ He: 2 (1 s 2) ◦ S: 6 (3 s 2 3 p 4)

Atomic Structure w/in a group The number of valence electrons changes from one to eight as you move from left to right across a period; when you reach Group 18, the pattern repeats. The main group elements are those in Groups 1, 2, 13, 14, 15, 16, 17, and 18 ◦ Group # is related to # of electrons for the main groups For elements in Groups 1 and 2, the group number equals the number of valence electrons For elements in Groups 13, 14, 15, 16, 17, and 18, the second digit in the group number is equal to the number of valence electrons
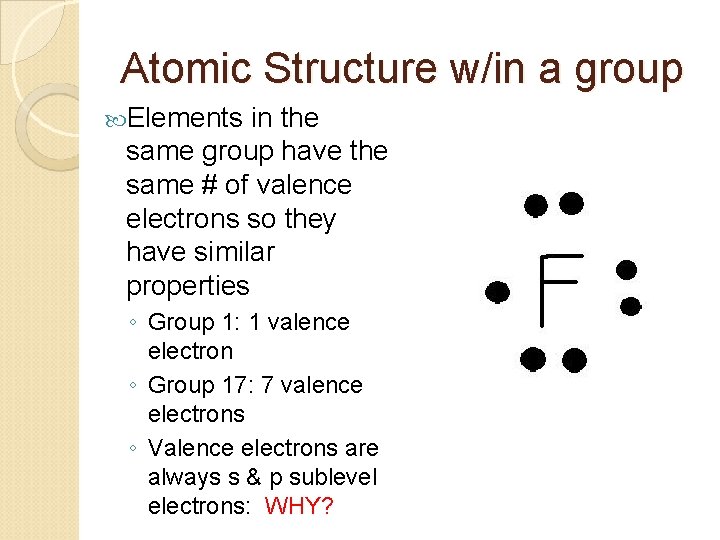
Atomic Structure w/in a group Elements in the same group have the same # of valence electrons so they have similar properties ◦ Group 1: 1 valence electron ◦ Group 17: 7 valence electrons ◦ Valence electrons are always s & p sublevel electrons: WHY?

Commonly Used Group Names Alkali metals in Group 1 ◦ Form bases w/ non metals Alkali means basic Alkaline earth metals in Group 2, Halogens in Group 17 ◦ Means salt, b/c they form saltlike compounds w/ metals Noble gases in Group
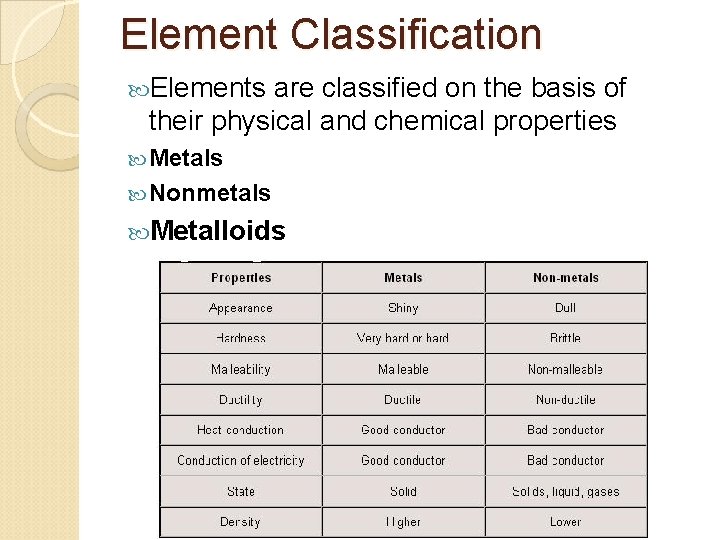
Element Classification Elements are classified on the basis of their physical and chemical properties Metals Nonmetals Metalloids

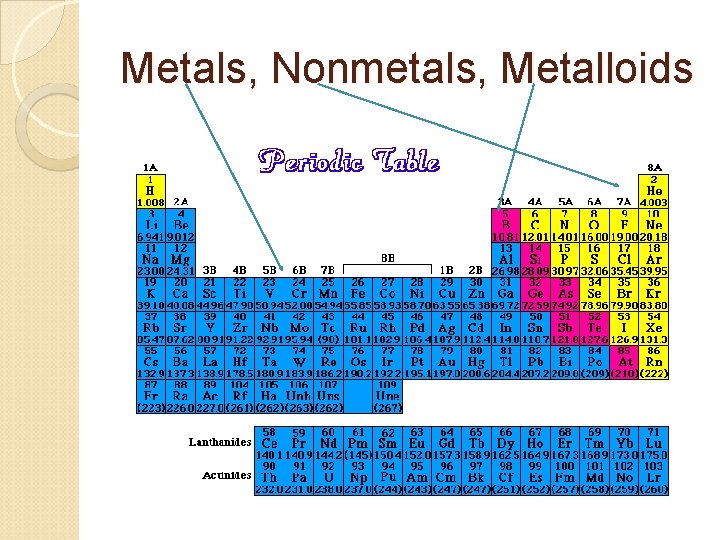
Metals Luster, conduct heat & electricity & bend, most are solids @ room temp w/ high melting points, typically not main group elements Groups 1 & 2 are metals & some of groups 13 -16 are metals Groups 3 -12 are transition elements, all are metals Inner Transition Elements (58 -71 & 90 -103), all are metals

Nonmetals Don’t conduct electricty, poor conductors of heat, low melting points, most are gases, solids lack luster, solids are brittle Have 5, 6, 7 or 8 valence electrons (except C has 4) Abundant in nature Carbon found in more compounds than any other elements


Metalloids Have some properties of metals & nonmetals Found on border between metals & nonmetals Some metalloids such as silicon, germanium (Ge), and arsenic (As) are semiconductors ◦ They conduct electricity better than a nonmetal but not as well as a metal ◦ Si made the computer
Metals, Nonmetals, Metalloids

Periodic Trends Atomic Radius ◦ Measure of the size of an atom ◦ Decreases as you move across the table More protons in the nucleus pull the electrons toward the center of the atom ◦ Increases as you move down the table Same # of valence electrons farther away from the nucleus & they are shielded by the inner shell electrons
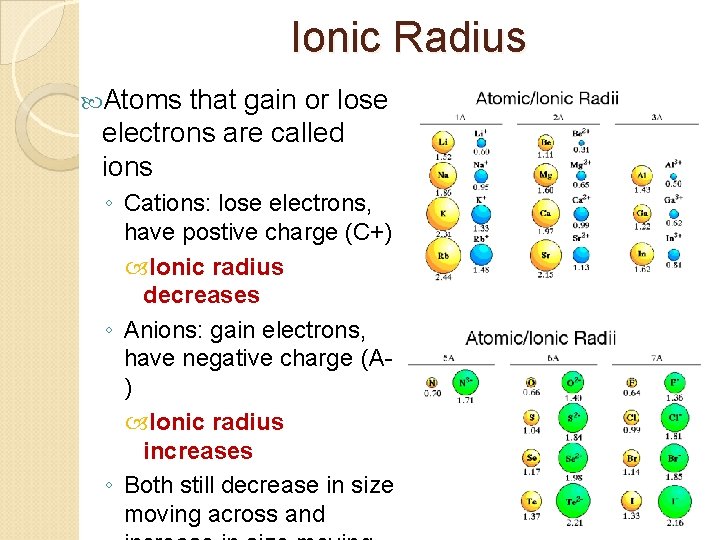
Ionic Radius Atoms that gain or lose electrons are called ions ◦ Cations: lose electrons, have postive charge (C+) Ionic radius decreases ◦ Anions: gain electrons, have negative charge (A) Ionic radius increases ◦ Both still decrease in size moving across and
Concept Check: Complete the Venn Metals Nonmetal s Metalloids
- Slides: 20