THE OZONE HOLE O O O 2016 Paul









- Slides: 14

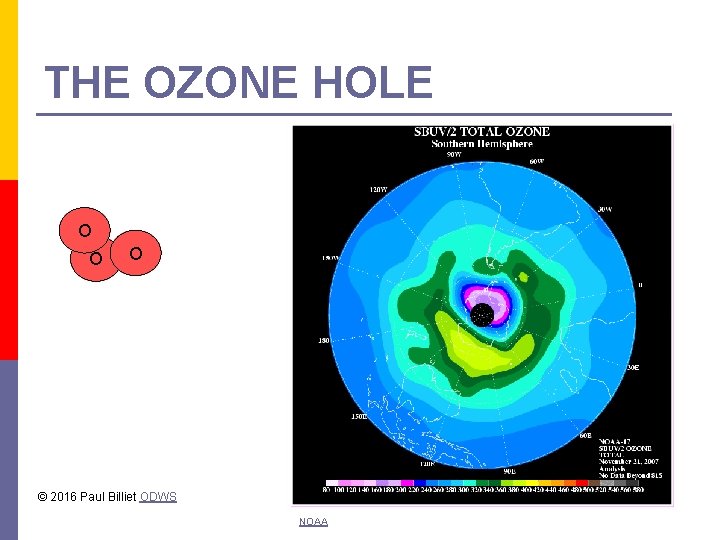
THE OZONE HOLE O O O © 2016 Paul Billiet ODWS NOAA

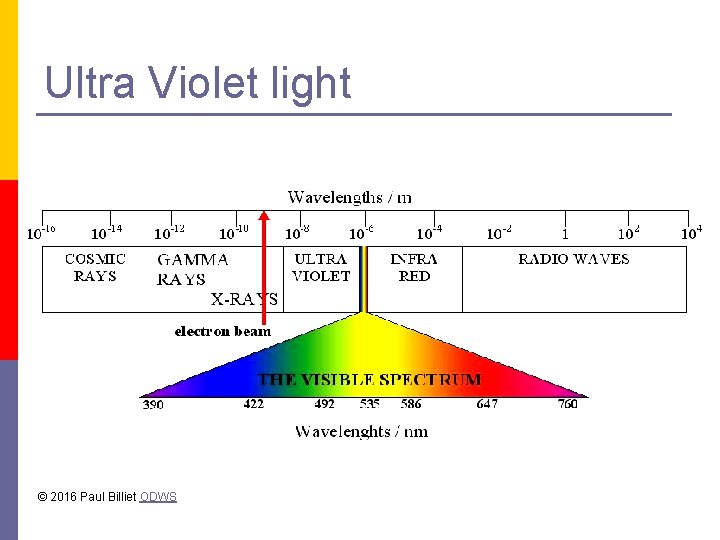
Ultra Violet light © 2016 Paul Billiet ODWS

UV light and life UV light is high energy radiation p When it strikes molecules it can cause them to beak into ions or free radicals p The free radicals in turn damage large molecules such as proteins and DNA p Damage to DNA causes mutations p It is a mutagen p Mutations can lead to cancer. p © 2016 Paul Billiet ODWS

UV light and life UV light can have beneficial effects too p It stimulates the transformation dietary steroids into Vit D (calciferol) p UV light is used to sterilise foods and medical equipment p Some animals can see UV light p Insects use it to guide them to nectar sources in flowers. p © 2016 Paul Billiet ODWS

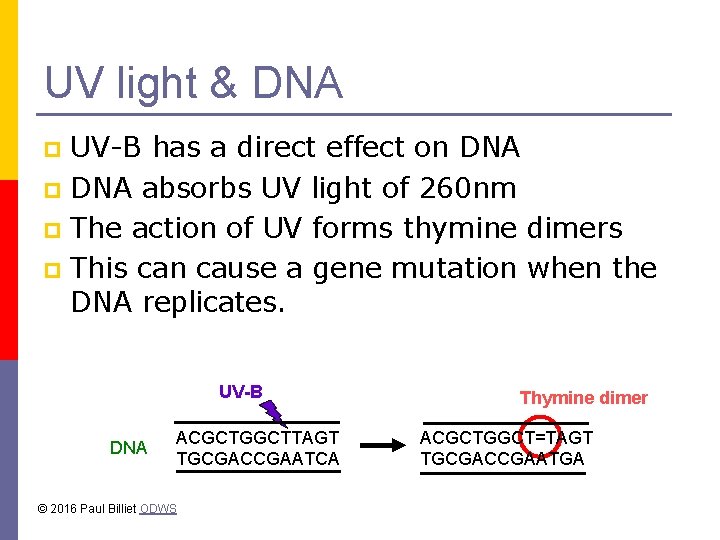
UV light & DNA UV-B has a direct effect on DNA p DNA absorbs UV light of 260 nm p The action of UV forms thymine dimers p This can cause a gene mutation when the DNA replicates. p UV-B DNA ACGCTGGCTTAGT TGCGACCGAATCA © 2016 Paul Billiet ODWS Thymine dimer ACGCTGGCT=TAGT TGCGACCGAATGA

Specific damage by UV light Sunburn p Clouding of the cornea and formation of cataract p Skin cancer (melanoma) p Reduced rates of photosynthesis. p © 2016 Paul Billiet ODWS

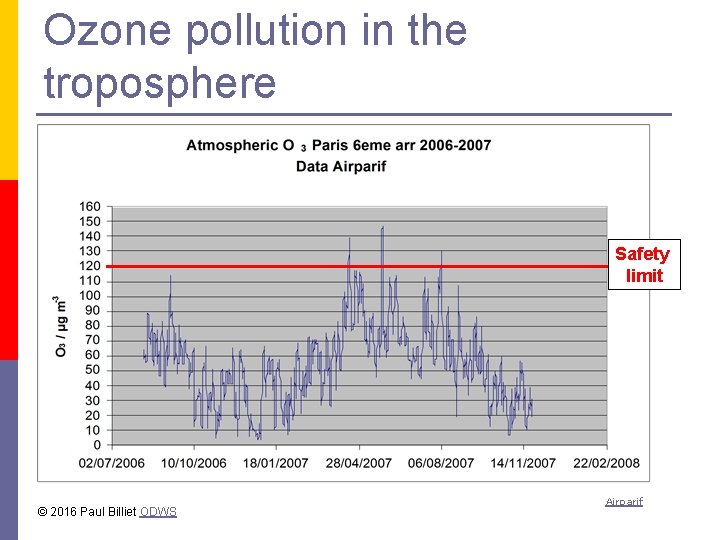
Ozone in the atmosphere Ozone (O 3) forms under the effect of UV light in the stratosphere p Ozone can also form in the troposphere p Naturally it forms from volcanic activity p It is produced by motor vehicules so it is a pollutant p Sunlight acts on NOx to produce O 3. p © 2016 Paul Billiet ODWS

Ozone pollution in the troposphere Safety limit © 2016 Paul Billiet ODWS Airparif

The ozone layer Ozone is an unstable gas p It rapidly breaks down p The ozone layer is only a few cm thick p If the rate of breakdown is faster than the rate of formation the ozone layer thins p This could develop into hole p An ozone hole was first observed over the Antarctic in 1985. p © 2016 Paul Billiet ODWS

The cause of the hole p p Chlorofluorocarbons (CFCs) The breakdown under the effect of UV light to release chlorine radicals (Cl) Especially under cold conditions The chlorine radicals react with O 3 converting it to O 2 and more radicals (Cl. O) 2 Cl + 2 O 3 2 Cl. O + 2 O 2 2 Cl. O + M Cl 2 O 2 + M Cl 2 O 2 + sunlight 2 Cl + O 2 © 2016 Paul Billiet ODWS 2 O 3 3 O 2

What are CFCs? Used as propellants in aerosol spray cans p Used as refrigerants in fridges, freezers and air conditioning units. p © 2016 Paul Billiet ODWS

What can be done? Reduce the use of CFCs p They are already banned in aerosols (1987) p BUT they are still used as refrigerants p Recycle fridges and air conditioning plants. p © 2016 Paul Billiet ODWS

The current situation The holes developing over the pole suggest that they may be showing an improvement p BUT CFC molecules take 30 years to rise up to the stratosphere p The chlorine radicals last a long time p The peak ozone damage was supposed to be in 2000 p Damage could go on another 70 years. p © 2016 Paul Billiet ODWS

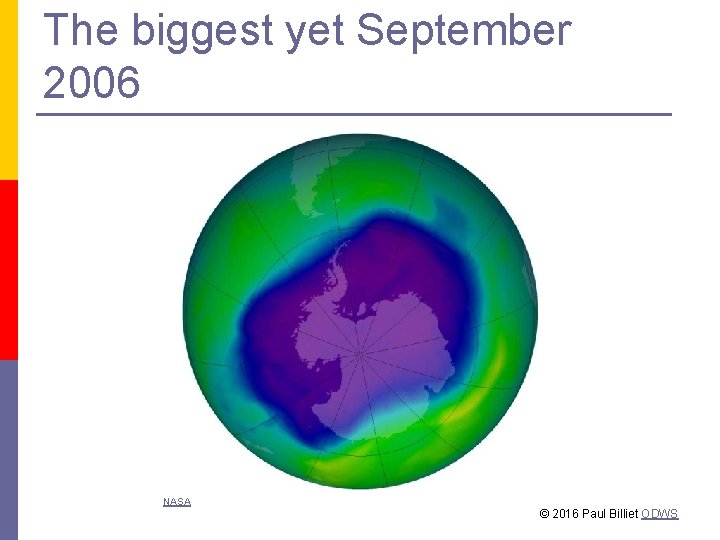
The biggest yet September 2006 NASA © 2016 Paul Billiet ODWS