The Number of Absorptions Protons have different chemical
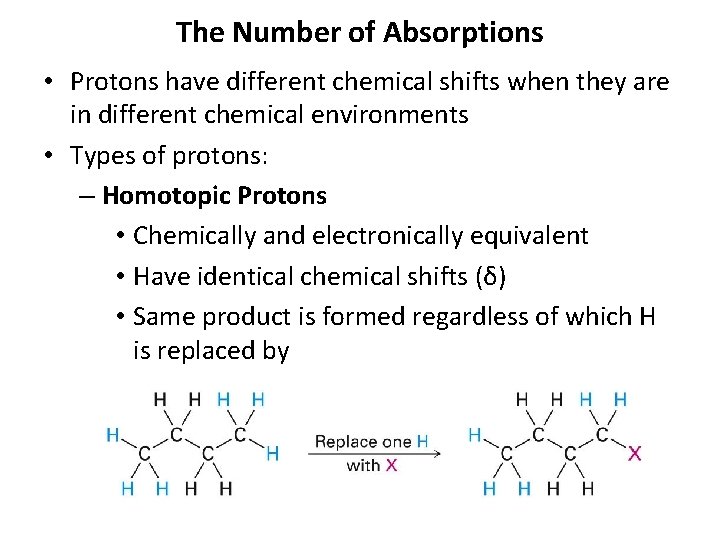
The Number of Absorptions • Protons have different chemical shifts when they are in different chemical environments • Types of protons: – Homotopic Protons • Chemically and electronically equivalent • Have identical chemical shifts (δ) • Same product is formed regardless of which H is replaced by
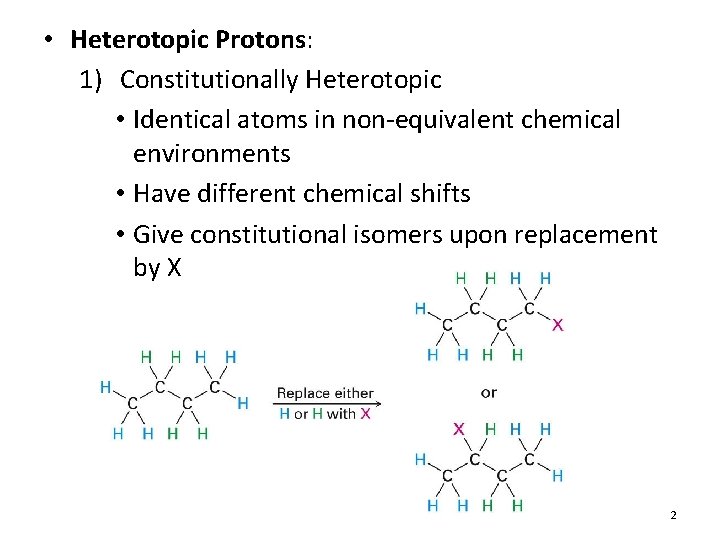
• Heterotopic Protons: 1) Constitutionally Heterotopic • Identical atoms in non-equivalent chemical environments • Have different chemical shifts • Give constitutional isomers upon replacement by X 2
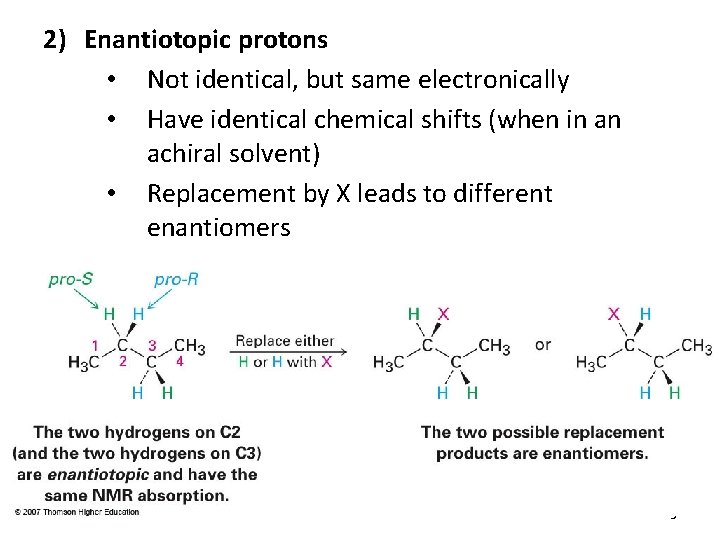
2) Enantiotopic protons • Not identical, but same electronically • Have identical chemical shifts (when in an achiral solvent) • Replacement by X leads to different enantiomers 3
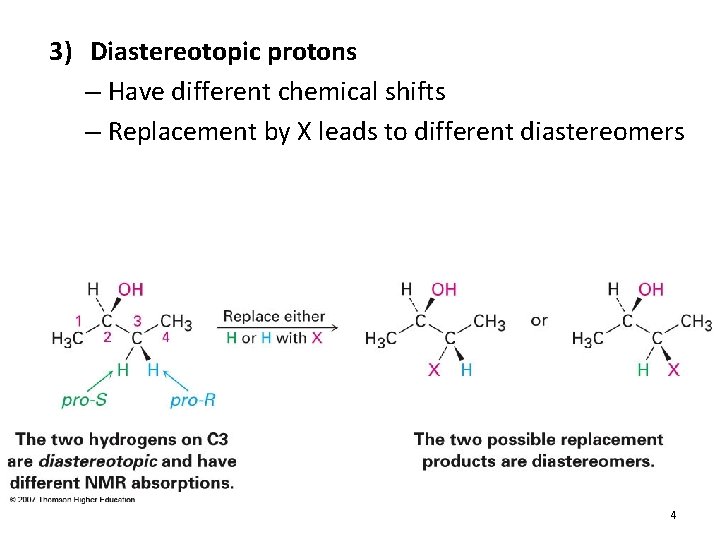
3) Diastereotopic protons – Have different chemical shifts – Replacement by X leads to different diastereomers 4
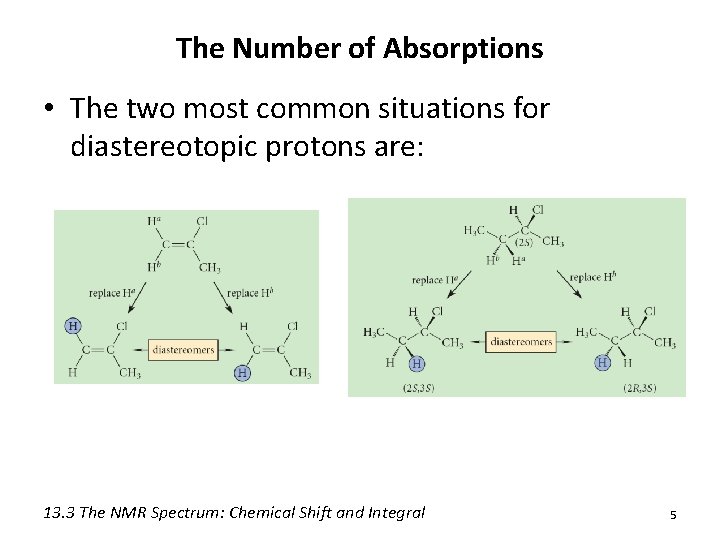
The Number of Absorptions • The two most common situations for diastereotopic protons are: 13. 3 The NMR Spectrum: Chemical Shift and Integral 5
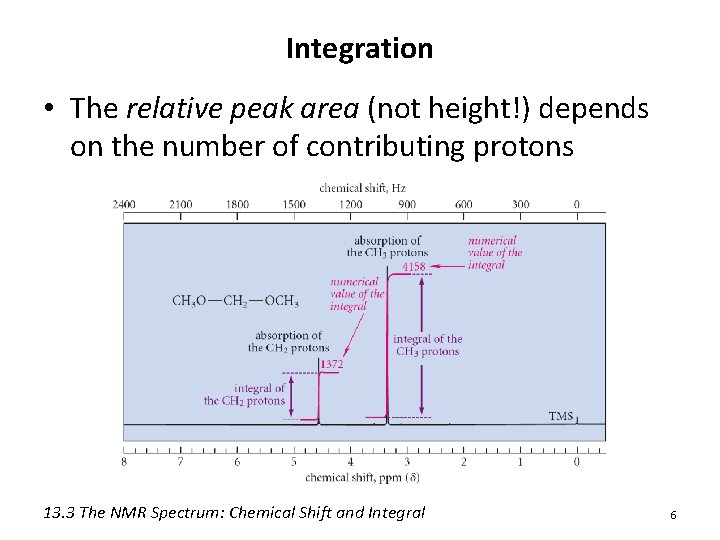
Integration • The relative peak area (not height!) depends on the number of contributing protons 13. 3 The NMR Spectrum: Chemical Shift and Integral 6
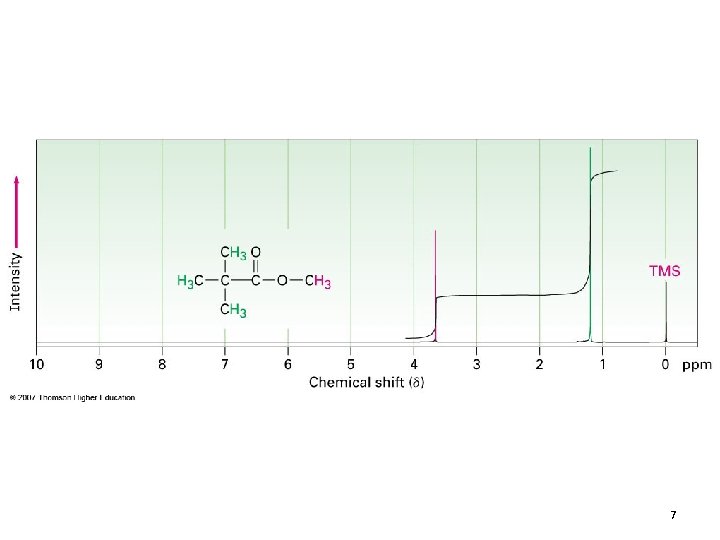
7
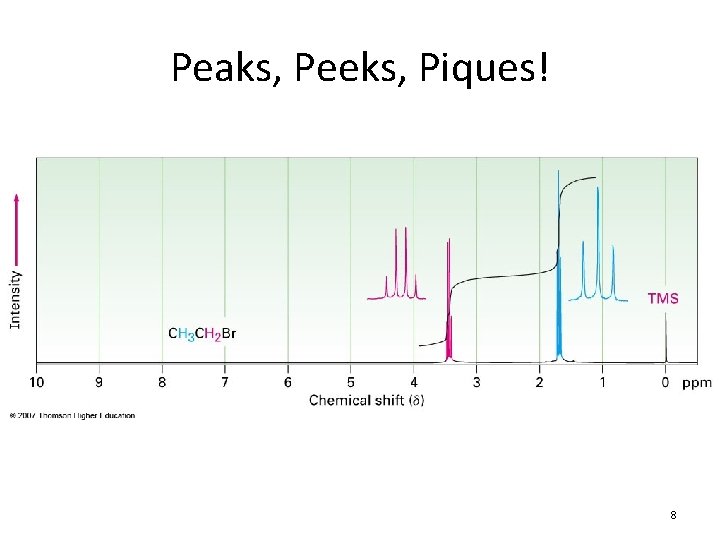
Peaks, Peeks, Piques! 8

The n+1 Rule • n + 1 rule: protons that have n equivalent neighboring protons show n + 1 peaks in their NMR spectrum – Called “Splitting” or “Spin-Spin Splitting” – Arises from the effect that one set of protons has on neighboring protons • Allows you to figure out how many equivalent protons are adjacent to your proton(s) in question – If your absorbance is: • 1 peak – No H’s on adjacent carbons – Singlet 9
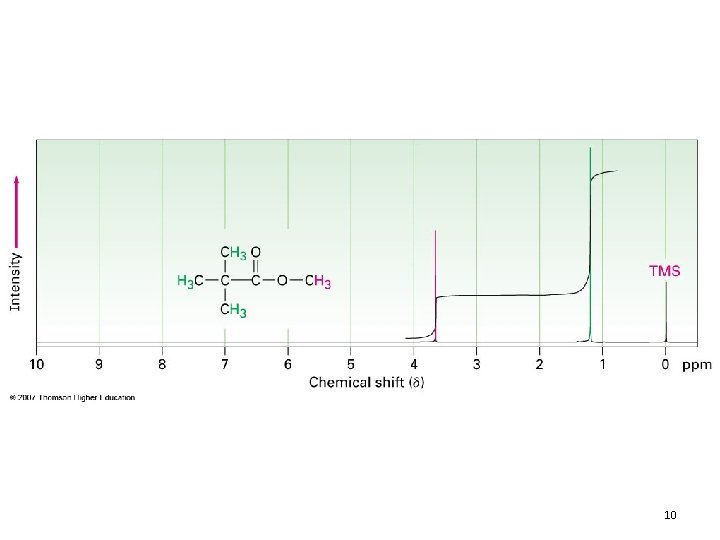
10

• 2 peaks – One H on adjacent carbon – Doublet • 3 peaks – Two adjacent protons – Triplet • Hence, splitting provides connectivity information 11
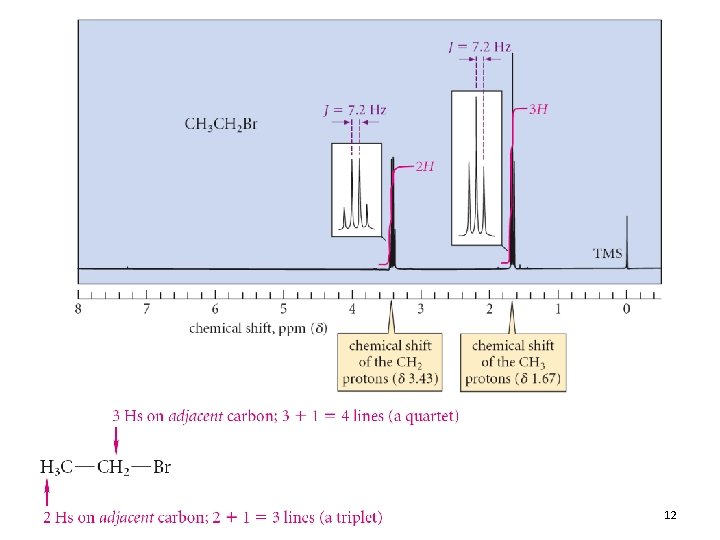
12
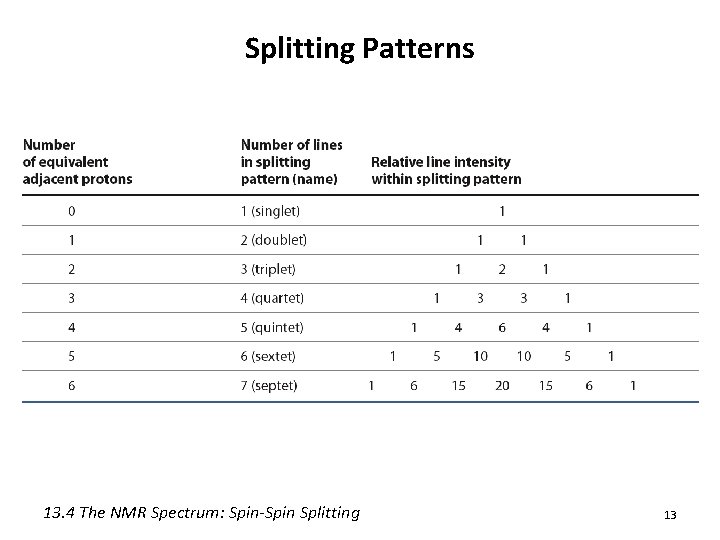
Splitting Patterns 13. 4 The NMR Spectrum: Spin-Spin Splitting 13
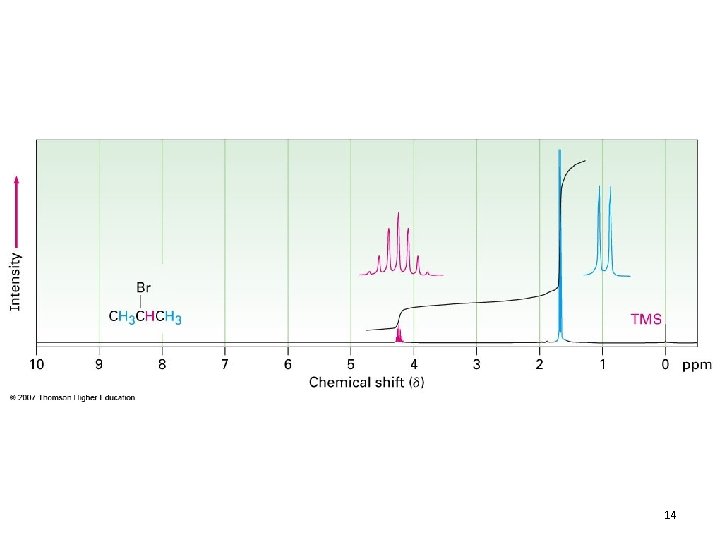
14

Spin-Spin Splitting • Splitting is always mutual: If Ha splits Hb, then Hb must split Ha • Ha and Hb are then said to be coupled • Splitting is not observed between chemically equivalent protons 13. 4 The NMR Spectrum: Spin-Spin Splitting 15
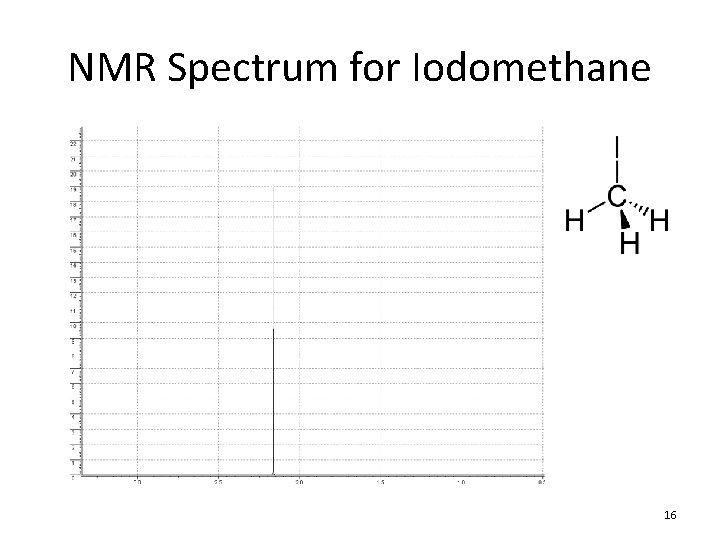
NMR Spectrum for Iodomethane 16
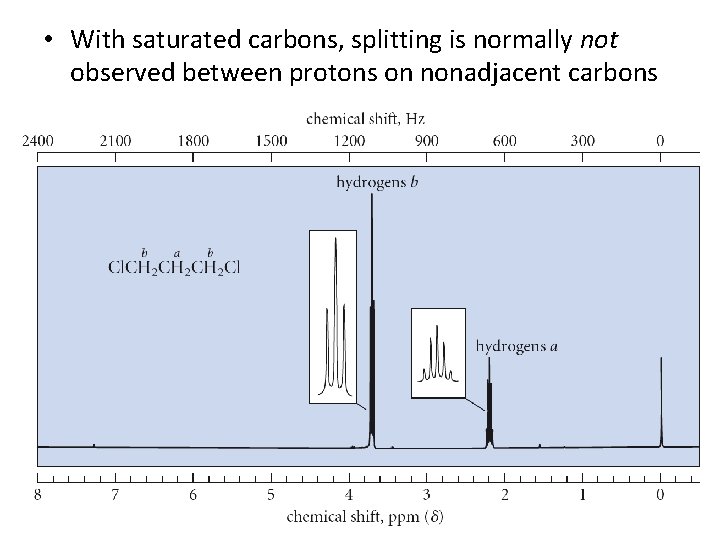
• With saturated carbons, splitting is normally not observed between protons on nonadjacent carbons 17

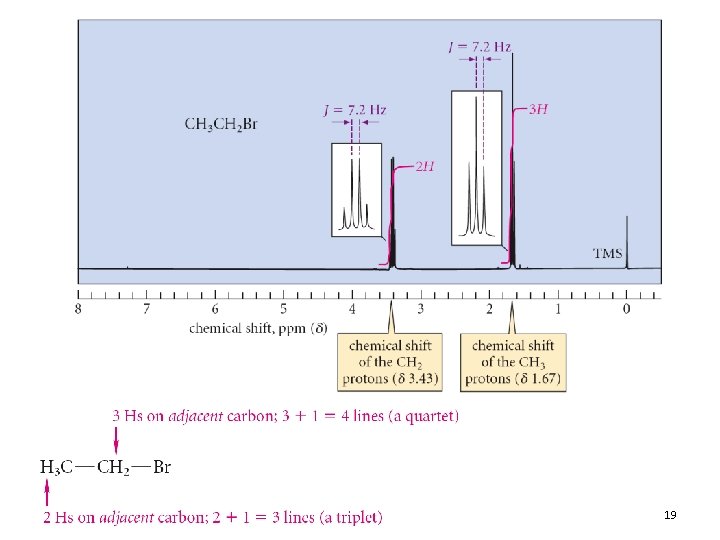
Coupling Constants • The spacing between adjacent peaks of a splitting pattern is the coupling constant (J) • The value of J is reported in Hz • Two coupled protons must have the same value of J • The coupling constant does not vary with the operating frequency of the spectrometer 13. 4 The NMR Spectrum: Spin-Spin Splitting 18
19
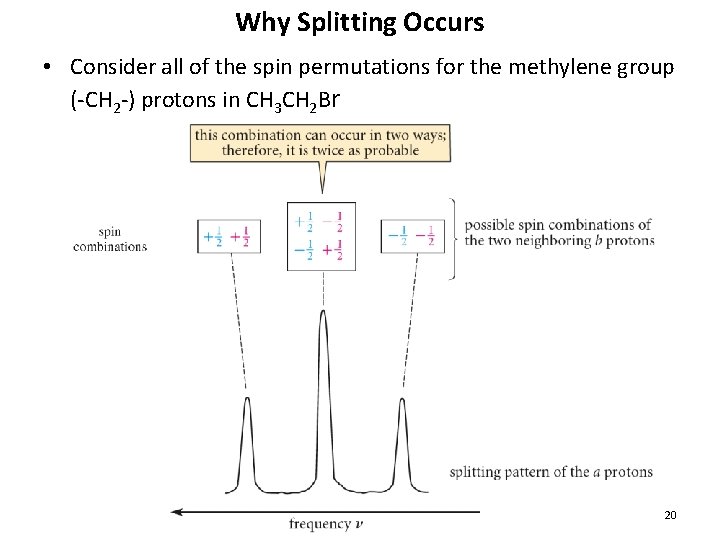
Why Splitting Occurs • Consider all of the spin permutations for the methylene group (-CH 2 -) protons in CH 3 CH 2 Br 20

NMR Information • The three elements of an NMR spectrum (chemical shift, integration, splitting) can piece a molecular structure together • The information in an NMR spectrum is oftentimes reported in text form • For ethyl bromide: 13. 4 The NMR Spectrum: Spin-Spin Splitting 21
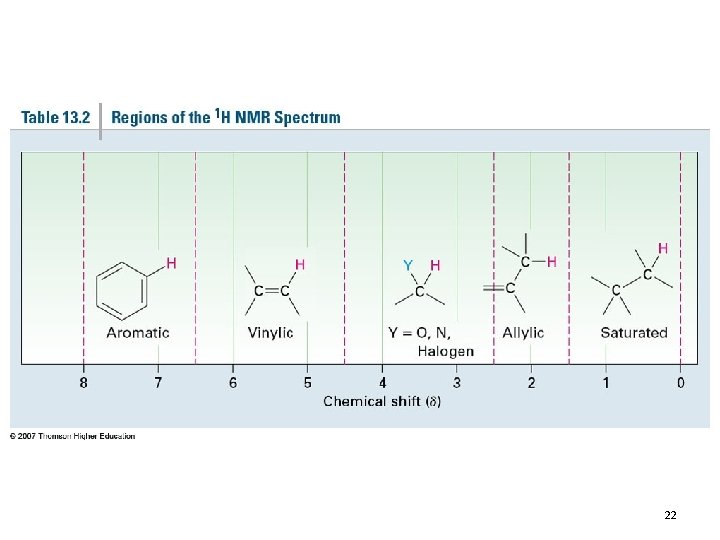
22
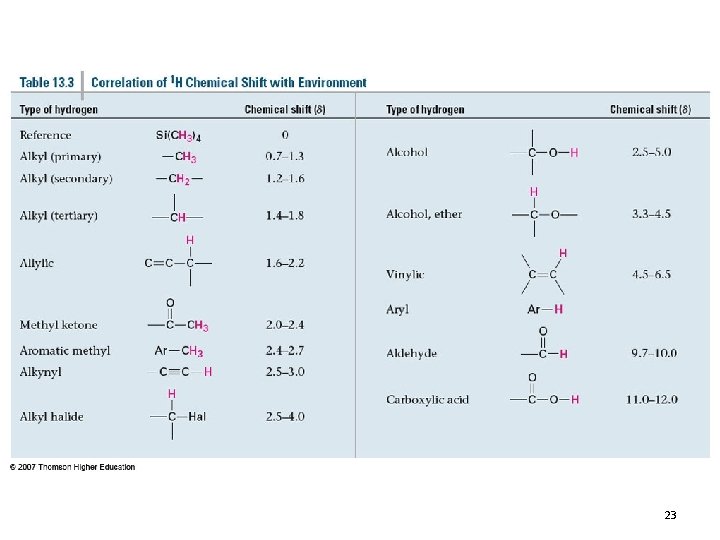
23
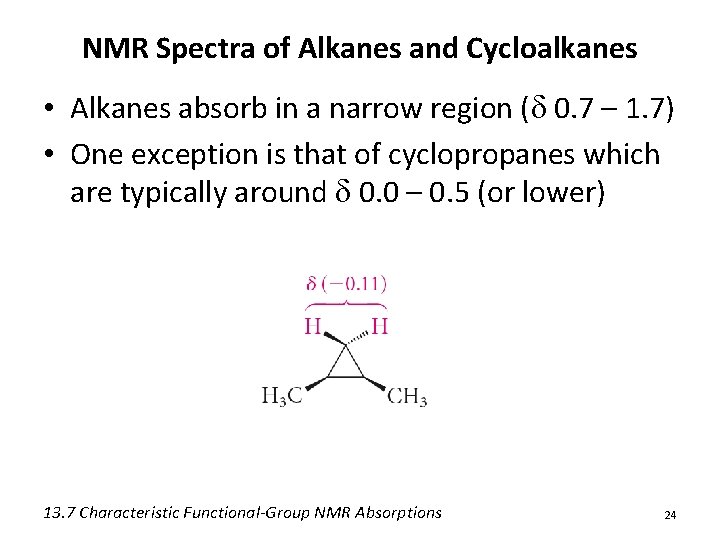
NMR Spectra of Alkanes and Cycloalkanes • Alkanes absorb in a narrow region (d 0. 7 – 1. 7) • One exception is that of cyclopropanes which are typically around d 0. 0 – 0. 5 (or lower) 13. 7 Characteristic Functional-Group NMR Absorptions 24
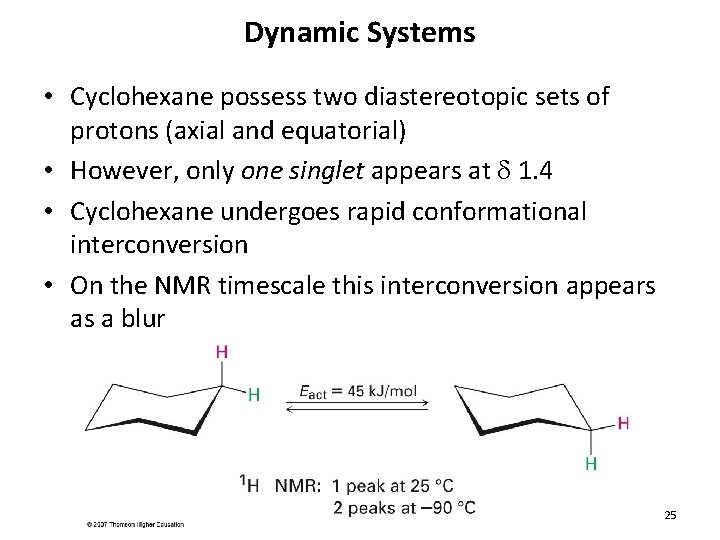
Dynamic Systems • Cyclohexane possess two diastereotopic sets of protons (axial and equatorial) • However, only one singlet appears at d 1. 4 • Cyclohexane undergoes rapid conformational interconversion • On the NMR timescale this interconversion appears as a blur 25

26
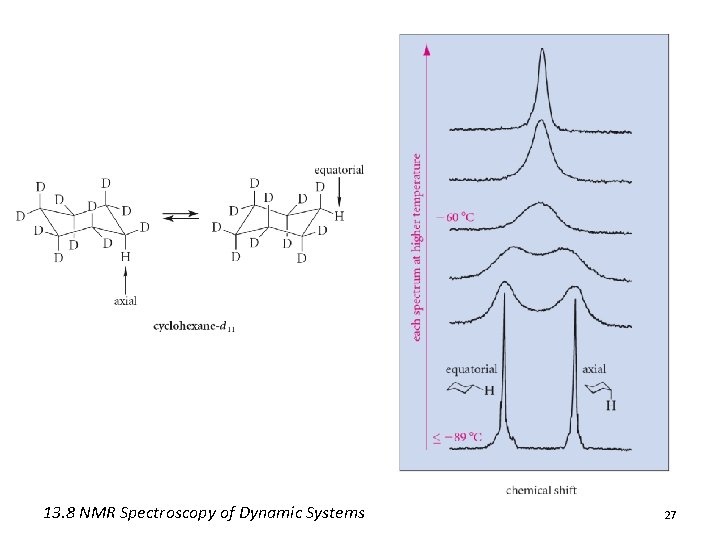
13. 8 NMR Spectroscopy of Dynamic Systems 27
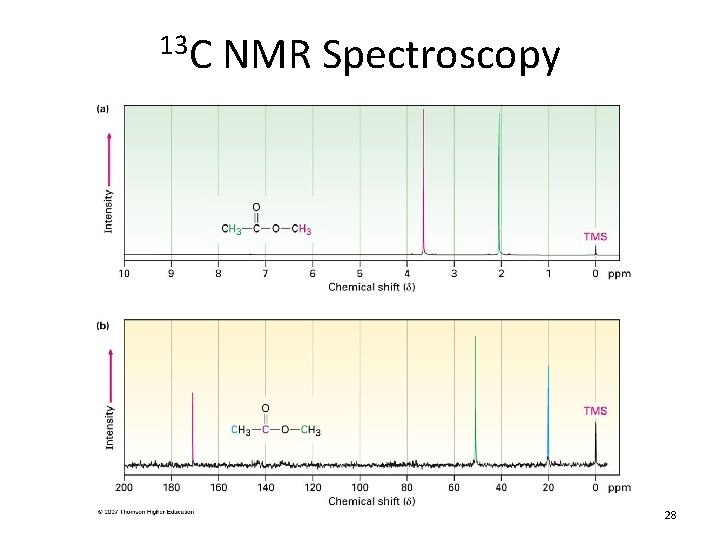
13 C NMR Spectroscopy 28
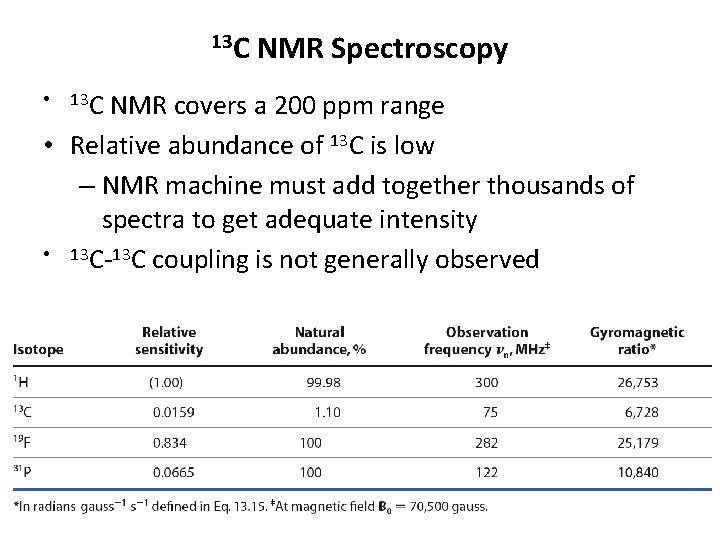
13 C NMR covers a 200 ppm range • Relative abundance of 13 C is low – NMR machine must add together thousands of spectra to get adequate intensity • 13 C-13 C coupling is not generally observed • 13 C NMR Spectroscopy
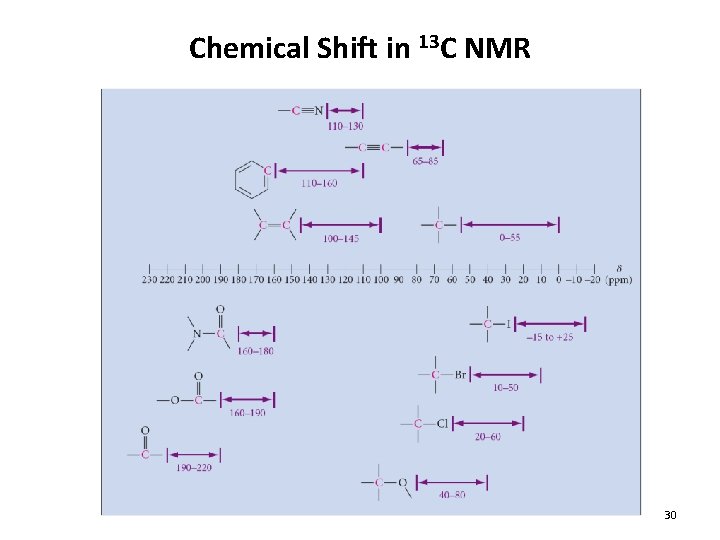
Chemical Shift in 13 C NMR 30
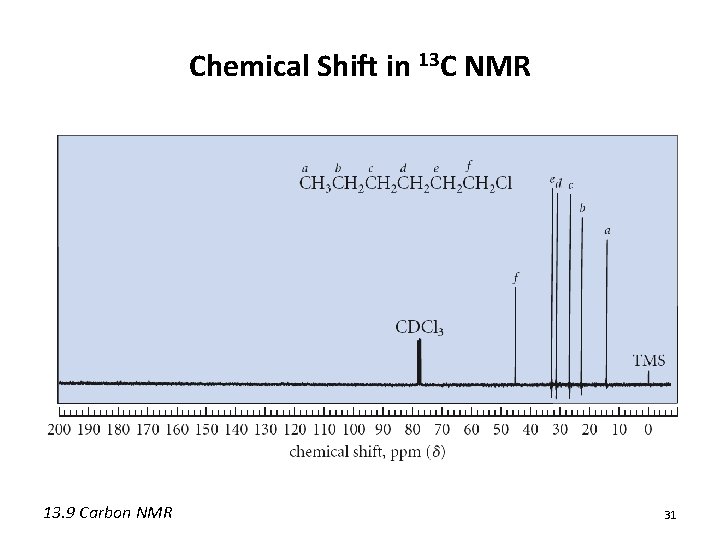
Chemical Shift in 13 C NMR 13. 9 Carbon NMR 31

Chemical Shift in 13 C NMR • The chemical shift is also influenced by the number of attached carbons • Typically d(quaternary) > d(tertiary) > d(secondary) > d(primary) 13. 9 Carbon NMR 32
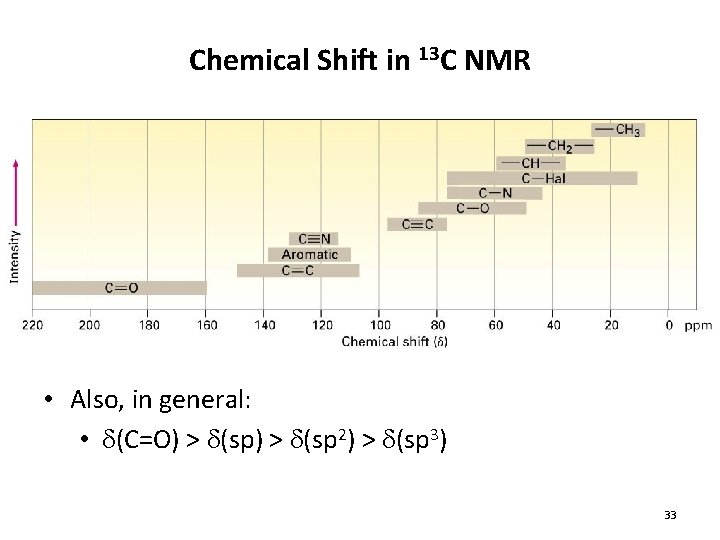
Chemical Shift in 13 C NMR • Also, in general: • d(C=O) > d(sp 2) > d(sp 3) 33
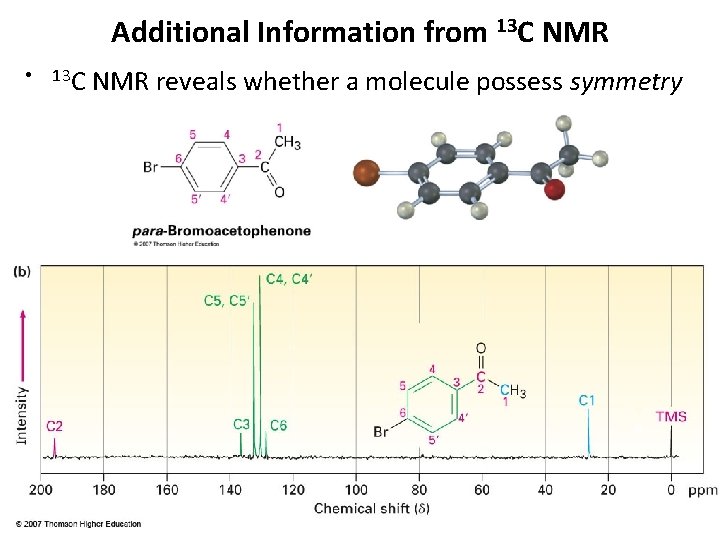
Additional Information from 13 C NMR • 13 C NMR reveals whether a molecule possess symmetry 13. 9 Carbon NMR 34
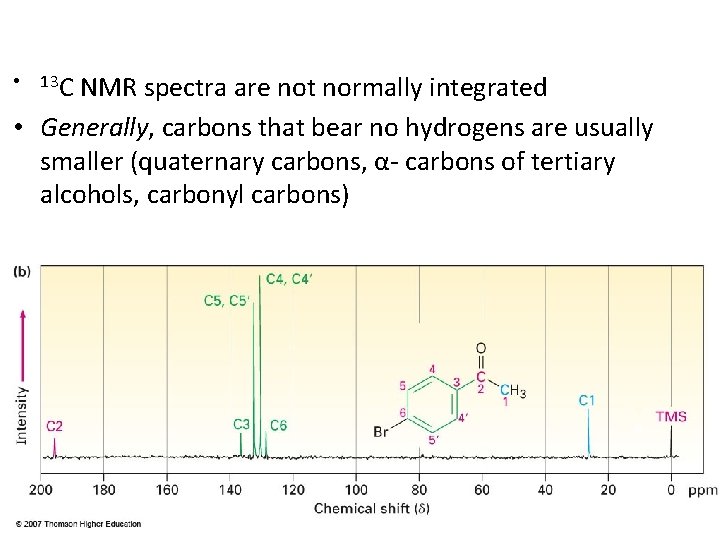
NMR spectra are not normally integrated • Generally, carbons that bear no hydrogens are usually smaller (quaternary carbons, α- carbons of tertiary alcohols, carbonyl carbons) • 13 C 35

DEPT 13 C NMR • Distortionless enhancement with polarization transfer • The DEPT technique yields separate spectra for methyl, methylene, and methine carbons • Each line in these spectra corresponds to a line in the complete 13 C NMR spectrum • Quaternary carbons do not appear in any of the DEPT spectra 13. 9 Carbon NMR 36
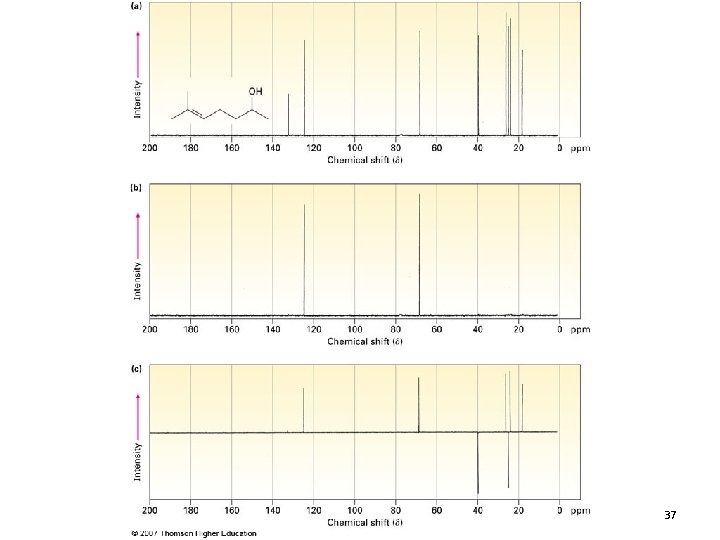
37
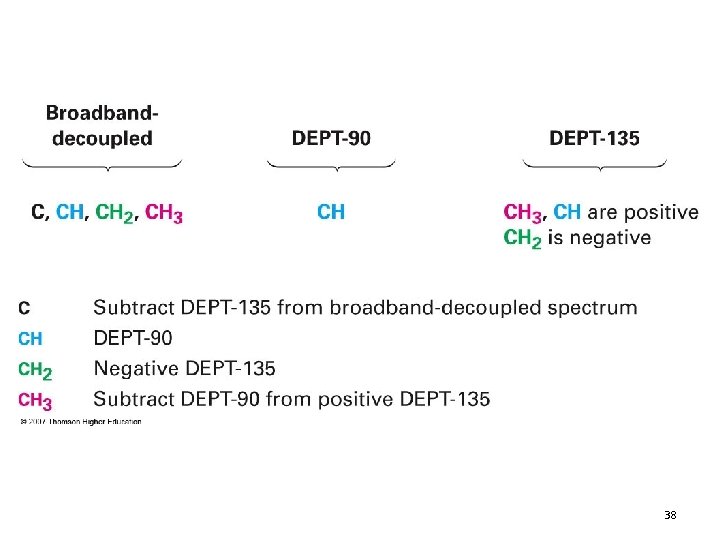
38

DEPT 13 C NMR 13. 9 Carbon NMR 39

Combined Spectroscopy • Some suggestions for solving structures 1. Find the molecular mass if possible (MS) 2. Use elemental analysis to calculate the molecular formula 3. Identify functional groups or fragments (IR, NMR) 4. Determine if there is symmetry (13 C NMR, but possibly from 1 H NMR too) 13. 10 Solving Structure Problems with Spectroscopy 40

Combined Spectroscopy 5. Write out partial structures and all possible complete structures that are consistent with the spectra and molecular formula 6. Rationalize all spectra for consistency with the proposed structure 13. 10 Solving Structure Problems with Spectroscopy 41

FT-NMR • Modern instruments employ pulse-Fouriertransform NMR (FT-NMR) • In FT-NMR all proton spins are excited instantaneously with a broad band rf pulse • The spectrum is obtained by analyzing the emission of rf energy as the spins reestablish equilibrium • This permits the collection of a large number of scans (50 – 20, 000) in a relatively short time 13. 11 The NMR Spectrometer 42

FT-NMR • A computer stores, analyzes, and mathematically sums the scans • Random electronic noise sums to zero when averaged over many scans • This dramatically enhances the signal-to-noise • This technique is particularly important to 13 C NMR considering the low natural abundance of the 13 C nucleus 13. 11 The NMR Spectrometer 43

Other Useful Forms of NMR • Solid-state NMR: Can study the properties of solid substances (coal, drugs, polymers) • Phosphorous NMR (31 P): Can be used to study biological processes (including intact cells or even whole organisms) • NMR tomography or magnetic resonance imaging (MRI): Physicians can image organs without X-rays or other harmful radiation 13. 12 Other Uses of NMR 44
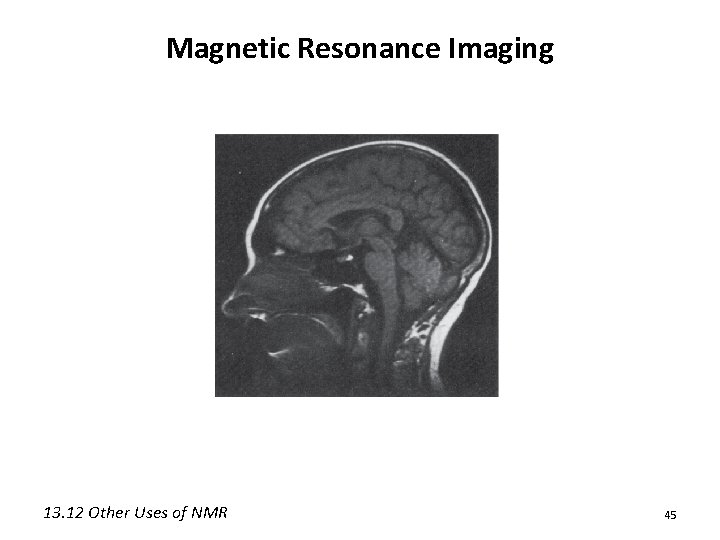
Magnetic Resonance Imaging 13. 12 Other Uses of NMR 45
- Slides: 45