The Pediatric Research Center Clinical Research Center Introduction


























- Slides: 31

The Pediatric Research Center Clinical Research Center Introduction and Study Submission Guidelines

PRC Leadership • Anne Fitzpatrick, Ph. D, MSCR, APRN Program Director of the PRC Assistant Professor, Pediatric Pulmonology Larry Greenbaum, MD, Ph. D Associate Program Director of the PRC Professor and Division Director, Pediatric Nephrology Stephanie Meisner, RN, BSN, CCRP Clinical Research Manager of the PRC Children’s Healthcare of Atlanta 2

PRC Staff Michelle Popler, RN, BSN Senior Research Nurse Jessica Heyer, RN, BSN Research Nurse Elizabeth Liu, EMT Research Coordinator/Patient Care Tech. Children’s Healthcare of Atlanta 3

Where Are We? Location: Tower 2 Butterfly elevators 2 nd Fl, inside the TDICU Hours: 8: 00 – 5: 30 PM Phone number: 404 -785 -0400 Facilities available for Coordinators: • Fax/Copy/Scan documents in TDICU nurses station • Family lounge for patients – i. e. Ice & coffee • Computer station in PRC Children’s Healthcare of Atlanta 4

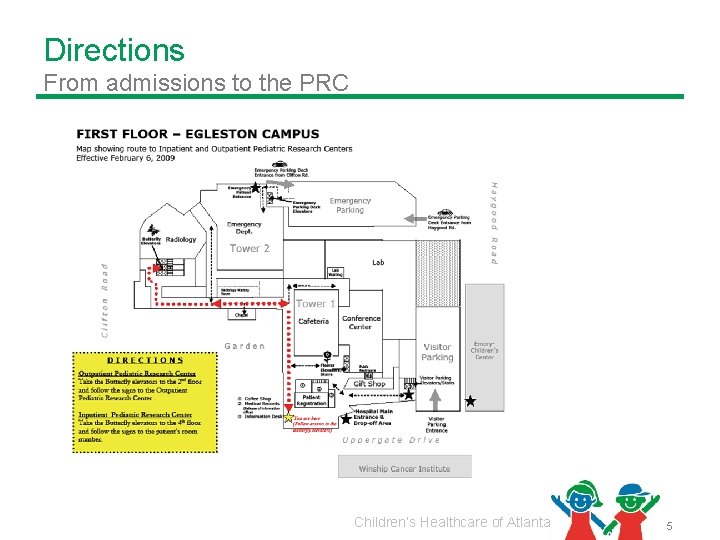
Directions From admissions to the PRC Children’s Healthcare of Atlanta 5

PRC Services Provides facilities & services for clinical research: § 4 bed outpatient unit § 4 bed inpatient unit § Nursing resources § Core lab § Bio-nutrition § Family-centered amenities Investigational Research Pharmacist provides Investigational Drug Services Jim Rhodes, james. rhodes@choa. org Research lab provides lab processing and shipping services Diana Worthington-White, diana. worthington-white@choa. org Clinical Interaction Site of the Atlanta Clinical and Translational Science Institute (ACTSI). Funded in part by the National Institutes of Health through CTSA. Children’s Healthcare of Atlanta 6

PRC Nursing Services – Nursing assessment – Vital Signs and Measurements – Phlebotomy – IV infusions – Roundtable Review – Protocol Order Review – Scheduling Assistance – Safety Oversight Children’s Healthcare of Atlanta 7

How to get your study in the PRC Click here for 1 page guide or visit: http: //www. pedsresearch. org/clinical-research/pediatric-researchcenter Children’s Healthcare of Atlanta 8

How to get your study in the PRC P R C 1 P A G E Children’s Healthcare of Atlanta 9

PRC Guidelines can be found here: http: //www. pedsresearch. org/clinical-research/pediatric-research-center Children’s Healthcare of Atlanta 10

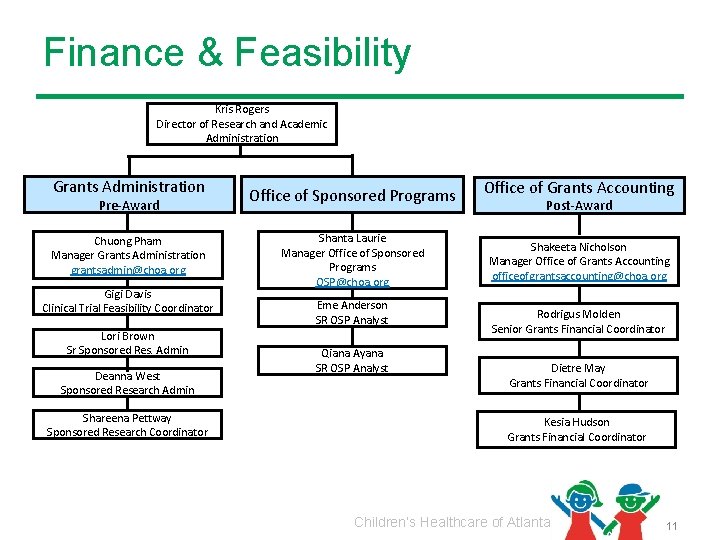
Finance & Feasibility Kris Rogers Director of Research and Academic Administration Grants Administration Pre-Award Chuong Pham Manager Grants Administration grantsadmin@choa. org Gigi Davis Clinical Trial Feasibility Coordinator Lori Brown Sr Sponsored Res. Admin Deanna West Sponsored Research Admin Shareena Pettway Sponsored Research Coordinator Office of Sponsored Programs Shanta Laurie Manager Office of Sponsored Programs OSP@choa. org Eme Anderson SR OSP Analyst Qiana Ayana SR OSP Analyst Office of Grants Accounting Post-Award Shakeeta Nicholson Manager Office of Grants Accounting officeofgrantsaccounting@choa. org Rodrigus Molden Senior Grants Financial Coordinator Dietre May Grants Financial Coordinator Kesia Hudson Grants Financial Coordinator Children’s Healthcare of Atlanta 11

Finance Breakdown • • • Pre-Award Functions Provide institutional information and assist with form preparation Feasibility Analysis Review and edit proposals Facilitate and manage internal routing and review Negotiate and execute contracts and subcontracts • • • Post-Award Functions Budget Management Grant Invoicing Monitor Deliverables Electronics Financial Drawdowns Coordination Grant Closeout Billing Trackers Children’s Healthcare of Atlanta 12

CHOA Routing Checklist Complete Intent to Submit Form: http: //www. pedsresearch. org/clinical-research/resources/RAS Email study protocol to : prc@choa. org Stepanie. meisner@choa. org Send DAF and budget requests to ALL CHOA ancillary services including the PRC DAF & Budget Forms: Click here for Forms Children’s Healthcare of Atlanta 13

ACTSI Submission ACTSI submission info. : http: //www. atlantactsi. org/areas/crn/protocol_submission/index. html Submitting to What? • The Scientific Advisory Committee (SAC) • Meets once/month • Reviews all protocols submitted to ACTSI for scientific soundness and execution • Members include investigators (MD’s & Ph. D’s), statisticians, nutritionists and nurses. Children’s Healthcare of Atlanta 14

ACTSI Submission Submit below items to: cnetwor@emory. edu E X A M P L E 1. ACTSI Form 2. IRB documents (can be pending) 3. PRC Draft Orders Children’s Healthcare of Atlanta 15

SAC Meeting Schedule: 2014 • Completed submissions head to SAC for review • Should be submitted 3 weeks prior to scheduled SAC meeting • Check the SAC Meeting Schedule • 2014 Schedule here Committee Meeting Date Submission Deadline SAC 2 January 14, 2014 (Tue) December 24, 2013 SAC 3 February 13, 2014 (Thu) January 23, 2014 SAC 1 March 13, 2014 (Thu) February 20, 2014 SAC 2 April 8, 2014 (Tue) March 18, 2014 SAC 3 May 8, 2014 (Thu) April 17, 2014 SAC 1 June 12, 2014 May 22, 2014 (Thu) SAC 2 July 8, 2014 (Tue) June 17, 2014 SAC 3 August 14, 2014 (Thu) July 24, 2014 SAC 1 September 11, August 21, 2014 (Thu) SAC 2 October 14, 2014 (Tue) SAC 3 November 13, October 23, 2014 (Thu) SAC 1 December 11, 2014 (Thu) September 23, 2014 November 20, 2014 Children’s Healthcare of Atlanta 16

PRC Checklist • Submit Order sets to: PRC@choa. org using our Order Set Template • PRC staff verifies orders with study protocol • Coordinator needs to verify returned edited orders • Finalize orders Any changes should be sent and verified with PRC prior to use • Once approved by SAC, schedule roundtable meeting with PRC team • Orders will be reviewed at roundtable Children’s Healthcare of Atlanta 17

PRC Checklist • Provide PRC with ACTSI SAC approval letter • Organize In-service needs: Research lab, pharmacy, EKG, ECHO, etc. • Order necessary equipment and supplies Children’s Healthcare of Atlanta 18

Obtaining CR-Assist Access CR-Assist: ACTSI research appointment hub Info: http: //www. atlantactsi. org/areas/crn/crassist/index. html § Contact Khushbu Amin for access and send completed study form to Kamin 5@emory. edu CR-Assist Access Form § Contact Audrey Kelly at Audrey. kelly@emoryhealthcare. org for CR-Assist training & study visit set up after Khushbu’s approval Children’s Healthcare of Atlanta 19

Obtaining CR-Assist Access CR-Assist Study Setup Form E X A M P L E Children’s Healthcare of Atlanta 20

Other ACTSI Research Resources More Info on Services offered: http: //www. atlantactsi. org/areas/crn/services%20 available. htm l http: //www. atlantactsi. org/areas/crn/services/index. html • Comprehensive biostatistical and epidemiological support (BERD Program) • Biomedical Informatics Program (BIP) • Community engagement expertise (CERP) • Pediatrics Expertise • Research Technology Resources • Core Facilities • National Recruitment Assistance Children’s Healthcare of Atlanta 21 • REDCap

PRC Scheduling Steps Once study built in CR-Assist: • Enter appointment request through CR-Assist • PRC will approve and schedule PRC staff pre-registers patient in EPIC • Patient registers with registration in CHOA lobby on 1 st visit • Patient must be re-registered after 1 year Subject comes to PRC for consent and treatment Children’s Healthcare of Atlanta 22

Not a CHOA Employee? How to Get Credentialed: § Contact CHOA Credentialing Services at below numbers for appropriate application packet (non-clinical, chart review, etc. ) § Complete top section of Credentialing Checklist § Scan form to CHOA Clinical Research Credentialing Rep: Saadia Khizer saadia. khizer@choa. org (404 -785 -9893) CHOA Credentialing Contacts: Name Title CHOA Ext. Provider Last Name Kuklinski, Lisa Manager 404 -785 -7485 N/A Lowry, Susan Supervisor 404 -785 -7487 N/A Morris, Enriquita Credentialing Coordinator 404 -785 -7492 A, B, V, X, Y, Z Brzezinski, Vickie Credentialing Coordinator 404 -785 -6798 J, L, T, W Guda, Susan Credentialing Coordinator 404 -785 -2198 M, N, O, P Children’s Healthcare of Atlanta 23

For Patient Chart Access/Patient Contact: You will need: • Background Consent Form • Complete Drug Screen • Immunization records with Employee Health *Flu shot and TB test will be required if not already obtained* Children’s Healthcare of Atlanta 24

For Clinical Credentialing: You will need: • Contact Susan Guda in Credentialing Services at (404) 7852198 • Complete “Credentialing and Scope of Practice” form and application packet obtained from Credentialing Services Children’s Healthcare of Atlanta 25

End of Route Steps Agreements and Sub-Agreements Emory and CHOA • Contract site determined by investigator (Emory Investigator=Emory PRIME Award) • Sub-contract site = Performance site – Where the research activities are done (CHOA for PRC) • MUST be completed PRIOR to subject being seen in PRC Children’s Healthcare of Atlanta 26

Post Approval Processes Required updates and Renewals • • Protocol and Agreement Amendments IRB modification, renewal and event reporting – CHOA/Emory OCR communications Grants Management – renewals, reporting, on-going routing Sponsor / Monitor communication, monitoring visits, queries DSMB reporting Billing q. Patient care billing • Coordinators must complete billing trackers q. Facility and Professional Services fees • Must be included on billing trackers • Subject must be PRE-registered for bill-hold before EVERY visit: http: //www. choa. org/Pediatric-Research/Research-Visit -Pre-Registration Children’s Healthcare of Atlanta 27

Study Completion Publications • MUST CITE ACTSI support in publications: http: //www. atlantactsi. org/about/citation. html Citing ACTSI: "Supported by the National Center for Advancing Translational Sciences of the National Institutes of Health under Award Number UL 1 TR 000454. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. " KL 2 Scholars should also list KL 2 TR 000455 and TL 1 Trainees should also list TL 1 TR 000456. • Share with PRC when published • Publication of findings in peer reviewed journal • Goal to share knowledge and improve care • Must register study with http: //clinicaltrials. gov/ • (NIH service, public access, ICMJE) Children’s Healthcare of Atlanta 28

Study Completion After the study • Notify all services that study has ended, including PRC • IRB Completion • Records retention – IRB / finance 2 years , Subject records – 10 years /determined by sponsor • Billing and grant completion • Sponsor Agreement Termination • Sub-Agreement termination Children’s Healthcare of Atlanta 29

Coordinator Reminders Remind patients of NPO status Pre-Register pt. on choa. org/research for bill-hold Complete pre-appointment patient trackers Complete patient trackers immediately after appt. Schedule patient in CR-Assist at least 2 -3 weeks prior to appointment • If necessary, notify ancillary services (pharmacy, EKG, ECHO, etc. ) of patient schedule changes • Bring SIGNED Orders to study visit • Adhere to PRC Guidelines • • • Children’s Healthcare of Atlanta 30

PRC Contacts Important Emails: PRC@choa. org Manager: Stephanie. Meisner@choa. org Nurses: Michelle. Popler@choa. org Jessica. Heyer@choa. org Tech: Elizabeth. Liu@choa. org Children’s Healthcare of Atlanta 31