The Periodic Table 6 1 Mendeleevs Period Table

The Periodic Table 6. 1

Mendeleev’s Period Table • Set up by atomic mass v. atomic number-modern • Elements in groups have similar properties
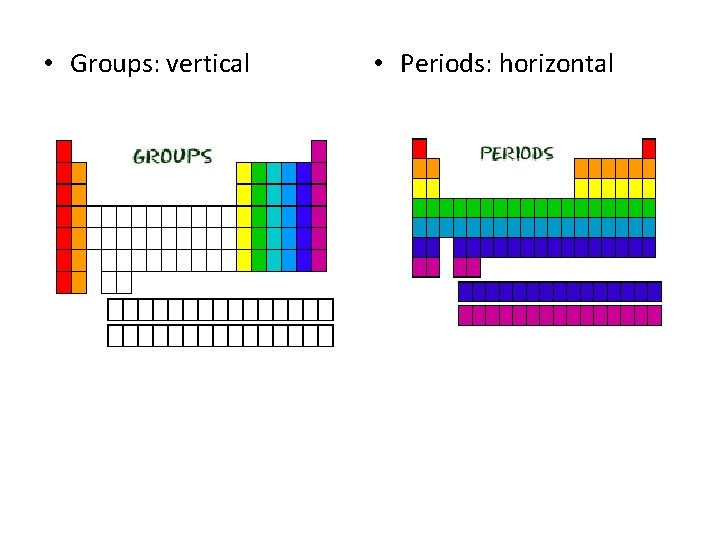
• Groups: vertical • Periods: horizontal
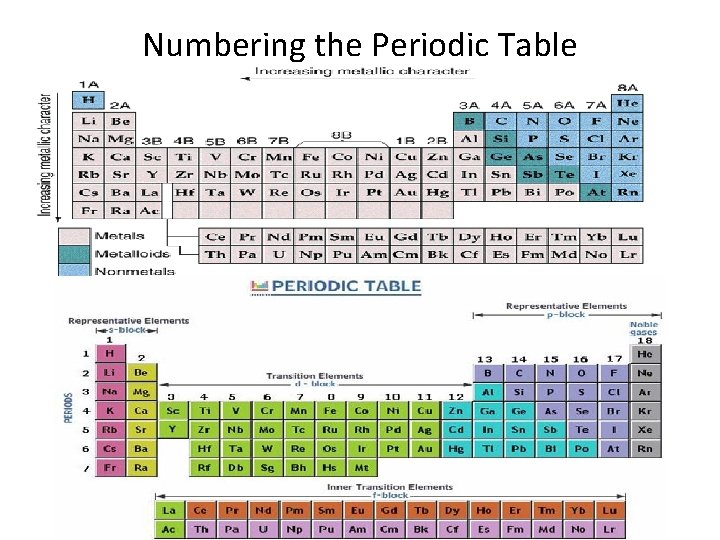
Numbering the Periodic Table
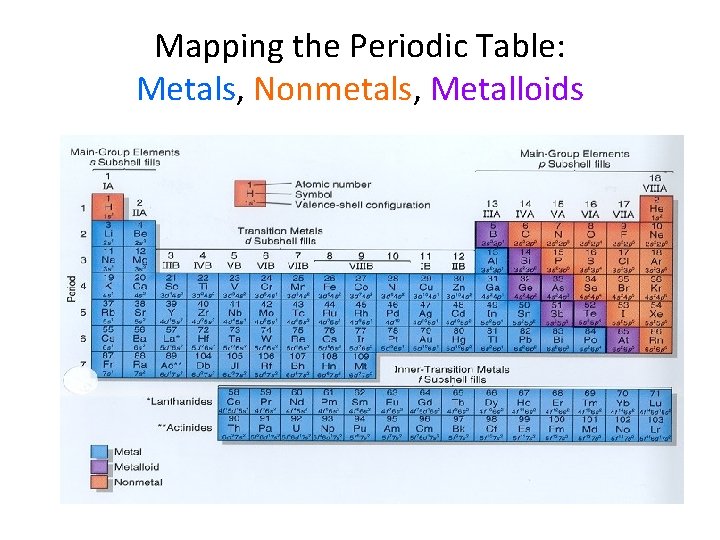
Mapping the Periodic Table: Metals, Nonmetals, Metalloids

Metal Properties 1. 2. 3. 4. 5. High luster or sheen – reflects light Conducts electricity Conducts heat Ductile (can be drawn into wires) Malleable (can be hammered into sheets - bends) 6

Non metals Properties 1. 2. 3. 4. Dull – no sheen Insulator – doesn’t conduct heat or electricity Brittle – will shatter not hammer into sheets Most are gases at room temperature – P, S, Se, I are solid – Br is liquid 7

Metalloid (semimetal) Properties • Has characteristics of both metals and nonmetals 1. Tend to be semi-conductors (conduct electricity under right conditions. 2. Can be shiny (have a sheen) 3. Can be brittle 8

Extended Periodic Table
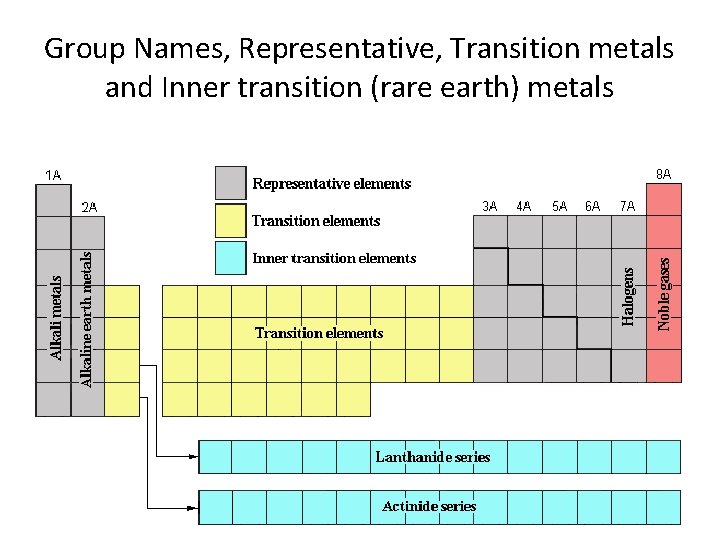
Group Names, Representative, Transition metals and Inner transition (rare earth) metals

Ch. 6. 2 and 6. 3 • Electron configuration • Ions and charges • Periodic Trends
Electron configuration on the Periodic Table
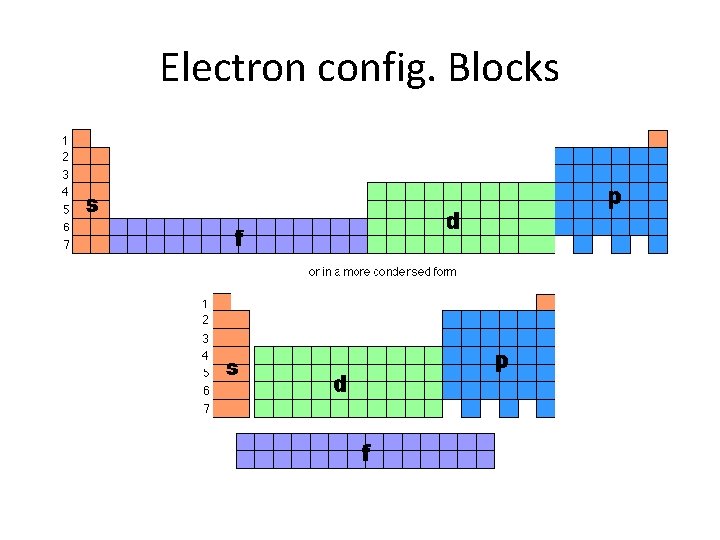
Electron config. Blocks
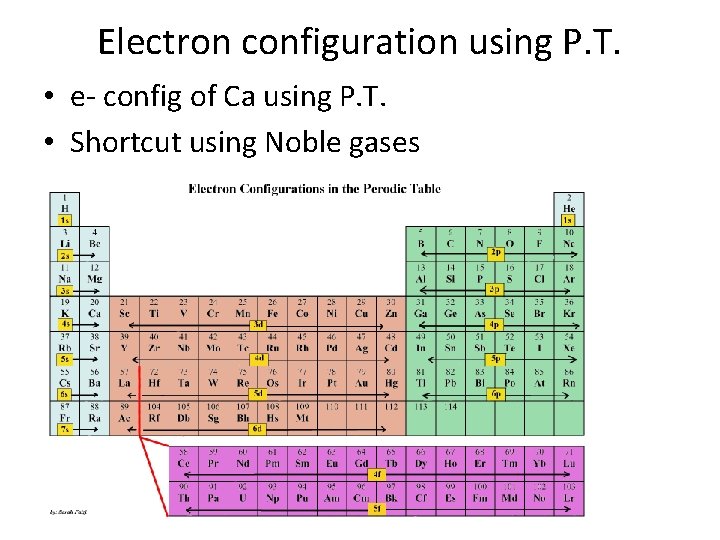
Electron configuration using P. T. • e- config of Ca using P. T. • Shortcut using Noble gases

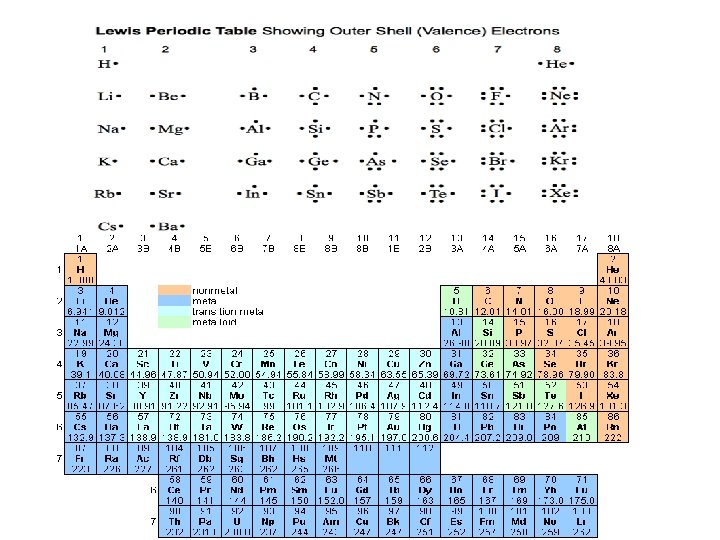
Valence electrons • Valence Electrons (v. e. ): – electrons used in bonding elements – Electrons in the highest energy level • s and p sublevels – maximum is 8 – Valence electrons is the same as the group number (for representative elements) • Example: Mg is group 2 A, has 2 v. e.

Valence electrons in P. T.

Ions • Atoms gain or lose electrons to obtain a noble gas configuration – Cations: positive charge bc they LOSE electrons. • Ca: loses 2 e- becomes +2 charge to be like Ar (Ca+2) – Anions: negative charge bc they GAIN electrons • Cl: gains 1 e- be come -1 charge to be like Ar (Cl -)
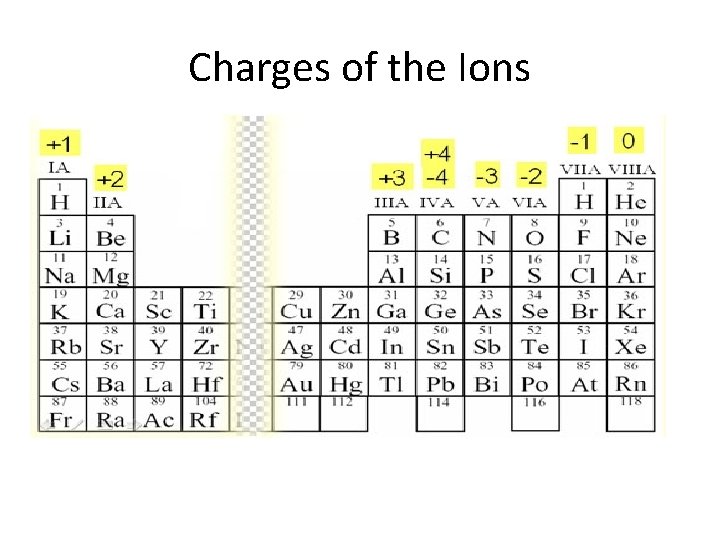
Charges of the Ions

Ch. 6. 3 Definitions • Atomic Size (radius): one half the distance between the nuclei of 2 atoms of the same element when 2 atoms are joined. – Ions: atom (or group of atoms) that has a positive or negative charge by losing or gaining electrons. • Anions: larger than original atom • Cations: smaller than original atom
Periodic Table Trends
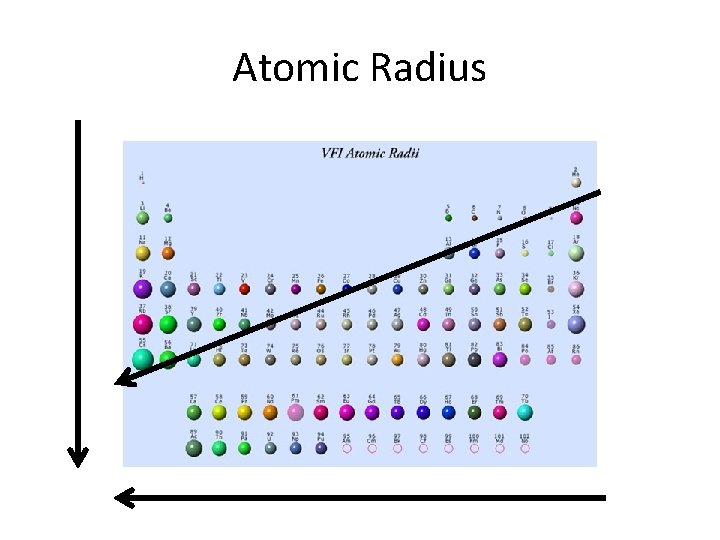
Atomic Radius

Vocabulary • Ionization Energy: energy required to remove the first electron from an atom. • Electronegativity: the ability of an atom to attract an electron when an atom is in a compound. (number value assigned arbitrarily 0. 7 - 4)
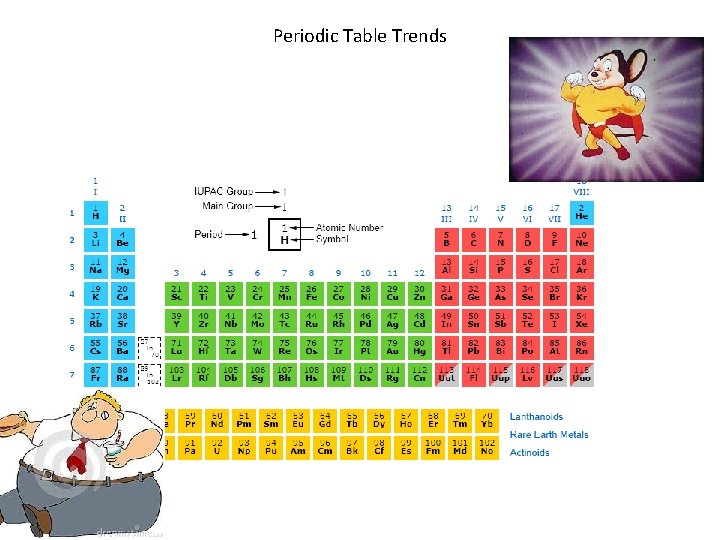
Periodic Table Trends
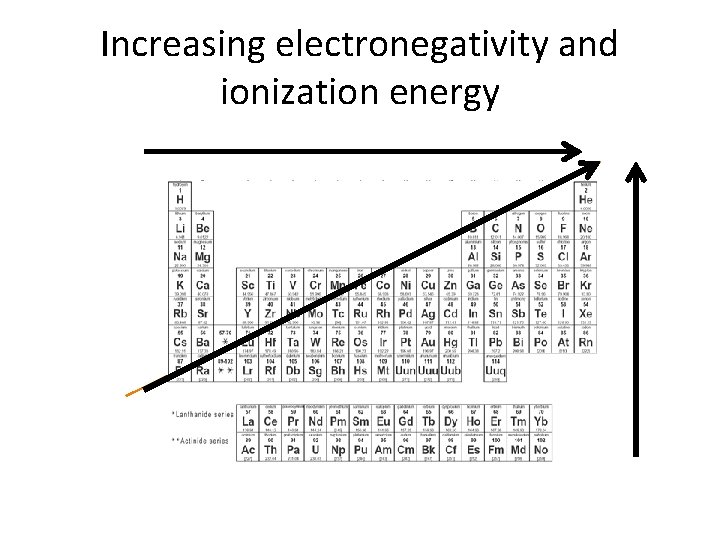
Increasing electronegativity and ionization energy
- Slides: 25