The Oral Microbiome and Salivary Biomarkers in Health

The Oral Microbiome and Salivary Biomarkers in Health and Disease Farah Ibrahim Al-Marzooq MBCh. B , MMed. Sc , Ph. D Sharjah Institute for Medical Research, University of Sharjah, UAE & Natheer Al-Rawi College of Dental Medicine, University of Sharjah,

n o i t c u d o r Int
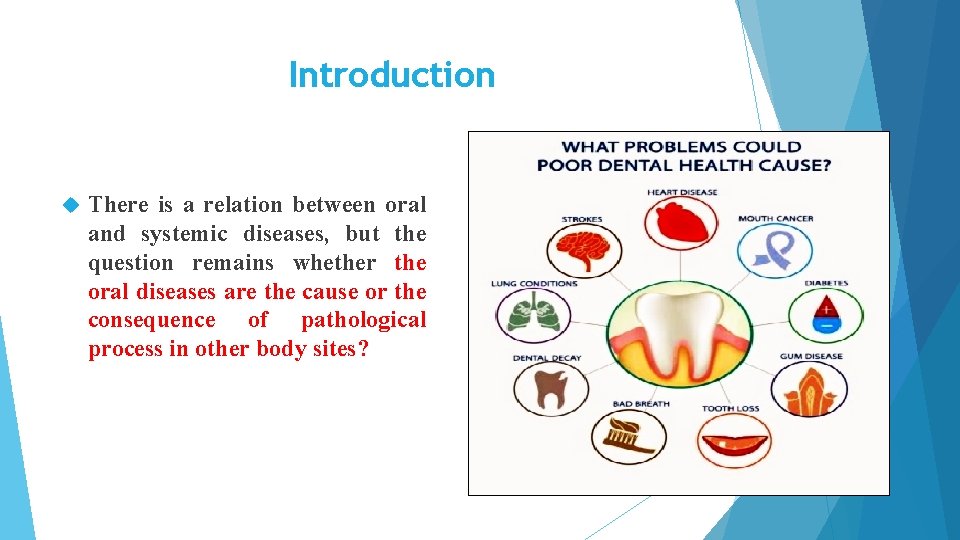
Introduction There is a relation between oral and systemic diseases, but the question remains whether the oral diseases are the cause or the consequence of pathological process in other body sites?

Introduction Obesity : a body mass index (BMI) >30. 0 kg/m 2 It is a major public health problem today. Obesity is a risk factor for several chronic diseases, most notably hypertension, dyslipidemia, coronary heart disease & diabetes. Diabetes is a chronic disease that occurs either when the pancreas does not produce enough insulin (Type 1 diabetes) or when the body cannot effectively use the insulin it produces (Type 2 diabetes) (WHO, 2016) International Diabetes Federation statistics , 2015 In 2015 1/11 adults (415 million) have diabetes By 2040 1/10 adults (642 million) will have diabetes

Introduction Biomarkers A biomarker, or biological marker, generally refers to a measurable indicator of physiologic health, a pathogenic process, or a pharmacologic response to a therapeutic intervention. Whether produced by healthy individuals or by individuals affected by a particular systemic disease, these molecules can be used to monitor health status, disease onset, treatment response and outcome. Human saliva is an ultra-filtrate from plasma; thus, it can be used as an alternative to serum for the detection of diagnostic biomarkers (Jinhua et al. , 2012)

Introduction Resist-in: (resist insulin) an adipo-cytokine, produced by adipocytes and macrophages. It was originally proposed as the link between obesity and insulin resistance in mice. Insulin resistance is a fundamental aspect of the etiology of type 2 diabetes. Previous studies reported a positive correlation between serum and salivary resistin, which were both correlated to BMI in type 2 diabetes patients (Jinhua et al. , 2012) Data suggests that resistin could be one of the molecular links connecting obesity, diabetes and periodontitis, and may serve as a marker that links periodontal disease with other systemic diseases (Archana et al. , 2014).

Introduction After smoking, obesity is the second strongest risk factor for inflammatory periodontal tissue destruction (Jagannathachary et al. , 2010) The relationship between diabetes and periodontal disease : Diabetes is a risk factor for severe periodontal diseases Periodontitis is a risk factor for worsening blood glucose control in patients with diabetes and may also increase the risk of diabetic complications
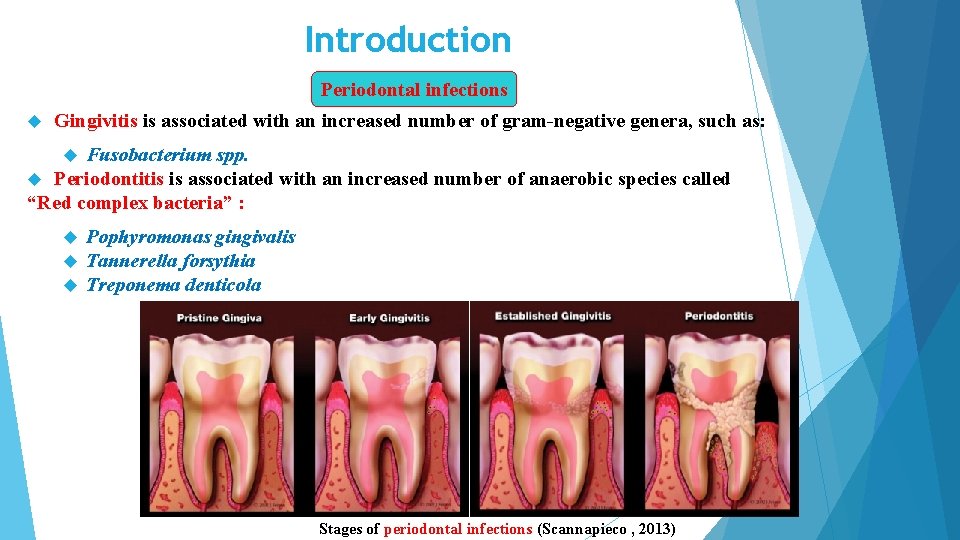
Introduction Periodontal infections Gingivitis is associated with an increased number of gram-negative genera, such as: Fusobacterium spp. Periodontitis is associated with an increased number of anaerobic species called “Red complex bacteria” : Pophyromonas gingivalis Tannerella forsythia Treponema denticola Stages of periodontal infections (Scannapieco , 2013)

Introduction The oral microbiome plays a relevant role in human health and it is a key element in a variety of oral and systemic diseases. Traditional culture techniques used to identify oral bacteria have significant shortcomings : • • • Difficulty in culturing: some oral bacteria need strict anaerobic conditions & some need special media as they are very fastidious in nature. Non-cultivability of many oral species. Low sensitivity: bacteria in limited amount can not be detected Only viable bacteria can grow on culture; therefore, strict sampling and transport conditions are essential Time-consuming and expensive (Topcuoglu & Kulekci , 2015). The use of molecular techniques facilitates the characterization of both cultivable and non-cultivable members of the oral microbiome. This leads to the identification of many oral bacterial species which have never been cultivated and identified. ≥ 1, 000 phylotypes could potentially colonize the oral cavity (Scannapieco, 2013)

Objectives We aim to compare the bacterial community and the level of selected biomarkers in the saliva of adults in health and disease conditions. Obese (diabetics and non-diabetics) were compared with non-obese adults: Resistin Bacteria (as a biomarker of insulin resistance) associated with dental infections

y g o l o d o h t e M

Methodology A cross sectional study (December 2015 - April 2016) Studypopulation Patients attending to the University of Sharjah Dental Hospital were invited to participate in the study Consent form Medical history & random blood glucose measurement Anthropometric measures: (weight & height) BMI calculation : weight (kg) divided by squared height (m 2) BMI ≥ 30 : obese , BMI < 30 : nonobese * *CDC (2016)
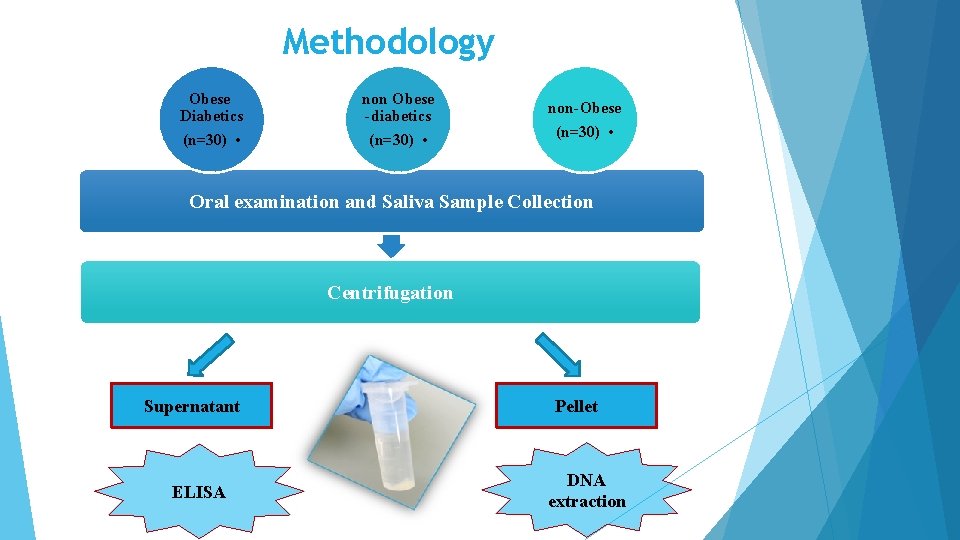
Methodology Obese Diabetics (n=30) • non Obese -diabetics (n=30) • non-Obese (n=30) • Oral examination and Saliva Sample Collection Centrifugation Supernatant ELISA Pellet DNA extraction

Methodology Quantitative measurement of Resistin concentrations in saliva samples Determination of salivary resistin concentration by sandwich ELISA Absorbance values (at wavelength of 450 nm) were measured using Micro Plate Reader (Hospitex Diagnostics, Italy) A Standard Curve was generated using the standard concentrations (0. 25 -16 ng/m. L) on the x-axis and the corresponding mean 450 nm absorbance on the y-axis.

Methodology Real-time PCR was carried out with Rotor-Gene® Q PCR thermocycler (Qiagen, Germany). Amplification was performed using master mix containing Eva Green as a florescent dye Oral Infection Bacterial species (target gene) Reference Porphyromonas gingivalis (16 S r. RNA) Kuboniwa et al. , 2004 Treponema denticola (16 S r. RNA) Yoshida et al. , 2004 Tannerella forsythia (16 S r. RNA) Shelburne et al. , 2000 Actinobacillus actinomycetemcomitans (ikt. A) Yoshida et al. , 2003 Gingivitis Fusobacterium spp. (16 S r. RNA) Suzuki et al. , 2004 Dental caries Bifidobacterium (16 S r. RNA) Matsuki et al. , 2002 Universal primers (16 S r. RNA) Nadkarni et al. , 2002 Periodontitis The fold-difference (N) in the number of the target organism-specific gene copies relative to the number of 16 S r. RNA gene copies was determined as follows: N = 2 - ∆ Ct = Ct target – Ct Universal primers 16 S r. RNA (Kabeerdoss, et al. 2013).

n o i s s u c s i D & Results
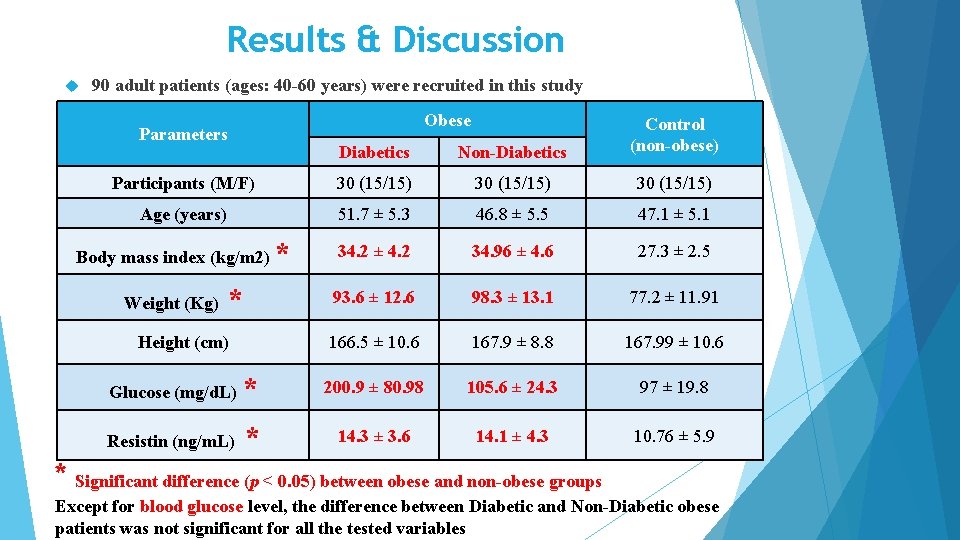
Results & Discussion 90 adult patients (ages: 40 -60 years) were recruited in this study Obese Diabetics Non-Diabetics Control (non-obese) Participants (M/F) 30 (15/15) Age (years) 51. 7 ± 5. 3 46. 8 ± 5. 5 47. 1 ± 5. 1 34. 2 ± 4. 2 34. 96 ± 4. 6 27. 3 ± 2. 5 93. 6 ± 12. 6 98. 3 ± 13. 1 77. 2 ± 11. 91 166. 5 ± 10. 6 167. 9 ± 8. 8 167. 99 ± 10. 6 Parameters Body mass index (kg/m 2) Weight (Kg) * Height (cm) * Glucose (mg/d. L) * 200. 9 ± 80. 98 105. 6 ± 24. 3 97 ± 19. 8 Resistin (ng/m. L) * 14. 3 ± 3. 6 14. 1 ± 4. 3 10. 76 ± 5. 9 * Significant difference (p < 0. 05) between obese and non-obese groups Except for blood glucose level, the difference between Diabetic and Non-Diabetic obese patients was not significant for all the tested variables
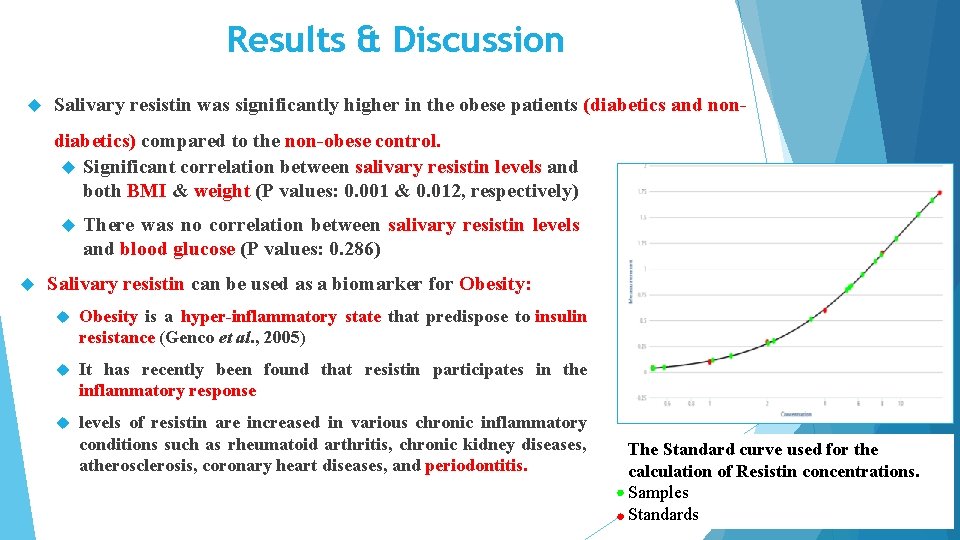
Results & Discussion Salivary resistin was significantly higher in the obese patients (diabetics and nondiabetics) compared to the non-obese control. Significant correlation between salivary resistin levels and both BMI & weight (P values: 0. 001 & 0. 012, respectively) There was no correlation between salivary resistin levels and blood glucose (P values: 0. 286) Salivary resistin can be used as a biomarker for Obesity: Obesity is a hyper-inflammatory state that predispose to insulin resistance (Genco et al. , 2005) It has recently been found that resistin participates in the inflammatory response levels of resistin are increased in various chronic inflammatory conditions such as rheumatoid arthritis, chronic kidney diseases, atherosclerosis, coronary heart diseases, and periodontitis. The Standard curve used for the calculation of Resistin concentrations. Samples Standards
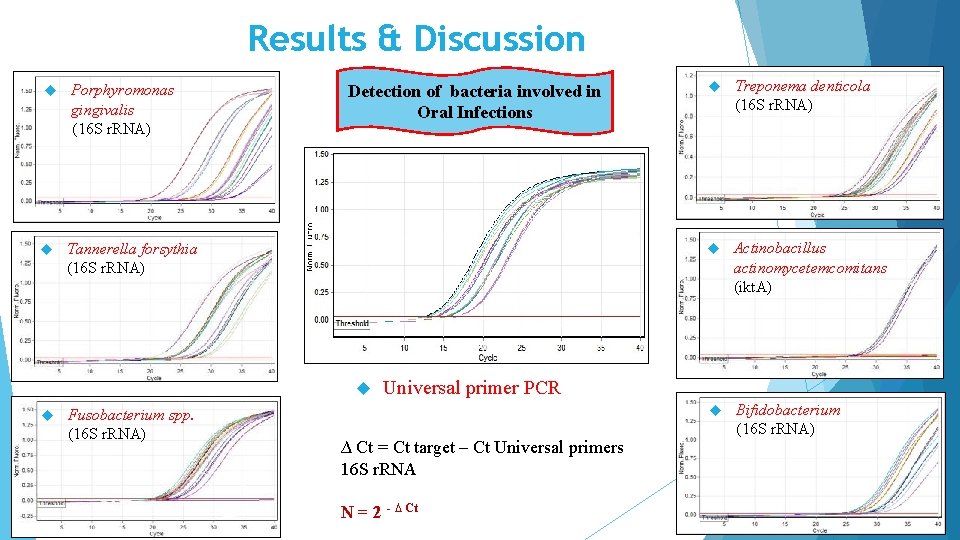
Results & Discussion Porphyromonas gingivalis (16 S r. RNA) Detection of bacteria involved in Oral Infections Tannerella forsythia (16 S r. RNA) Fusobacterium spp. (16 S r. RNA) Treponema denticola (16 S r. RNA) Actinobacillus actinomycetemcomitans (ikt. A) Bifidobacterium (16 S r. RNA) Universal primer PCR ∆ Ct = Ct target – Ct Universal primers 16 S r. RNA N = 2 - ∆ Ct
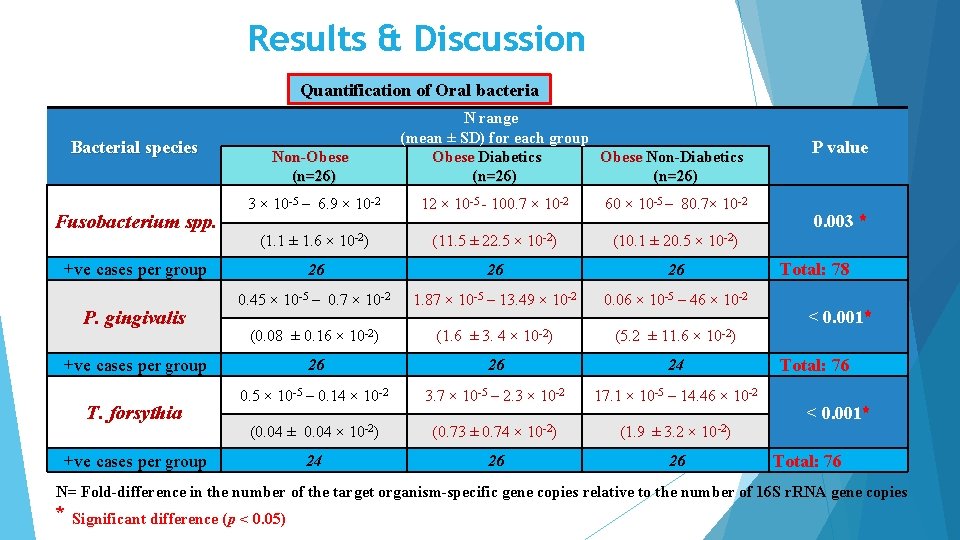
Results & Discussion Quantification of Oral bacteria Bacterial species Fusobacterium spp. Non-Obese (n=26) 3 × 10 -5 – 6. 9 × 10 -2 12 × 10 -5 - 100. 7 × 10 -2 60 × 10 -5 – 80. 7× 10 -2 (1. 1 ± 1. 6 × 10 -2) (11. 5 ± 22. 5 × 10 -2) (10. 1 ± 20. 5 × 10 -2) 26 26 26 0. 45 × 10 -5 – 0. 7 × 10 -2 1. 87 × 10 -5 – 13. 49 × 10 -2 0. 06 × 10 -5 – 46 × 10 -2 (0. 08 ± 0. 16 × 10 -2) (1. 6 ± 3. 4 × 10 -2) (5. 2 ± 11. 6 × 10 -2) 26 26 24 0. 5 × 10 -5 – 0. 14 × 10 -2 3. 7 × 10 -5 – 2. 3 × 10 -2 17. 1 × 10 -5 – 14. 46 × 10 -2 (0. 04 ± 0. 04 × 10 -2) (0. 73 ± 0. 74 × 10 -2) (1. 9 ± 3. 2 × 10 -2) 24 26 26 +ve cases per group P. gingivalis +ve cases per group T. forsythia N range (mean ± SD) for each group Obese Diabetics Obese Non-Diabetics (n=26) +ve cases per group P value 0. 003 * Total: 78 < 0. 001* Total: 76 N= Fold-difference in the number of the target organism-specific gene copies relative to the number of 16 S r. RNA gene copies * Significant difference (p < 0. 05)
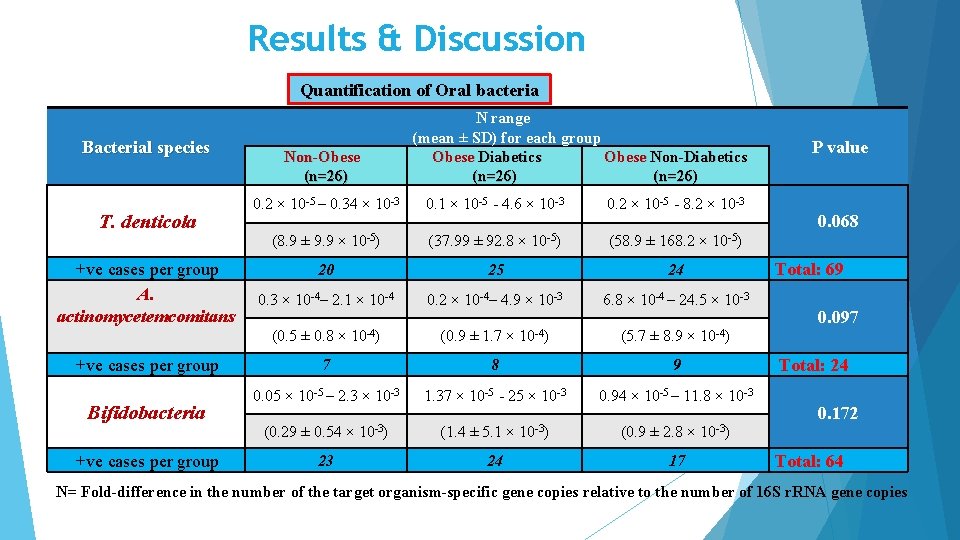
Results & Discussion Quantification of Oral bacteria Bacterial species T. denticola +ve cases per group A. actinomycetemcomitans +ve cases per group Bifidobacteria +ve cases per group Non-Obese (n=26) N range (mean ± SD) for each group Obese Diabetics Obese Non-Diabetics (n=26) 0. 2 × 10 -5 – 0. 34 × 10 -3 0. 1 × 10 -5 - 4. 6 × 10 -3 0. 2 × 10 -5 - 8. 2 × 10 -3 (8. 9 ± 9. 9 × 10 -5) (37. 99 ± 92. 8 × 10 -5) (58. 9 ± 168. 2 × 10 -5) 20 25 24 0. 3 × 10 -4– 2. 1 × 10 -4 0. 2 × 10 -4– 4. 9 × 10 -3 6. 8 × 10 -4 – 24. 5 × 10 -3 (0. 5 ± 0. 8 × 10 -4) (0. 9 ± 1. 7 × 10 -4) (5. 7 ± 8. 9 × 10 -4) 7 8 9 0. 05 × 10 -5 – 2. 3 × 10 -3 1. 37 × 10 -5 - 25 × 10 -3 0. 94 × 10 -5 – 11. 8 × 10 -3 (0. 29 ± 0. 54 × 10 -3) (1. 4 ± 5. 1 × 10 -3) (0. 9 ± 2. 8 × 10 -3) 23 24 17 P value 0. 068 Total: 69 0. 097 Total: 24 0. 172 Total: 64 N= Fold-difference in the number of the target organism-specific gene copies relative to the number of 16 S r. RNA gene copies
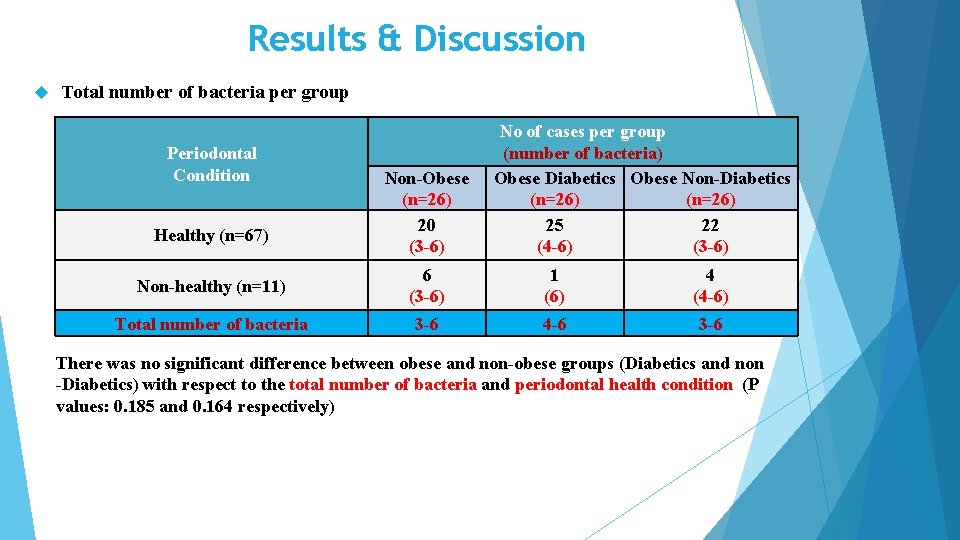
Results & Discussion Total number of bacteria per group Periodontal Condition Healthy (n=67) Non-Obese (n=26) 20 (3 -6) No of cases per group (number of bacteria) Obese Diabetics Obese Non-Diabetics (n=26) 25 22 (4 -6) (3 -6) Non-healthy (n=11) 6 (3 -6) 1 (6) 4 (4 -6) Total number of bacteria 3 -6 4 -6 3 -6 There was no significant difference between obese and non-obese groups (Diabetics and non -Diabetics) with respect to the total number of bacteria and periodontal health condition (P values: 0. 185 and 0. 164 respectively)
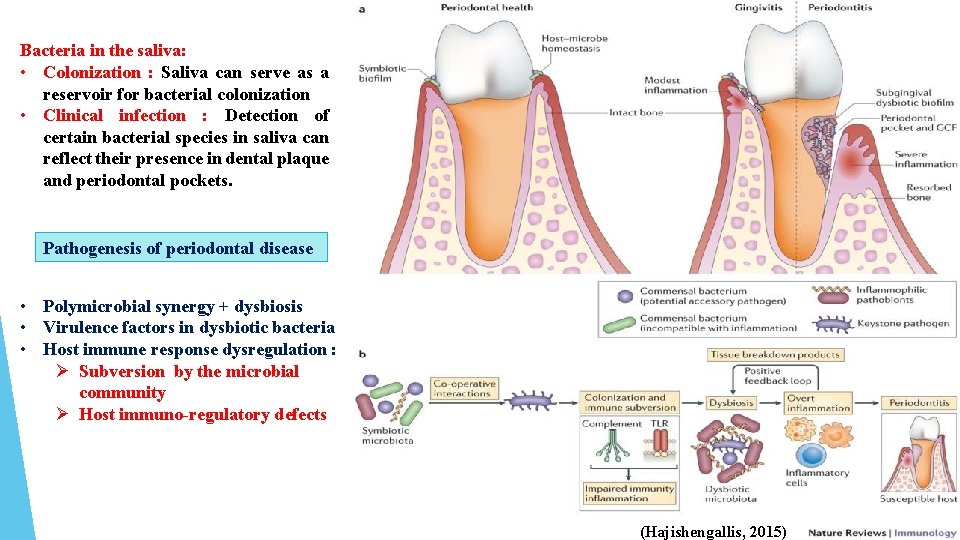
Bacteria in the saliva: • Colonization : Saliva can serve as a reservoir for bacterial colonization • Clinical infection : Detection of certain bacterial species in saliva can reflect their presence in dental plaque and periodontal pockets. Pathogenesis of periodontal disease • Polymicrobial synergy + dysbiosis • Virulence factors in dysbiotic bacteria • Host immune response dysregulation : Ø Subversion by the microbial community Ø Host immuno-regulatory defects (Hajishengallis, 2015)

Results & Discussion No correlation was found between the levels of salivary resistin and: The total number of oral bacteria. quantity of different oral bacteria. Both salivary resistin & certain oral bacterial species [Fusobacterium (associated with gingivitis), P. gingivalis and T. forsythia (associated with periodontitis)] were detected in significantly higher quantities in the obese patients (diabetics and nondiabetics) compared to the non-obese control Previous research showed incremental elevation of resistin with periodontal disease activity and a reduced level of resistin, after periodontal therapy (Archana et al, 2014). In animal studies, it was found that the expression of resistin can be upregulated by microbial antigens such as lipopolysaccharide, a component of the cell wall of Gram-negative bacteria that has been demonstrated to induce inflammatory reactions (Lu et al, 2002). The connection is likely made through inflammation, initiated and propagated by the actions of oral biofilms, which exacerbates chronic systemic inflammation in obese individuals. Increased systemic inflammation has been linked to insulin resistance and the development of diabetes, as well as its complications (Scannapieco, 2013)
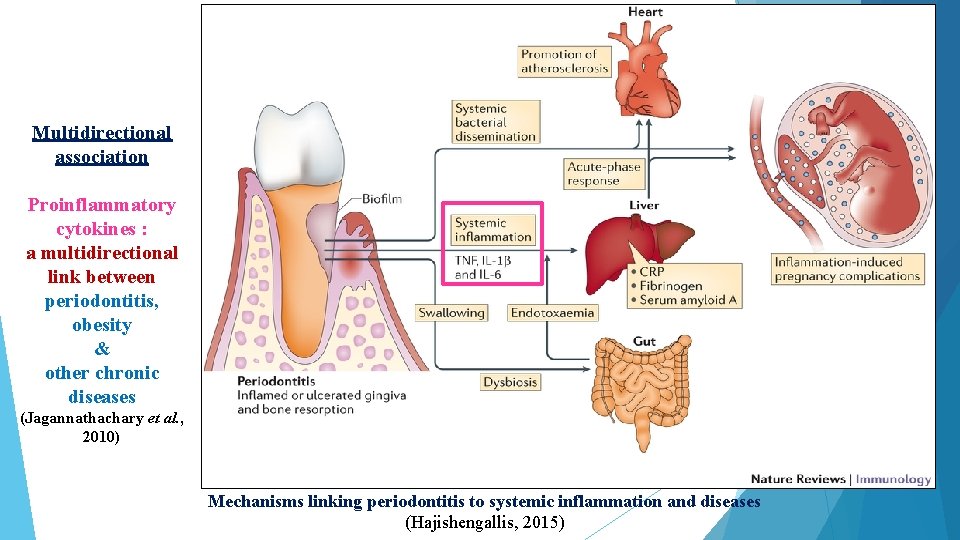
Multidirectional association Proinflammatory cytokines : a multidirectional link between periodontitis, obesity & other chronic diseases (Jagannathachary et al. , 2010) Mechanisms linking periodontitis to systemic inflammation and diseases (Hajishengallis, 2015)

Conclusion
Conclusion Saliva as a diagnostic fluid This study highlighted the importance of saliva as a non-invasive sample for the detection of biomarkers and microbes associated with oral and systemic diseases. Detection of biomarkers Saliva collection is a non-invasive It may represent an alternative for patients in whom blood drawing is difficult (e. g. children and older patients) Fast screening of large population it can be performed by individuals with modest training, including patients themselves. This may pave the way for the development of salivary screening tests that can be done by patients at home

Conclusion Saliva as a diagnostic fluid Detection of microbes Using saliva as a PCR template reflects the overall condition of the oral cavity Relative quantification is better than absolute quantification, which requires very precise sample collection (Yoshida et al. , 2002). Quantitative real-time PCR of oral biofilms : Diagnosis : establishing the etiology of oral infectious diseases, especially unculturable oral bacteria Prognosis: monitoring the effect of therapy and evaluating treatment. Salivary diagnosis is poised to revolutionize the delivery of health and dental care by providing chairside, non-invasive diagnosis and health monitoring, which can contribute to the development of personalized medicine and personalized dental medicine.

Recommendations Periodontal diseases are silent and chronic in nature; thus, many patients do not realize they have them. Meanwhile, physicians may not know that the patient has a condition that affects sugar control and makes diabetes management more difficult The relationship between oral and general health will challenge dentists & physicians to work together in managing patients with periodontal diseases and systemic diseases like diabetes. Patients with diabetes should consult a dentist for periodontal screening Patients with periodontal disease should be screened for diabetes if signs or symptoms are present. Dentists should explain to the obese individuals about the possible oral complications of obesity, and should follow up their oral condition in order to diminish morbidity associated with obesity.

References Archana D. , Rahul K. , Nagappa G. , D. Gopalakrishnan, and Paulami B. Resistin: A Potential Biomarker for Periodontitis Influenced Diabetes Mellitus and Diabetes Induced Periodontitis. Disease Markers. 2014; vol. 2014, Article ID 930206 Matsuki TK, Watanabe J, Fujimoto Y, Miyamoto T, Takada K, Matsumoto K, et al. Development of 16 S r. RNA-gene-targeted group-specific primers for the detection and identification of predominant bacteria in human feces. Appl Environ Microbiol. 2002; 68: 544551. Nadkarni MA, Martin FE, Jacques NA, Hunter N. Determination of bacterial load by real-time PCR using a broad-range (universal) probe and primers set. Microbiology. 2002; 148: 257 -266. Shelburne CE, Prabhu A, Gleason RM, Mullally BH, Coulter WA. Quantitation of Bacteroides forsythus in subgingival plaque comparison of immunoassay and quantitative polymerase chain reaction. J Microbiol Methods. 2000; 39: 97 -107. Suzuki N, Yoshida A, Saito T, Kawada M, Nakano Y. Quantitative microbiological study of subgingival plaque by real-time PCR shows correlation between levels of Tannerella forsythensis and Fusobacterium spp. J Clin Microbiol. 2004; 42: 2255 -2257. Yoshida A, Suzuki N, Nakano Y, Oho T, Kawada M, Koga T. Development of a 5′fluorogenic nuclease-based real-time PCR assay for quantitative detection of Actinobacillus actinomycetemcomitans and Porphyromonas gingivalis. J Clin Microbiol. 2003; 41: 863 -866. Yoshida A, Kawada M, Suzuki N, Nakano Y, Oho T, Saito T, Yamashita Y. Taq. Man real-time polymerase chain reaction assay for the correlation of Treponema denticola numbers with the severity of periodontal disease. Oral Microbiol Immunol. 2004; 19: 196 -200.

References Hajishengallis G. Periodontitis: from microbial immune subversion to systemic inflammation. Nature reviews Immunology. 2015; 15(1): 30 -44. Jinhua Y. , Hongfei G. , Jing Y. , Lu X. , and Ming L. Measurement of Salivary Resistin Level in Patients with Type 2 Diabetes. International Journal of Endocrinology. 2012; vol. 2012, Article ID 359724 Jagannathachary S, Kamaraj D. Obesity and periodontal disease. Journal of Indian Society of Periodontology. 2010; 14(2): 96 -100. Genco RJ, Grossi SG, Ho A, Nishimura F, Murayama Y. A proposed model linking inflammation to obesity, diabetes, and periodontal infections. J Periodontol. 2005; 76: 2075– 84 Scannapieco FA. The Oral Microbiome: Its Role in Health and in Oral and Systemic Infections. Clinical Microbiology Newsletter. 2013; 35(20): 163– 169 Suzuki N, Yoneda M, Hirofuji T. Mixed Red-Complex Bacterial Infection in Periodontitis. International Journal of Dentistry. 2013; 2013: 587279. Topcuoglu N & Kulekci G. 16 S r. RNA based microarray analysis of ten periodontal bacteria in patients with different forms of periodontitis. Anaerobe. 2015; 35(Pt A): 35 -40. Kuboniwa M, Amano A, Kimura KR, Sekine S, Kato S, Yamamoto Y, Okahashi N, Iida T, Shizukuishi S. Quantitative detection of periodontal pathogens using real-time polymerase chain reaction with Taq. Man probes. Oral Microbiol Immunol. 2004; 19: 168 -176. Lu SC, Shieh WY, Chen CY, Hsu SC, Chen HL. Lipopolysaccharide increases resistin gene expression in vivo and in vitro. FEBS Lett. 2002; 530 (1– 3): 158– 62

Acknowledgment University of Sharjah for supporting this project Members of the Wound Healing & Oral Diagnosis Research Group The Director and researchers in the Sharjah Institute for Medical Research, University of Sharjah, UAE The students (College of Dental Medicine, University of Sharjah, UAE) who collected the samples : Walaa Maher Yasmina Waleed Yassmina Yasser Farah Ghanim Ahmed Alghafri Ahmed Sheriff Feras Mohammed

Thanks for your kind attention
- Slides: 33