The PatientCentered Outcomes Research Institute PCORI Joe Selby

The Patient-Centered Outcomes Research Institute (PCORI) Joe Selby, MD MPH Executive Director, PCORI Florida State University System 4 th Annual Federal R&D Agency Workshop September 30, 2016
FOR TODAY PCORI – Clinical Research Done Differently PCORI’s Portfolio & Funding Opportunities Introducing PCORnet

Stakeholder-Driven Research From the Affordable Care Act, 2010 “The purpose of the Institute is to assist patients, clinicians, purchasers, and policy-makers in making informed health decisions by advancing the quality and relevance of evidence concerning the manner in which diseases, disorders, and other health conditions can effectively and appropriately be prevented, diagnosed, treated, monitored, and managed through research and evidence synthesis. . . and the dissemination of research findings with respect to the relative health outcomes, clinical effectiveness, and appropriateness of the medical treatments, services. . . ”

PCORI’s Mission PCORI helps people make informed health care decisions, and improves health care delivery and outcomes, by producing and promoting high integrity, evidence-based information that comes from research guided by patients, caregivers and the broader health care community.

PCORI was Established to Fund Comparative Clinical Effectiveness Research § Compares two or more options for screening, diagnosis, treatment that matter to patients (one option may be “usual care”) § Considers the range of outcomes that are important to patients § Conducted in real world populations and real world settings § Attends to differences in effectiveness and preferences across patient subgroups
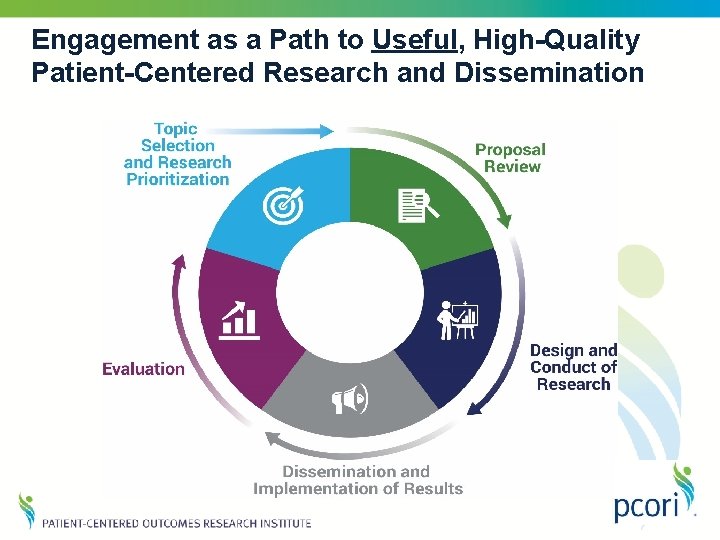
Engagement as a Path to Useful, High-Quality Patient-Centered Research and Dissemination
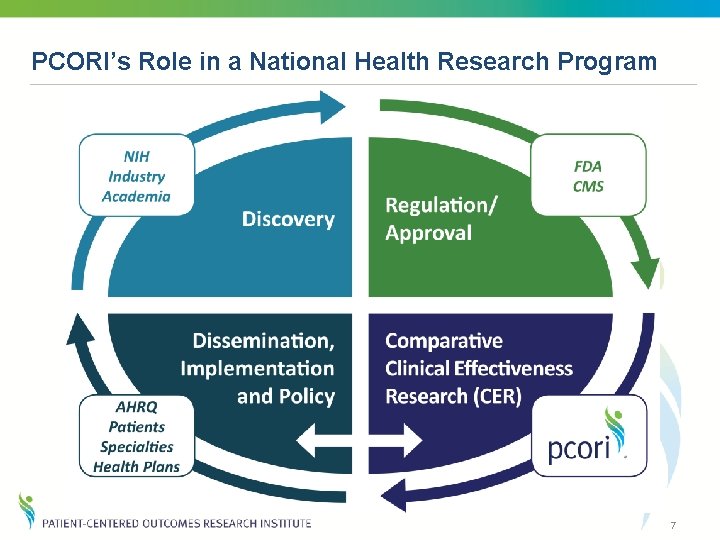
PCORI’s Role in a National Health Research Program 7

Adrian Felipe Hernandez, MD, MS, Duke University How Engaging Patients can Improve Research Real world effectiveness of warfarin among ischemic stroke patients with atrial fibrillation: observational analysis from Patient-Centered Research into Outcomes Stroke Patients Prefer and Effectiveness Research (PROSPER) study. Y Xian et al. , BMJ 2015; 351: h 3786

Impact of Involving Patients (The PROSPER Study) Study Design: Retrospective, observational cohort study within a registry of persons who had and survived an initial stroke (n=13, 000). Patient advisory panel helped to plan the study Research Question: Does use of warfarin (in A Fib), statins and antidepressants affect important clinical outcomes (3 analyses) Outcomes: Patient involvement changed the primary outcome from M. A. C. E. to “home time: days spent at home during follow-up. ”” Results: Among 12, 553 patients with atrial fibrillation after a stroke, those started on warfarin before discharge enjoyed 47 more days at home during an average of two years of follow-up, as well as lower rates of recurrent stroke, MI, death. “These findings support the routine use of warfarin for eligible ischemic stroke patients with atrial fibrillation, including those over 80 years of age, women, those with more severe strokes, and those with comorbid conditions” 9

Does Engagement Make a Difference? Recent systematic review* provides first evidence of the impact of patient and public involvement on research on health and social-care research. • Literature search from 1995 -2009 identified 66 studies • Analysis showed patient and public involvement enhanced quality and appropriateness of research • Impacts were described for all stages of research • But authors note the evidence base on impact of still needs significant enhancement *J Brett et al; Health Expectations 2014; 17(5): 637– 650.
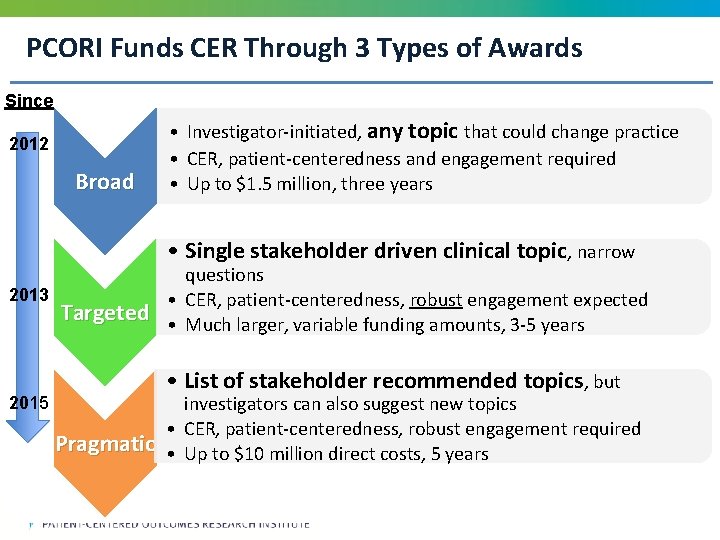
PCORI Funds CER Through 3 Types of Awards Since 2012 Broad • Investigator-initiated, any topic that could change practice • CER, patient-centeredness and engagement required • Up to $1. 5 million, three years • Single stakeholder driven clinical topic, narrow 2013 Targeted questions • CER, patient-centeredness, robust engagement expected • Much larger, variable funding amounts, 3 -5 years • List of stakeholder recommended topics, but 2015 Pragmatic investigators can also suggest new topics • CER, patient-centeredness, robust engagement required • Up to $10 million direct costs, 5 years

Targeted Research Funding Awards • PCOR treatment options in uterine fibroids* • Multifactorial fall injury prevention strategy in older persons** • Effectiveness of transitional care • Treatment options for African Americans and Hispanics/Latinos with uncontrolled asthma • Obesity treatment options set in primary care for underserved populations • Hypertension Disparities Reduction Awards in African-American and Rural populations** • Comparative effectiveness of new treatment options for Hepatitis C • Comparative effectiveness of novel oral anti-coagulants (NOACs) • Options for treatment-resistant depression • Comparative effectiveness of disease modifying treatments for MS

PCORI’s Pragmatic Clinical Studies Initiatives • Must address critical clinical choices faced by patients, caregivers, clinicians, systems • Topics of special interest identified by PCORI Advisory Panels, Institute of Medicine, Agency for Healthcare Research and Quality • Typically conducted in routine clinical settings • Though often large, protocols usually less complex than traditional trials • Requires engagement of major stakeholder organizations Opportunity Snapshot • Number of Anticipated Awards Per Funding Cycle: Six to Nine • Funds Available Per Cycle: Up to $90 Million • Maximum Project Duration: 5 Years • Maximum Direct Costs Per Project: $10 Million

Examples from First 19 Pragmatic Clinical Studies • Operative vs medical endocrine therapy for management of low-risk DCIS patients. • Nerve blocking regional anesthesia vs general anesthesia in older adults undergoing surgery for hip fracture on acute, inpatient morbidity, ability walk without assistance at 60 and 180 days, disability, pain, ability to return home after fracture, and mortality. • Exercise coaching program vs. usual care for older adults who have experienced a low-impact fracture as a result of a fall for preventing further injuries and improving health. • Surgical vs. antibiotic therapy for uncomplicated appendicitis for complications, subsequent appendectomy, safety, patient experience • Uncoated aspirin vs. low intensity warfarin vs. rivaroxaban for prevention of venous thrombosis after hip or knee replacement for aggregate clinical pulmonary embolism/deep vein thrombosis and all-cause mortality, bleeding and patient-reported outcomes.

Eugene Washington PCORI Engagement Awards: Launched in February 2014. Supports PCORI’s Engagement Imperative, as defined in our Strategic Plan, to: § Develop a patient-centered outcomes research (PCOR)-ready community to establish an infrastructure for engagement in PCOR; § Engage these communities in the research process; and § Promote dissemination of PCOR/CER evidence. Funds projects that build a community better able to participate in PCOR and CER, as well as serve as channels to disseminate related study results. 16

Engagement Awards Programmatic Support Categories Supports projects and meetings that most fall into one or more of the following categories: 17

Introducing

PCORnet’s Mission To make it faster, easier, and less costly to conduct health system and clinical research, both observational studies and randomized trials, than is now possible by harnessing the power of large amounts of health data and patient partnerships, and by making contracting negotiations and making IRB approval/oversight more efficient. To embed the research within health systems and use data and research findings to facilitate health system improvement. And in the process, transform the culture of clinical research from one directed by researchers acting as entrepreneurs to one driven by collaboration and the needs of patients, clinicians, systems and payers.
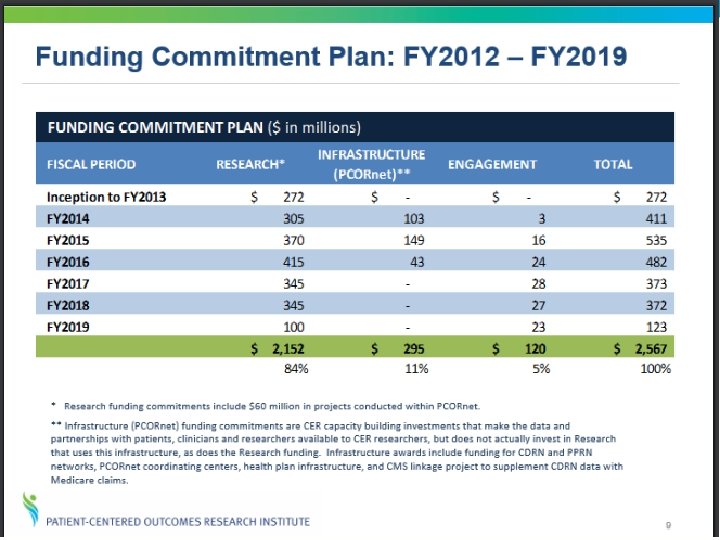
There’s a Site Near You Pragmatic This map depicts the number of PCORI-funded Patient. Powered or Clinical Data Research Networks that have coverage in each state.

PCORnet Common Data Model The Common Data Model 21
Expanding Data in the CDM Biospecimen & Genomic Data Encounters Condition Death Index Prescribing Patient Satisfaction Procedures Lab Results Demographic Claims

130 health systems across the country Over 60 data marts Data on over >100 million patients

ADAPTABLE Trial Start-Up Master Slide Set March, 2016 Clinical. Trials. gov: NCT 02697916

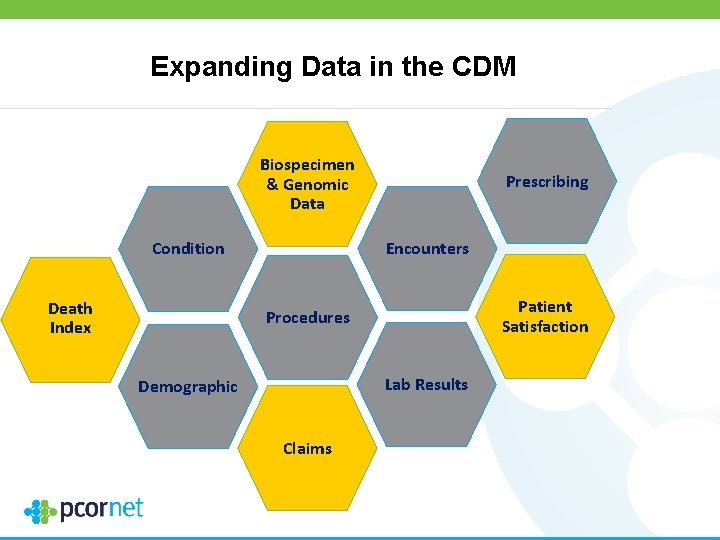
ADAPTABLE Study Design Patients with known ASCVD + ≥ 1 “enrichment factor”* Identified through EHR (computable phenotype) by CDRNs (PPRN patients that are already a part of a CDRN are eligible to participate. ) Patients contacted with trial information and link to e-consent; † Treatment assignment will be provided directly to patient Exclusion criteria ASA 81 mg QD ASA 325 mg QD • Age <18 years • ASA allergy or Electronic follow-up: Every 3– 6 months contraindication Supplemented with EHR/CDM/claims data (including pregnancy or nursing) • Significant GI bleed Duration: Enrollment over 24 months; within past 12 months maximum follow-up of 30 months • Significant bleeding Primary endpoint: disorder Composite of all-cause mortality, hospitalization • Requires warfarin, direct for MI, or hospitalization for stroke oral anticoagulant, or Primary safety endpoint: ticagrelor Hospitalization for major bleeding *Enrichment factors • Age >65 years • Creatinine >1. 5 mg/d. L • Diabetes mellitus (T 1 or 2) • Known 3 -vessel CAD • Current CVD or PAD • Known EF <50% by echo, cath, nuclear study • Current smoker A subset of participants who do not have internet access may be consented and followed via a parallel system. † Clinical. Trials. gov: NCT 02697916
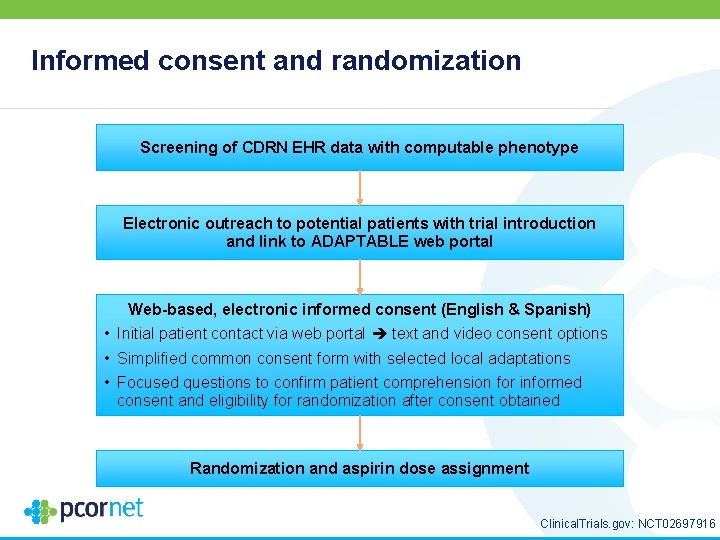
Informed consent and randomization Screening of CDRN EHR data with computable phenotype Electronic outreach to potential patients with trial introduction and link to ADAPTABLE web portal Web-based, electronic informed consent (English & Spanish) • Initial patient contact via web portal text and video consent options • Simplified common consent form with selected local adaptations • Focused questions to confirm patient comprehension for informed consent and eligibility for randomization after consent obtained Randomization and aspirin dose assignment Clinical. Trials. gov: NCT 02697916

PCORnet’s first Collaborative Research Groups Purpose The purpose is to explore the feasibility, efficiency, and usefulness of cross-Network forums, organized around specific clinical areas, for building capacity within PCORnet to generate multi-network research projects that answer research questions of high priority to patients, clinicians, and health system leaders. The PCORnet Council approved four exploratory CRG pilots to assess their feasibility, efficiency, and usefulness for building PCORnet capacity to form research communities that develop PCORnet studies. Topic Timeline/Plans Pediatrics Initial activities are planned for a 6 -month period; if successful Cardiovascular Health other CRGs will be added quickly. Leads Chris Forrest [PEDSnet] Betsy Shenkman [One. Florida] Mark Pletcher [Health e. Heart] Veronique Roger [LHSnet] Diversity Jen Devoe/Jon Puro [ADVANCE] Mitch Lunn [PRIDEnet] Health Systems Rainu Kaushal [NYC] Beth Mc. Glynn [PORTAL] 27

Evidence Synthesis—Immediate PCORI plans Initially, the Evidence Synthesis portfolio will focus on shortturnaround, rigorous, relevant CER or heterogeneity of treatment effect products • Strategic, selective analyses of existing trial data (e. g. , IPD MA focused on treatment variation) • Updating high-quality existing SR support up-to-date decision-making on critical CER topics • Most ES is conducted through and in collaboration with AHRQ and its EPC’s. Over time, PCORI will continue to refine its strategy in response to stakeholders’ needs for robust, relevant, non-duplicative evidence synthesis products 47

Thanks……. . and Stay in Touch! Pragmatic
- Slides: 29