The Ohio State University Nonequilibrium Thermodynamics Laboratories ChemiIonization

The Ohio State University Nonequilibrium Thermodynamics Laboratories Chemi-Ionization and Visible/UV Emission from Supersonic Flows of Combustion Products Saurabh Keshav, Yurii G. Utkin, J. William Rich and Igor V. Adamovich Dept. of Mechanical Engineering The Ohio State University Support AFOSR, Space Power and Propulsion Program

The Ohio State University Nonequilibrium Thermodynamics Laboratories Motivation q Control of UV / visible emission from high altitude rocket plumes q Study combustion diagnostic by chemi-ionization Objective q Study energy transfer, chemi-ionization, and emission in supersonic flows of combustion products
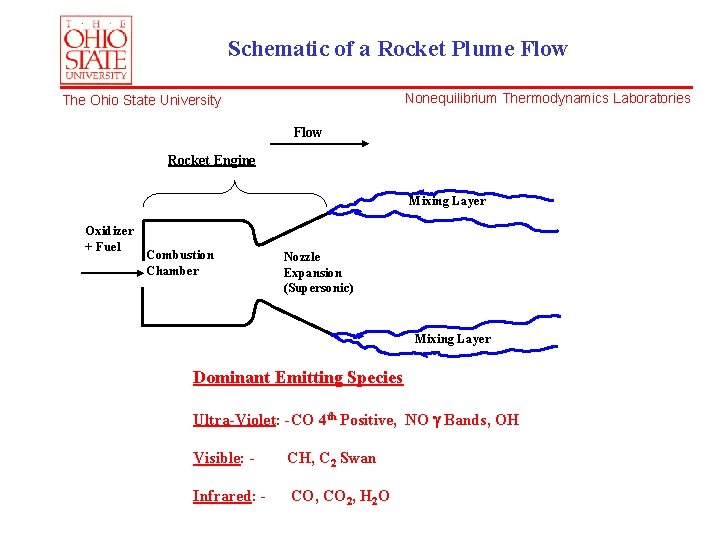
Schematic of a Rocket Plume Flow Nonequilibrium Thermodynamics Laboratories The Ohio State University Flow Rocket Engine Mixing Layer Oxidizer + Fuel Combustion Chamber Nozzle Expansion (Supersonic) Mixing Layer Dominant Emitting Species Ultra-Violet: -CO 4 th Positive, NO Bands, OH Visible: - CH, C 2 Swan Infrared: - CO, CO 2, H 2 O
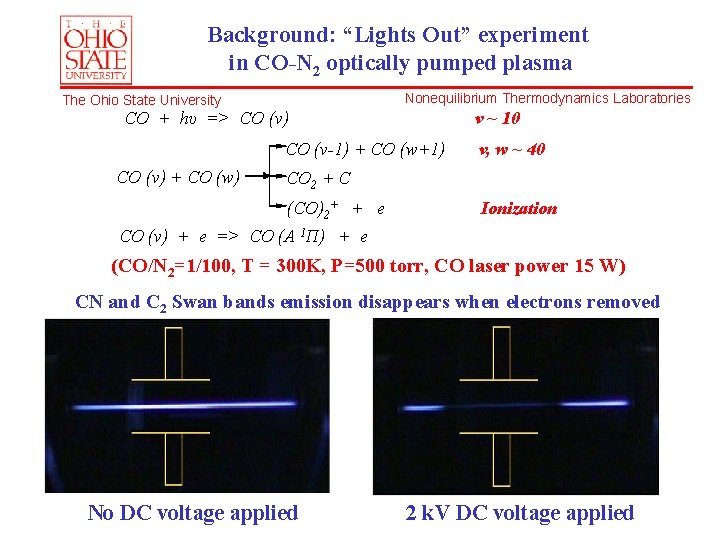
Background: “Lights Out” experiment in CO-N 2 optically pumped plasma Nonequilibrium Thermodynamics Laboratories The Ohio State University CO + hυ => CO (v) v ~ 10 CO (v-1) + CO (w+1) CO (v) + CO (w) v, w ~ 40 CO 2 + C (CO)2+ + e Ionization CO (v) + e => CO (A 1Π) + e (CO/N 2=1/100, T = 300 K, P=500 torr, CO laser power 15 W) CN and C 2 Swan bands emission disappears when electrons removed No DC voltage applied 2 k. V DC voltage applied
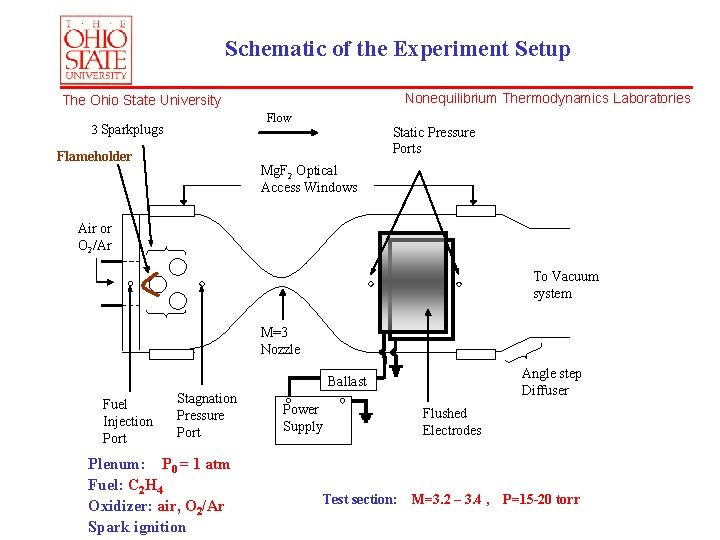
Schematic of the Experiment Setup Nonequilibrium Thermodynamics Laboratories The Ohio State University Flow 3 Sparkplugs Static Pressure Ports Flameholder Mg. F 2 Optical Access Windows Air or O 2/Ar To Vacuum system M=3 Nozzle Angle step Diffuser Ballast Fuel Injection Port Stagnation Pressure Port Plenum: P 0 = 1 atm Fuel: C 2 H 4 Oxidizer: air, O 2/Ar Spark ignition Power Supply Flushed Electrodes Test section: M=3. 2 – 3. 4 , P=15 -20 torr

Background theory on Chemi- Ionization and Electron Removal The Ohio State University Nonequilibrium Thermodynamics Laboratories Combustion Chamber q CH + O => CHO+ + e q Electronically excited, radiating species (CH*, C 2*, OH*, O*, H*) are created q Do electrons help excited species generation? Removal of Electrons from M = 3 flow q Applying voltage to electrodes to draw off electrons (Thomson discharge)
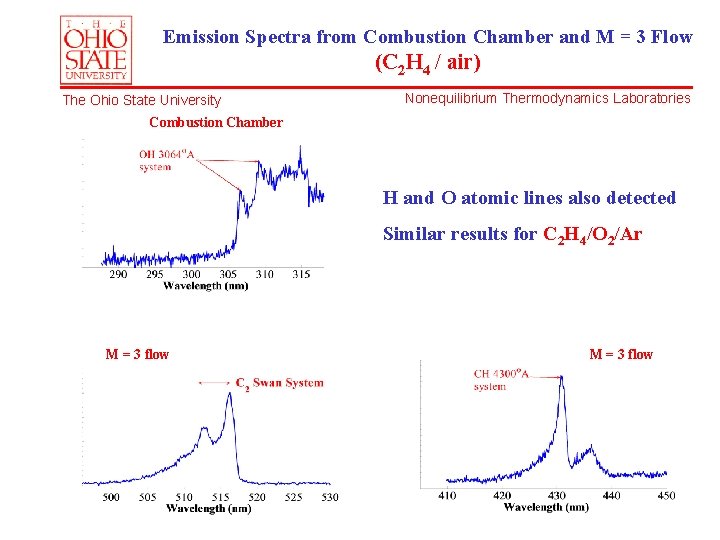
Emission Spectra from Combustion Chamber and M = 3 Flow (C 2 H 4 / air) The Ohio State University Nonequilibrium Thermodynamics Laboratories Combustion Chamber H and O atomic lines also detected Similar results for C 2 H 4/O 2/Ar M = 3 flow
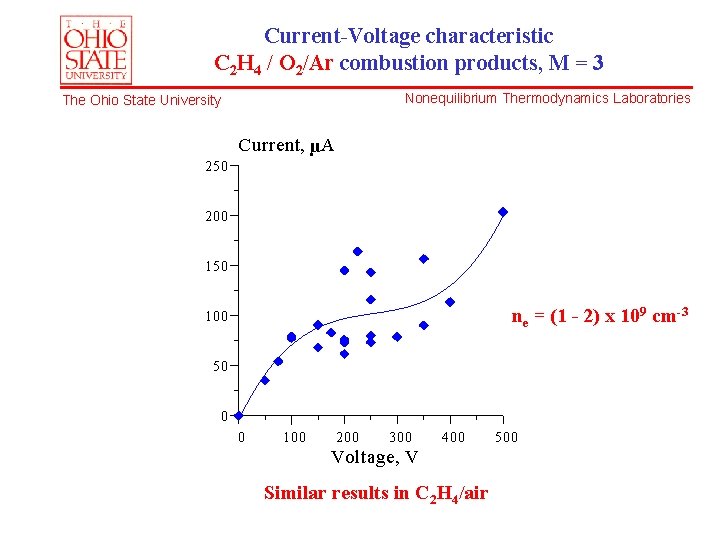
Current-Voltage characteristic C 2 H 4 / O 2/Ar combustion products, M = 3 Nonequilibrium Thermodynamics Laboratories The Ohio State University Current, µA 250 200 150 ne = (1 - 2) x 109 cm-3 100 50 0 0 100 200 300 400 Voltage, V Similar results in C 2 H 4/air 500
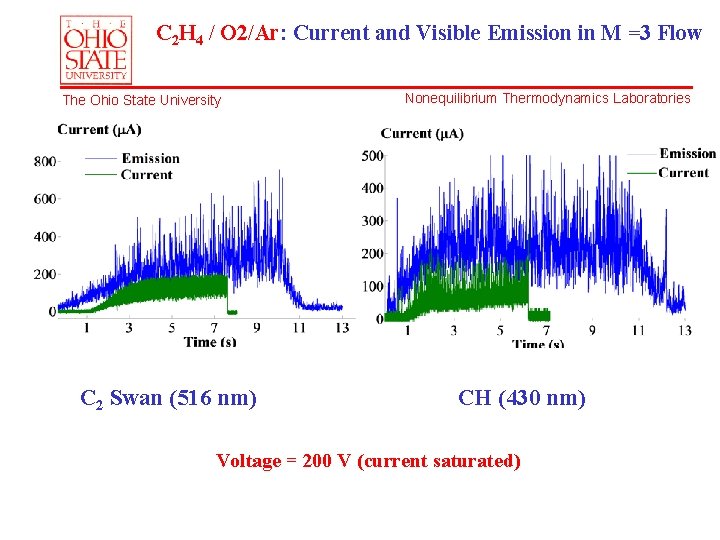
C 2 H 4 / O 2/Ar: Current and Visible Emission in M =3 Flow The Ohio State University C 2 Swan (516 nm) Nonequilibrium Thermodynamics Laboratories CH (430 nm) Voltage = 200 V (current saturated)
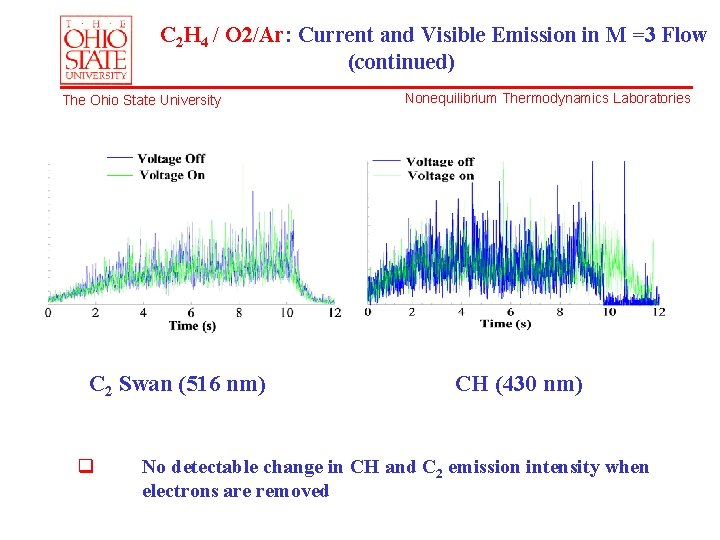
C 2 H 4 / O 2/Ar: Current and Visible Emission in M =3 Flow (continued) The Ohio State University C 2 Swan (516 nm) q Nonequilibrium Thermodynamics Laboratories CH (430 nm) No detectable change in CH and C 2 emission intensity when electrons are removed
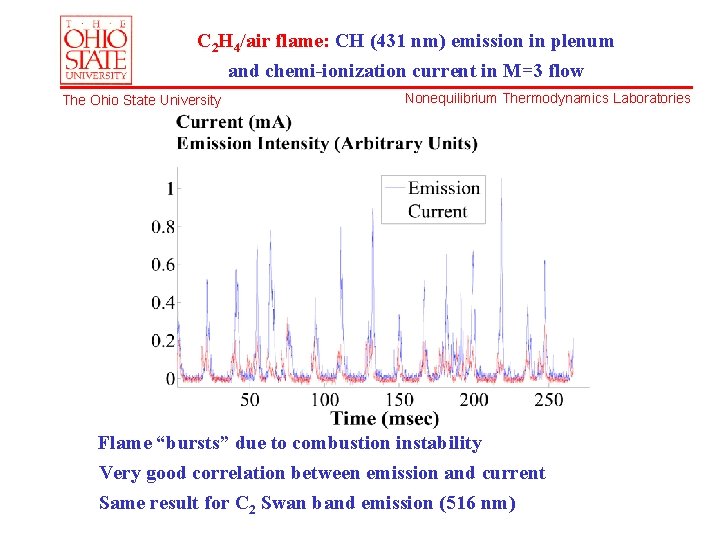
C 2 H 4/air flame: CH (431 nm) emission in plenum and chemi-ionization current in M=3 flow The Ohio State University Nonequilibrium Thermodynamics Laboratories Flame “bursts” due to combustion instability Very good correlation between emission and current Same result for C 2 Swan band emission (516 nm)

Summary The Ohio State University Nonequilibrium Thermodynamics Laboratories q Self sustained combustion and flameholding achieved q Electron density measured in supersonic flows of combustion products, correlated with flame emission q No effect on C 2 Swan band CH emission when electrons are removed

Future work The Ohio State University Nonequilibrium Thermodynamics Laboratories q Further measurements in M = 4 flows and different equivalence ratios in both steady and unsteady flames q Injection of air, N 2, CO, and NO into supersonic flow to study energy transfer from combustion products to these species
- Slides: 13