The Octet Rule When atoms form ions or










- Slides: 13

The Octet Rule • When atoms form ions or combine in compounds, they try to get the electron configuration of the nearest Inert Gas IN OTHER WORDS… they try to get 8 electrons in their outermost shell

Warm-up Exercise!

Where do we look on the Table to get into the Third energy level? • If the first energy level can have 2 e- and the second and subsequent energy levels can each have 8 e- , then what is the first atom to have an e- in the third energy level? Sodium

• It’s important to be able to quickly locate the atoms with a certain number of electrons in the outer shell, or highest energy orbital, because it helps you predict how it will interact with other atoms. • This outer shell determines the Valence of the atom.

Electrovalence (but we can just call it “Valence”) • Definition: Valence - the measure of how much an atom wants to bond with other atoms. Too easy, huh?

• …The valence electrons are the ONLY ones we have to worry about to determine an atoms bonding characteristic! • This outer shell determines the Valence of the atom.

The Octet Rule • When atoms form ions or combine in compounds, they try to get the electron configuration of the nearest Inert Gas IN OTHER WORDS… they try to get 8 electrons in their outermost shell

Valence Electrons • The electrons in the outermost shell – or energy level – of an atom • Periodic – which means they occur in a repeating pattern. – Elements in Group IA have 1 valence electron – Elements in Group IIA have 2 valence electrons – Can you guess how many valence electrons Si (Silicon) has? – Group IVA, so 4 • How about S (Sulfur)? – Group VIA, so 6 • Br (Bromine)? – Group VIIA, so 7

Different types of Bonding • There are 2 bond types : ionic and covalent • In ionic bonding one atom has a stronger attraction for electrons than the other, and “steals” an electron from a second atom • In covalent bonding the attraction for electrons is similar for two atoms. They share their electrons to obtain an octet.

IONIC BONDING • Occurs between a metal and a nonmetal. • The NON-metal “steals” the electron from the metal. WHY? (Think about our discussion of what makes a “happy” atom…

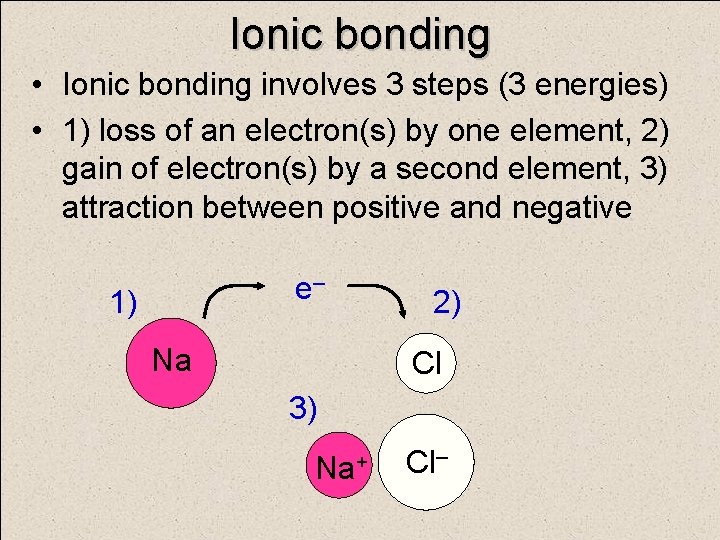
Ionic bonding • Ionic bonding involves 3 steps (3 energies) • 1) loss of an electron(s) by one element, 2) gain of electron(s) by a second element, 3) attraction between positive and negative e– 1) Na 2) Cl 3) Na+ Cl–

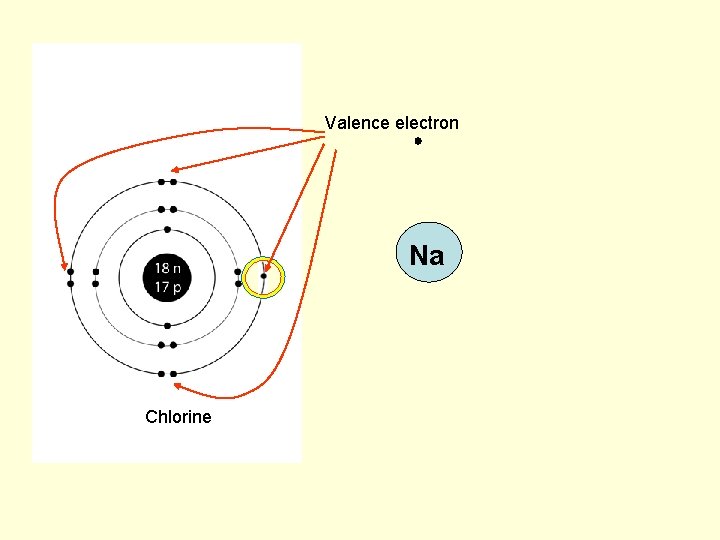
Valence electron Na Chlorine

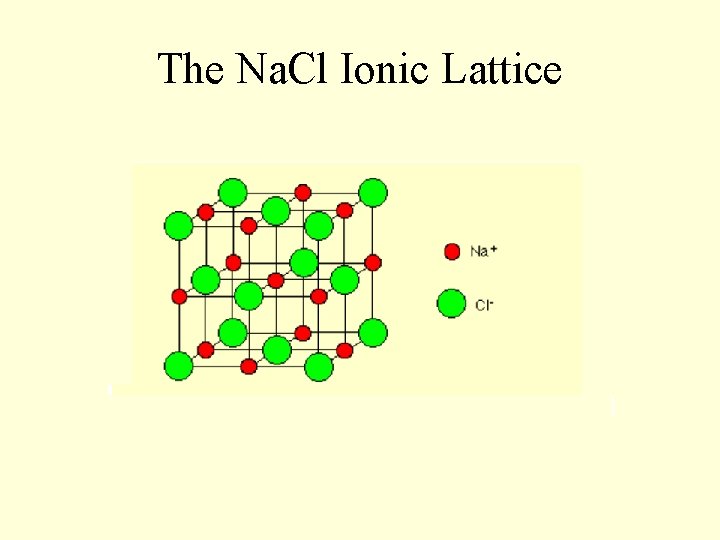
The Na. Cl Ionic Lattice