The Periodic Table 1 Electronic Structures of Atoms
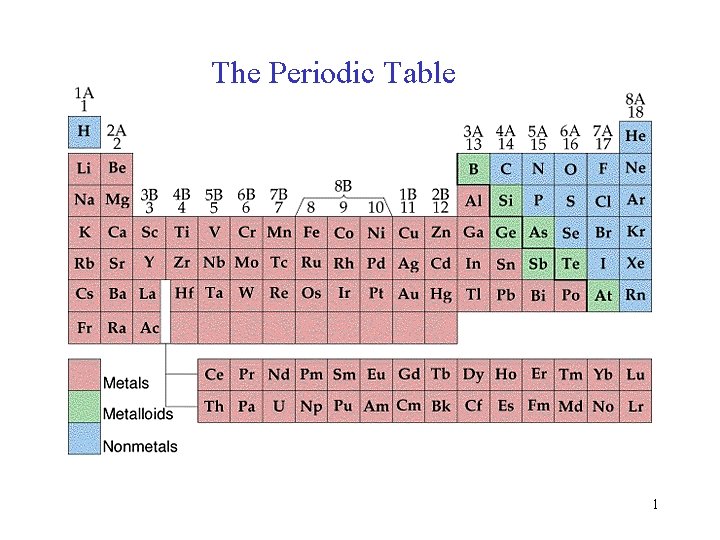
The Periodic Table 1
Electronic Structures of Atoms Where are the e-’s located and how are they arranged? 2

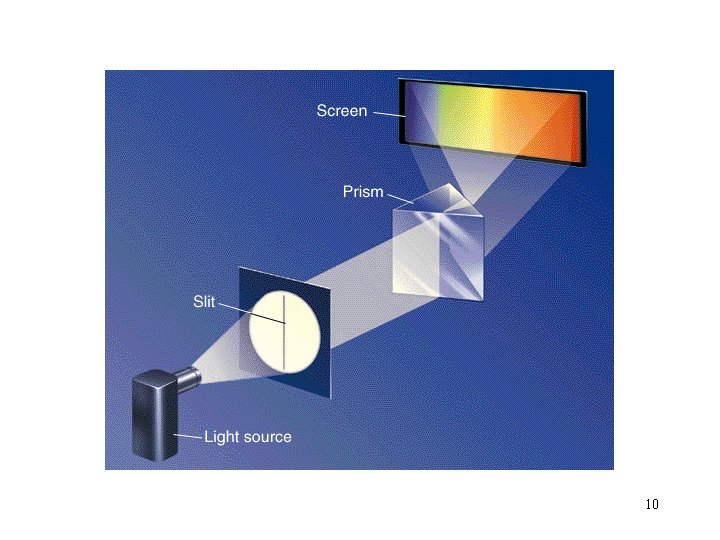
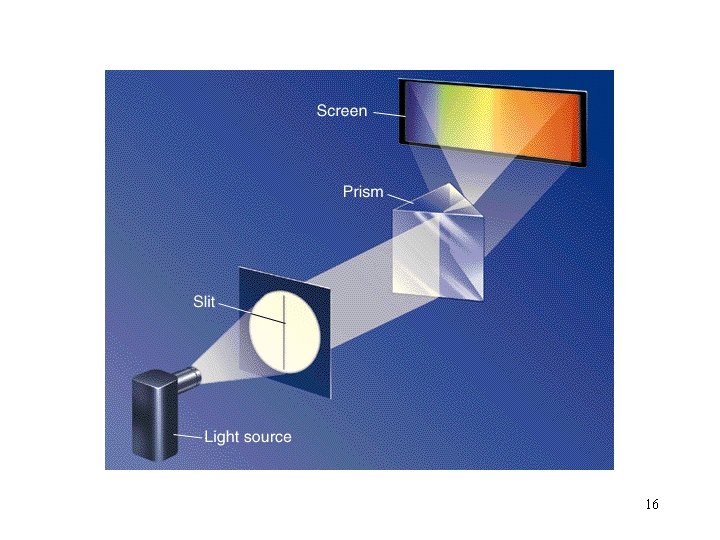
How can we probe an atom to find out? “Let’s use light. ” 3
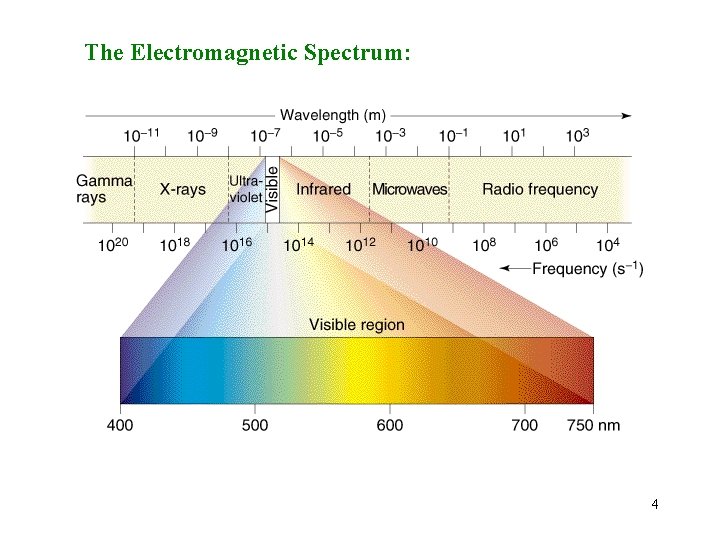
The Electromagnetic Spectrum: 4
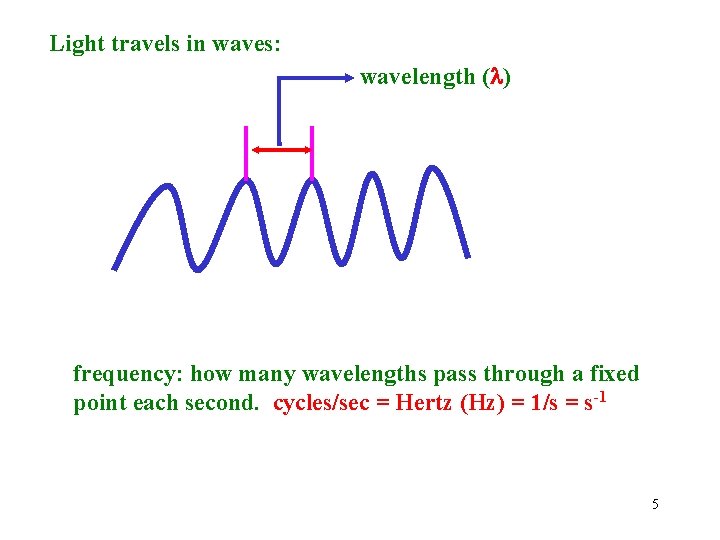
Light travels in waves: wavelength ( ) frequency: how many wavelengths pass through a fixed point each second. cycles/sec = Hertz (Hz) = 1/s = s-1 5
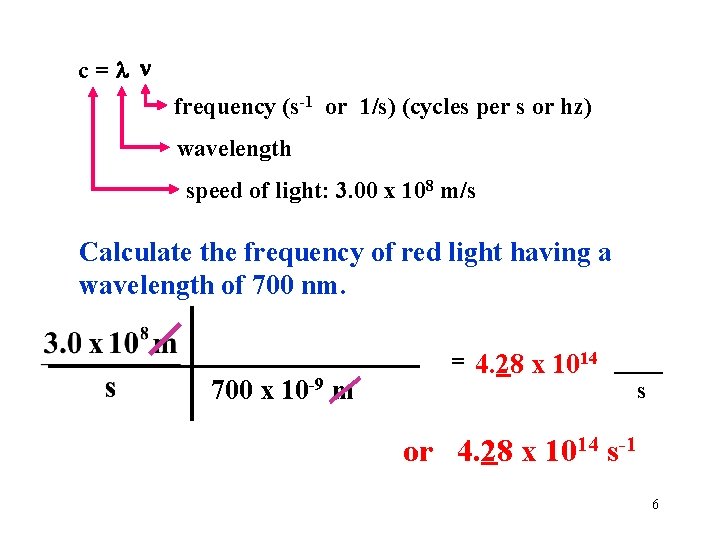
c= frequency (s-1 or 1/s) (cycles per s or hz) wavelength speed of light: 3. 00 x 108 m/s Calculate the frequency of red light having a wavelength of 700 nm. 700 x 10 -9 m = 4. 28 x 1014 ____ s or 4. 28 x 1014 s-1 6
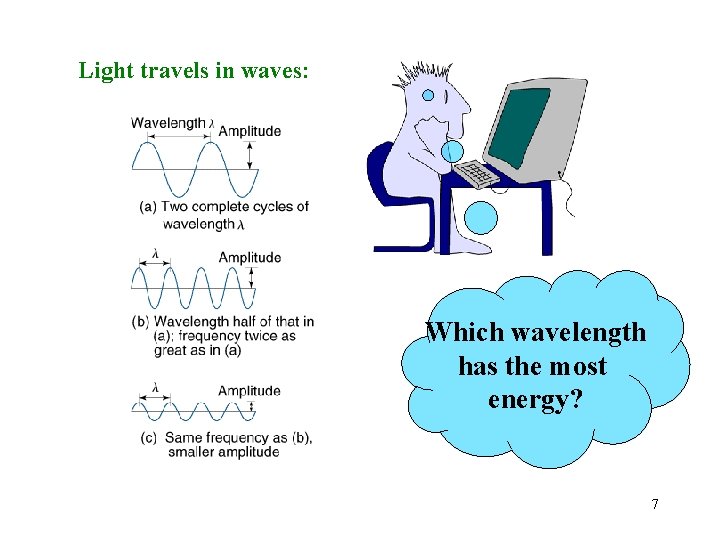
Light travels in waves: Which wavelength has the most energy? 7
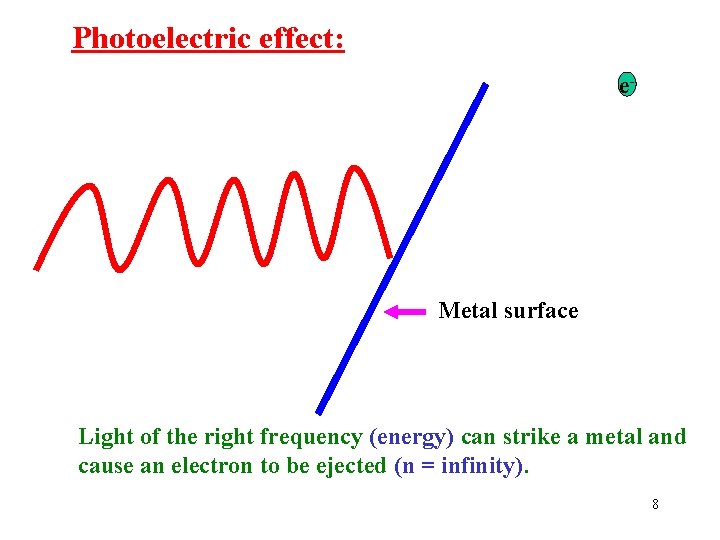
Photoelectric effect: e- Metal surface Light of the right frequency (energy) can strike a metal and cause an electron to be ejected (n = infinity). 8
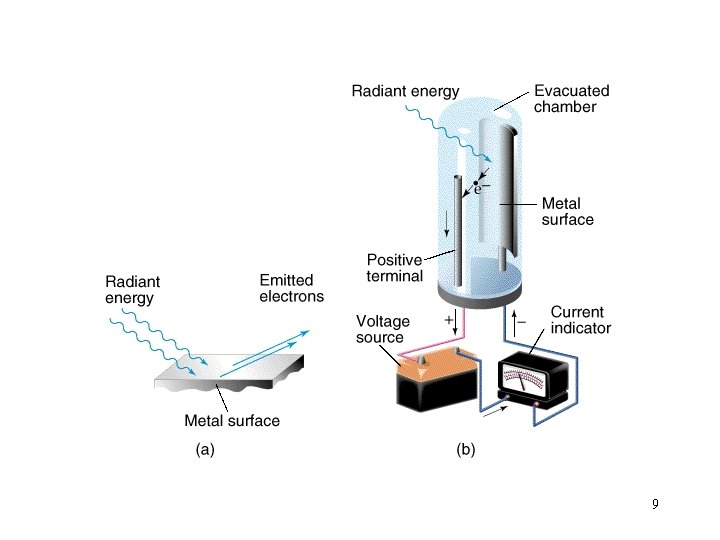
9
10
11
The nucleous - The first layer of e ’s e 12

“Energy is quantized” “light energy particles are called photons” E =h frequency Planck’s constant = 6. 63 x 10 -34 J • s Energy 13
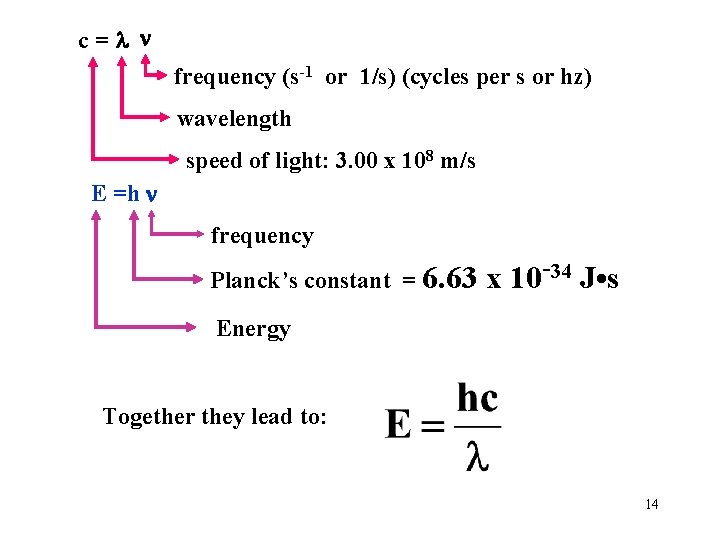
c= frequency (s-1 or 1/s) (cycles per s or hz) wavelength speed of light: 3. 00 x 108 m/s E =h frequency Planck’s constant = 6. 63 x 10 -34 J • s Energy Together they lead to: 14

Calculate the energy of red light vs. blue light. red light: 700 nm blue light: 400 nm red: E = 2. 85 x 10 -19 J blue: E = 4. 96 x 10 -19 J sunburn? ? ? uv 15
16
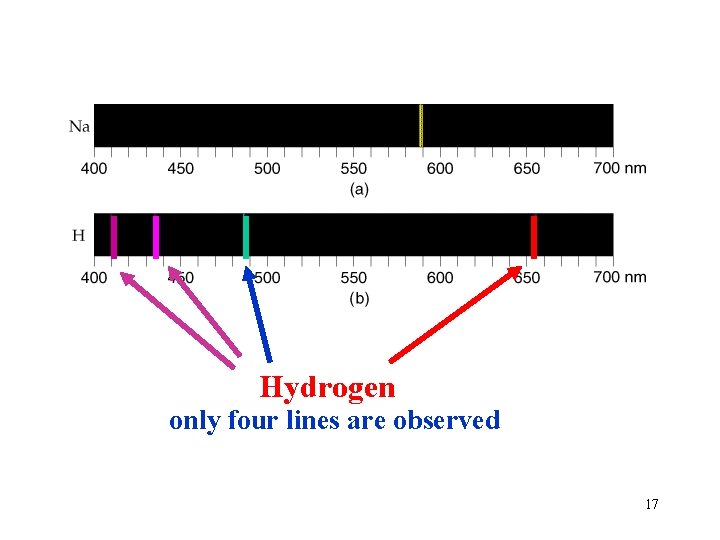
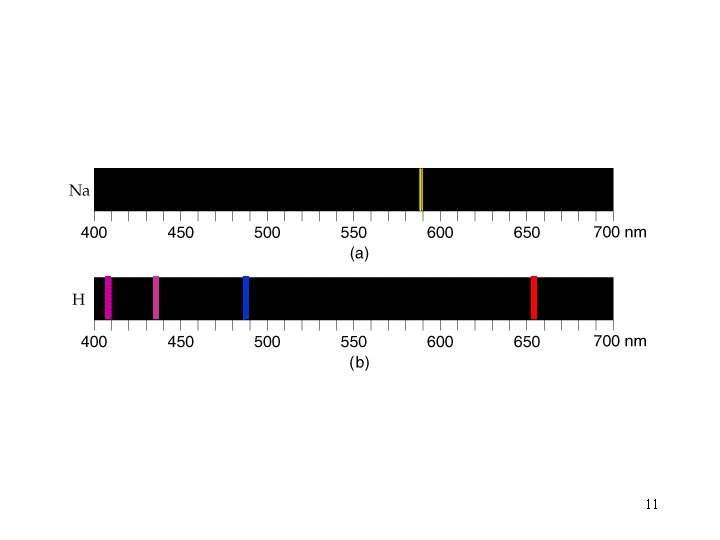
Hydrogen only four lines are observed 17
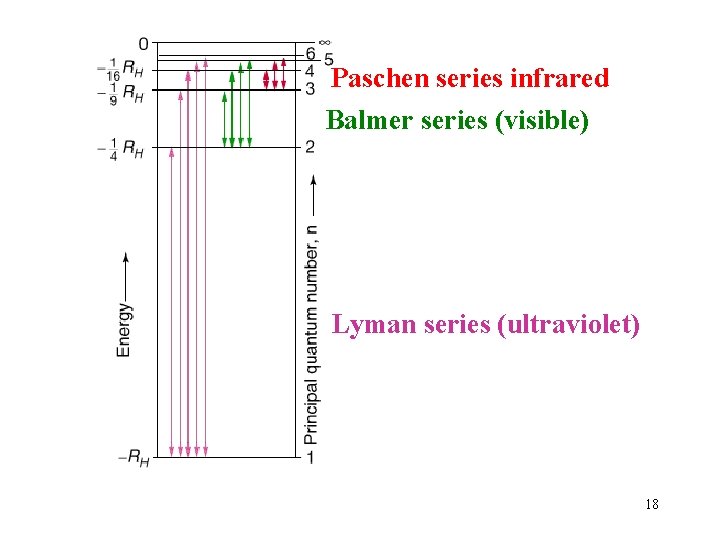
Paschen series infrared Balmer series (visible) Lyman series (ultraviolet) 18
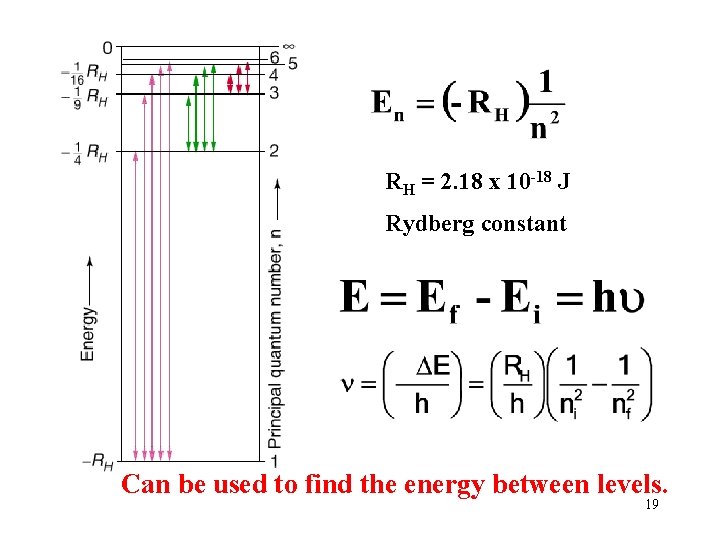
RH = 2. 18 x 10 -18 J Rydberg constant Can be used to find the energy between levels. 19

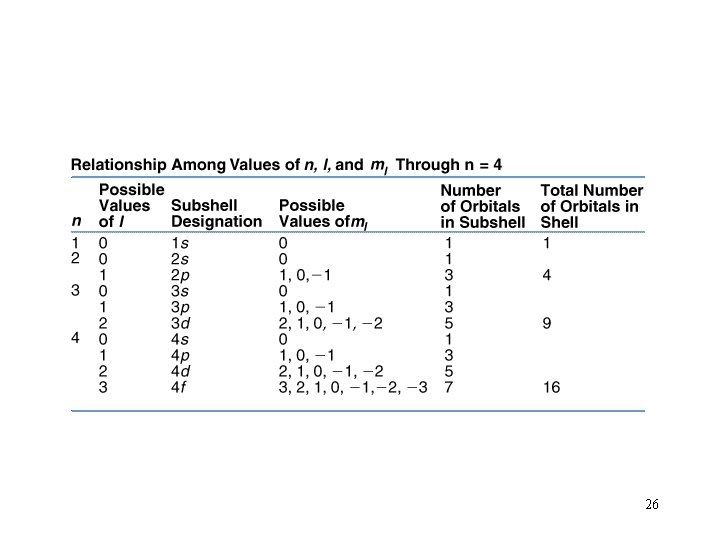
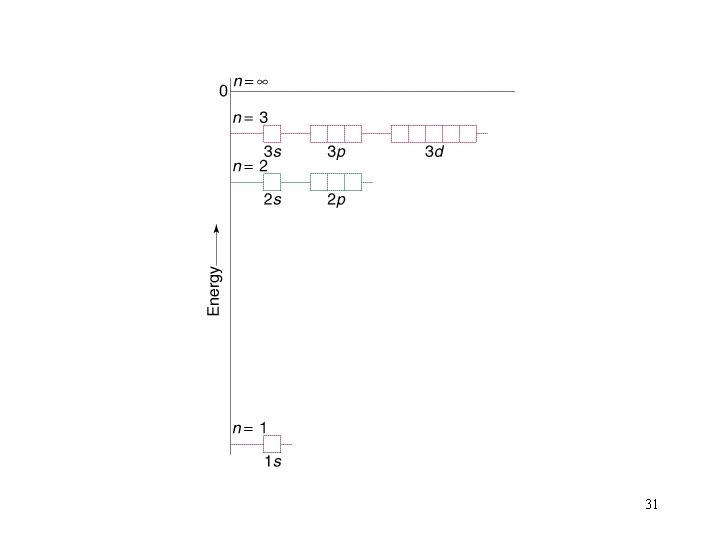
Quantum Numbers: type n = principle quantum no. values 1, 2, 3, etc meaning shell (period) l = angular momentum quant. # or azimuthal q# 0, 1, 2, 3. . . s, p, d, f, g, h. . subshell ml = magnetic q. # 0, 1, 2, 3, . . orbital ms = spin q. # ½ spin These 4 Quantum numbers give the general location of electrons within an atom and the general shape of the orbital in which they reside. 20
It is really an address system to write to electrons. 21

If you become frightened, close your eyes and repeat: “It’s only model” until you are once again calm. 22
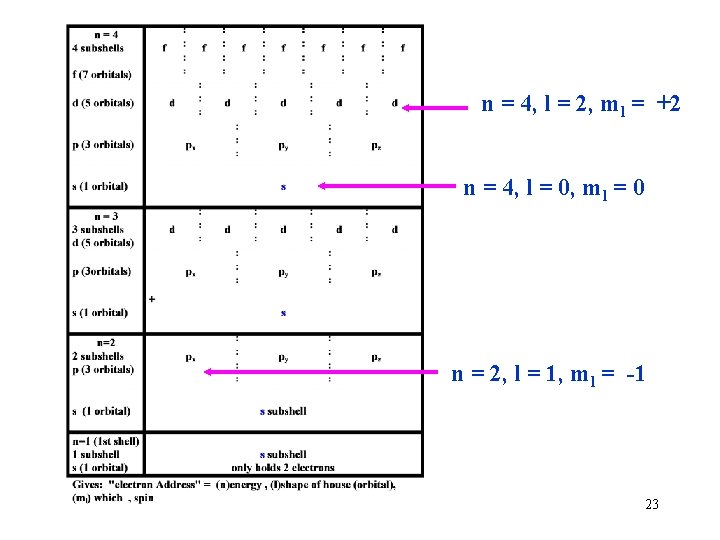
n = 4, l = 2, ml = +2 n = 4, l = 0, ml = 0 n = 2, l = 1, ml = -1 23

l = 3 ml =-3, -2, -1, 0, 1, 2, 3 n = 4 l = 2 ml =-2, -1, 0, 1, 2 l = 1 ml =-1, 0, 1 l=0 ml =0 l=2 n=3 l=1 l=0 n=2 n =1 24
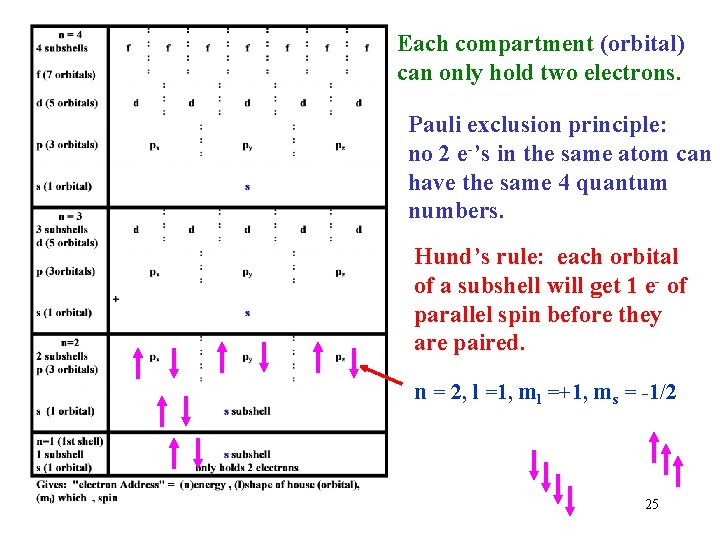
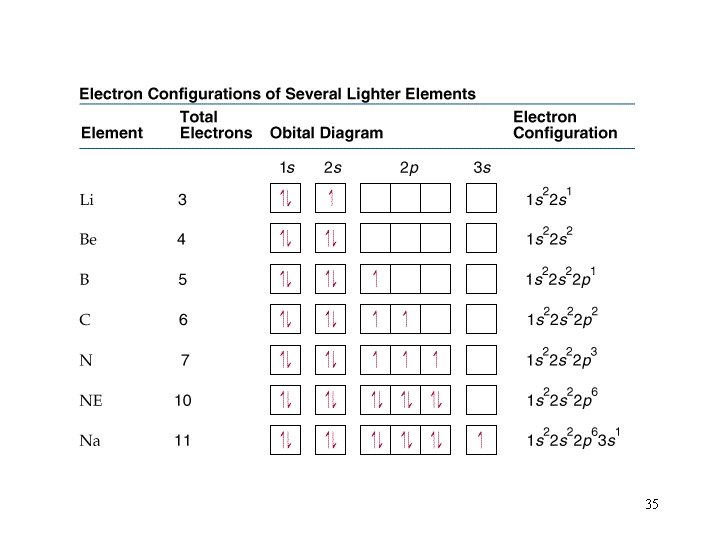
Each compartment (orbital) can only hold two electrons. Pauli exclusion principle: no 2 e-’s in the same atom can have the same 4 quantum numbers. Hund’s rule: each orbital of a subshell will get 1 e- of parallel spin before they are paired. n = 2, l =1, ml =+1, ms = -1/2 25
26

Orbital Shapes 27
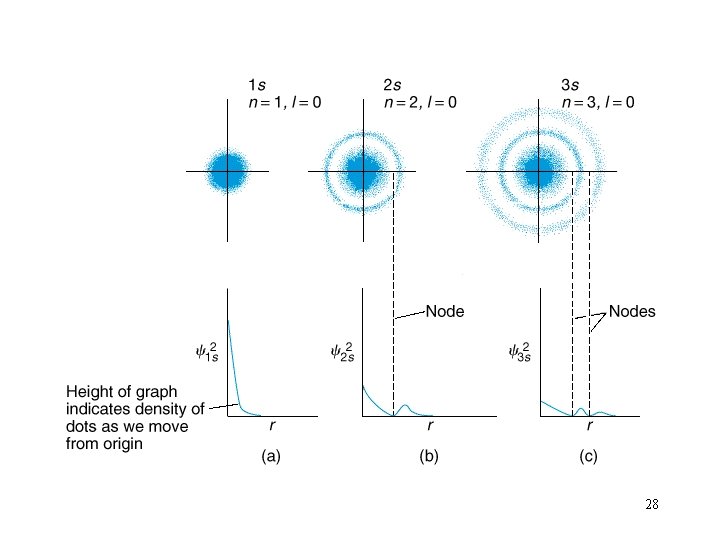
28
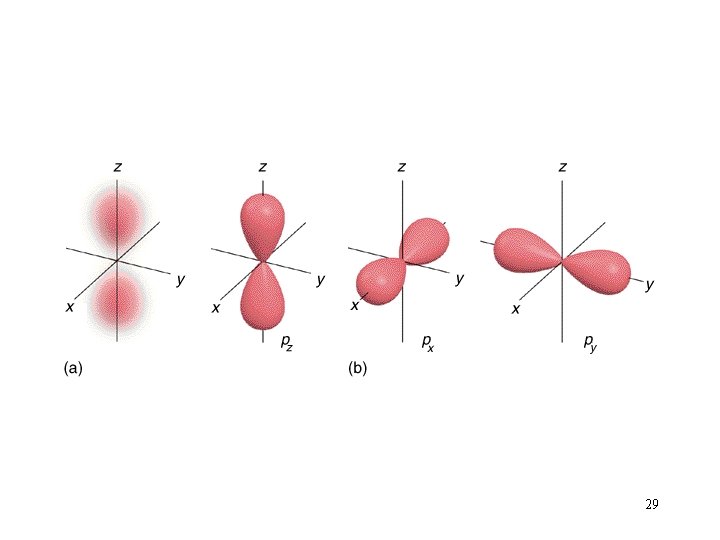
29
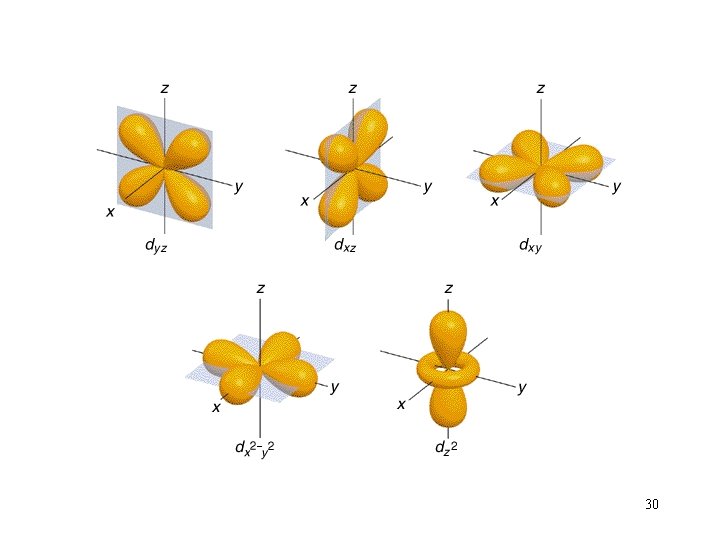
30
31

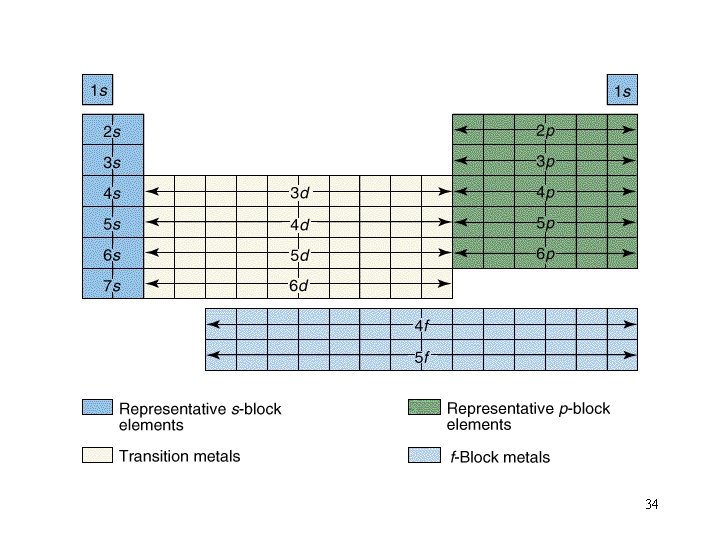
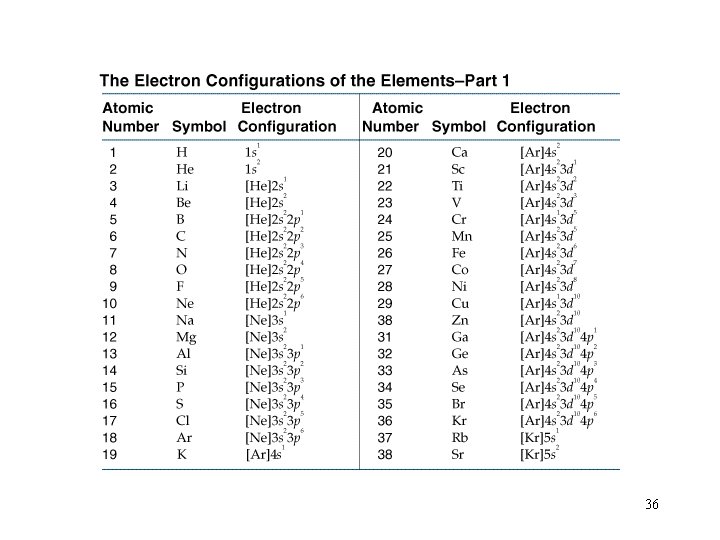
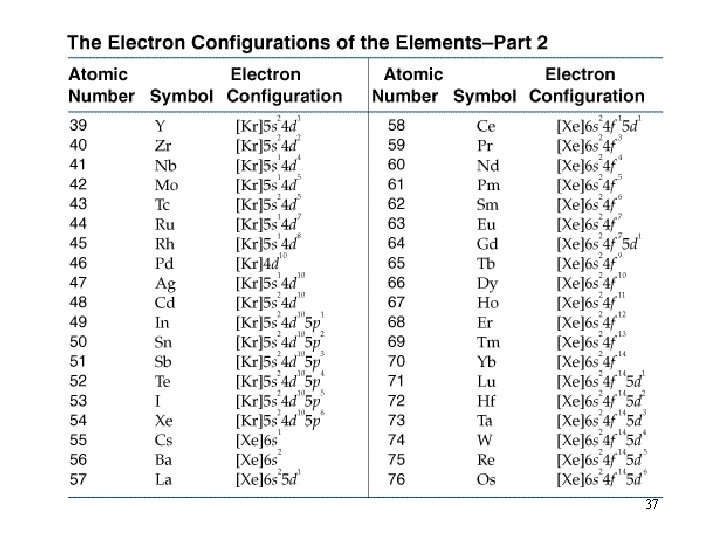
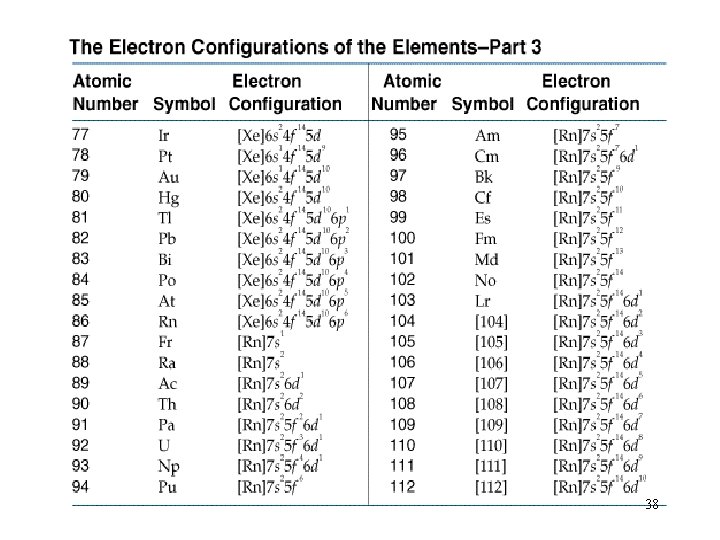
Filling Orders let’s use the periodic table to write out filling orders. 32
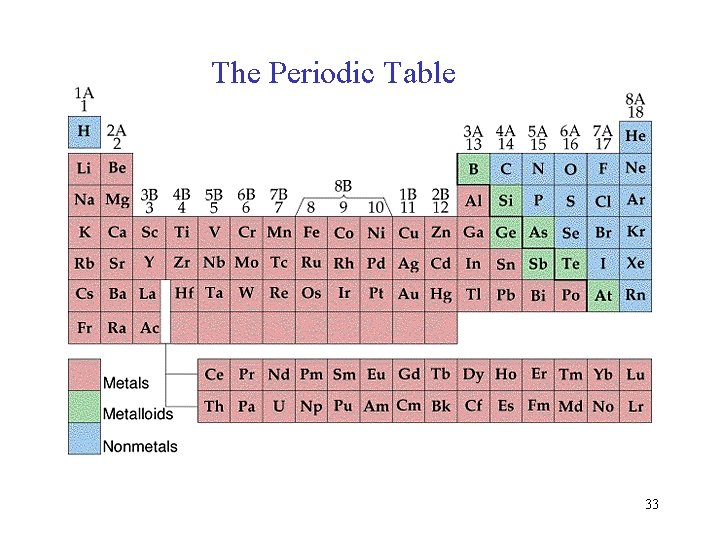
The Periodic Table 33
34
35
36
37
38
- Slides: 38