The Periodic Table and Periodic Law Chapter 6

The Periodic Table and Periodic Law Chapter 6 Honors Chemistry Glencoe

Section 6 -1: Development of the Modern Periodic Table I. History of the Periodic Table A. John Newlands – grouped elements into groups of 8, called the Law of Octaves B. Mendeleev and Meyer both grouped the elements with atomic mass and elemental properties.

1. 2. Mendeleev got the credit because he published first and demonstrated the usefulness of his PT. He arranged the elements in vertical columns based on their reactivity with other elements.

II. 3. He left some blank spaces in his table for undiscovered elements, but eventually, those elements were discovered. Modern Periodic Table A. Arranged according to atomic number (Moseley), NOT atomic mass. B. Periodic Law – elements are arranged according to repetitious properties.

1. Group – vertical column of elements; also called a chemical family. There are 18 groups. 2. Period/Shells/Energy Levels – horizontal row of elements; 7 periods on the table. Elements have strong resemblance among their members.
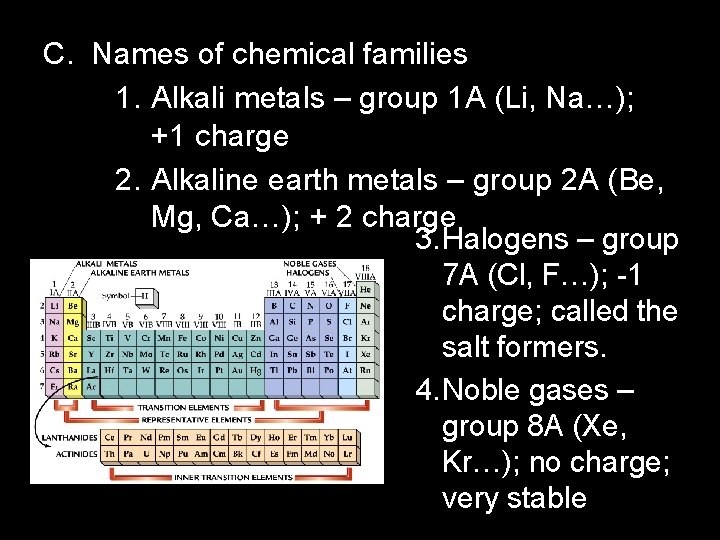
C. Names of chemical families 1. Alkali metals – group 1 A (Li, Na…); +1 charge 2. Alkaline earth metals – group 2 A (Be, Mg, Ca…); + 2 charge 3. Halogens – group 7 A (Cl, F…); -1 charge; called the salt formers. 4. Noble gases – group 8 A (Xe, Kr…); no charge; very stable

D. Periodic Table Subshells 1. Representative elements – s and p subshells 2. Transition metals – d subshell 3. Inner transition metals – f subshell

III. Metals, Nonmetals and Semi-metals A. Metals (left) 1. Have luster or shine 2. Good conductors of heat and electricity 3. Solid at room temperature (except mercury) 4. Malleable – thin sheets 5. Ductile – fine wire

B. Nonmetals 1. Lacks luster; dull 2. Poor conductors 3. Neither malleable nor ductile 4. Many are gases at RT (Bromine is a liquid) 5. Varied physical properties

C. Semimetals 1. Also called the metalloids 2. Properties intermediate of metals and nonmetals or some of each. 3. Properties not predictable; different for the different elements.

Section 6 -2: Classification of the Elements A. Type of electrons 1. Valence – outermost e- in the s and p blocks. How many valence e- are in [He] 2 s 2 2 p 5? What element is this? 2. Core – the remaining electrons B. Patterns in different groups 1. Alkali metals – ends with #s 1 2. Alkaline earth metals – ends with #s 2 3. Halogens - #p 5 4. Noble gases - #p 6
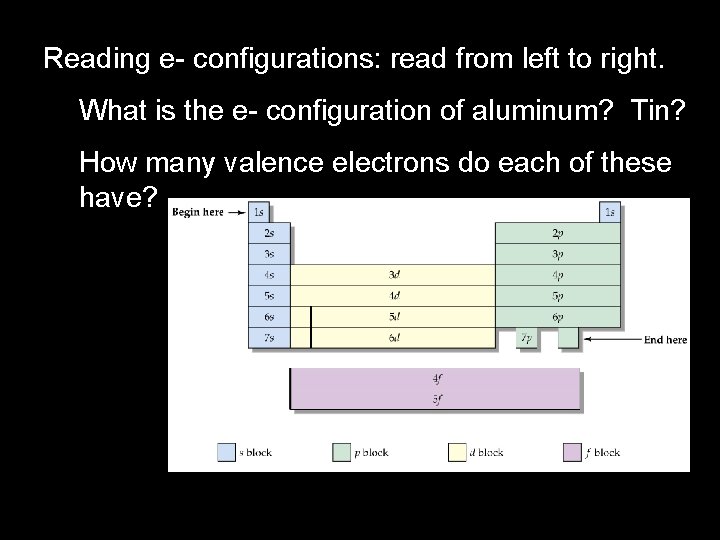
Reading e- configurations: read from left to right. What is the e- configuration of aluminum? Tin? How many valence electrons do each of these have?

C. Patterns in formation of ions 1. Atoms will tend to lose or gain electrons to become a stable noble gas. 2. Isoelectronic – elements that have the same e- configuration. Example: Chlorine ion and argon.

Sec 6 -3: Periodic Trends I. Atomic and Ionic Radii A. Atomic radius – ½ the distance from 2 nuclei Increasing atomic radius
B. Ionic Radius – ½ the distance from 2 nuclei of 2 ions

II. Electronegativity A. The attraction an atom has for a shared pair of electrons in a bond (pull on electrons). B. Increases going up and right on the PT.

III. Ionization Energy: energy required to remove 1 mole of e- from 1 mole of neutral gaseous atoms. A. Can predict which atoms will form certain ions. B. General Trend:

C. Successive Ionization Energy: energy used to remove successive e- (2 nd, 3 rd, etc. ) 1. Depends on the ions that the atom tends to form. 2. Magnesium: The IE to remove 1 st and the 2 nd will be low (easy to remove). The 3 rd IE is high because it already has a noble gas configuration 3. Which of the elements would you expect to have the highest 1 st IE?
- Slides: 18