The next generation for MR valve related devices









- Slides: 15

The next generation for MR valve related devices FDA-PMDA HBD Session March 4, 2019

Barathi Sethuraman, Ph. D. Employee: Abbott Disclosure

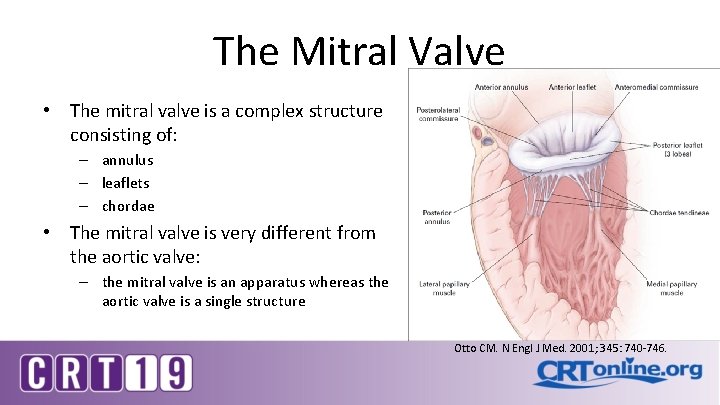
The Mitral Valve • The mitral valve is a complex structure consisting of: – annulus – leaflets – chordae • The mitral valve is very different from the aortic valve: – the mitral valve is an apparatus whereas the aortic valve is a single structure Otto CM. N Engl J Med. 2001; 345: 740 -746.

Mitral Regurgitation (MR) • Primary MR is a valvular problem (leaflet or chordae) or • Secondary MR is a ventricular problem (tethering of structurally normal leaflets or annular dilation)

Transcatheter Mitral Valve Therapies Repair • For primary MR: – Edge to edge repair – Artificial chordae • For secondary MR: – Edge to edge repair – Direct or indirect annuloplasty – LV reshaping Replacement • Transapical • Transseptal

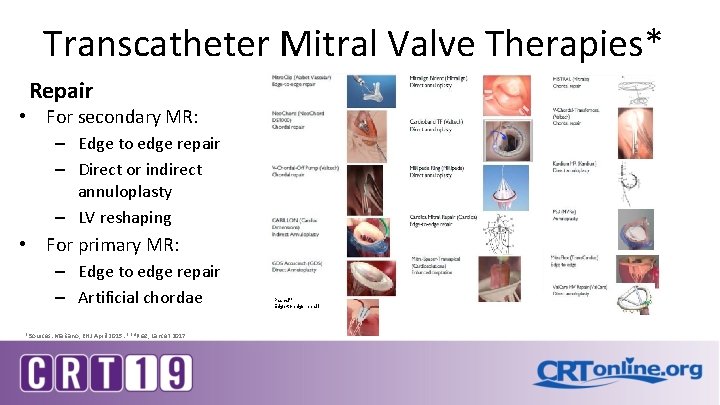
Transcatheter Mitral Valve Therapies* Repair • For secondary MR: – Edge to edge repair – Direct or indirect annuloplasty – LV reshaping • For primary MR: – Edge to edge repair – Artificial chordae *Sources: Maisano, EHJ April 2015: ** 4 Praz, Lancet 2017 Pascal** Edge-to-edge repairl

The Mitra. Clip™ System (Edge-to-Edge Repair) • Currently the only device approved by the FDA to treat prohibitive risk patients with primary MR • Access through the femoral vein • Currently available in two sizes: NTR & XTR Mitra. Clip® NTR Mitra. Clip® XTR +5 mm* Mitra. Clip® NTR The original Mitra. Clip® NT size, with an improved Clip Delivery System. Mitra. Clip® XTR Longer arms for easier grasping and better reach, with an improved Clip Delivery System.

Clinical Evidence for Mitra. Clip™ • • EVEREST II REALISM ACCESS-EU Mitra. Clip Japan TVT registry COAPT EXPAND

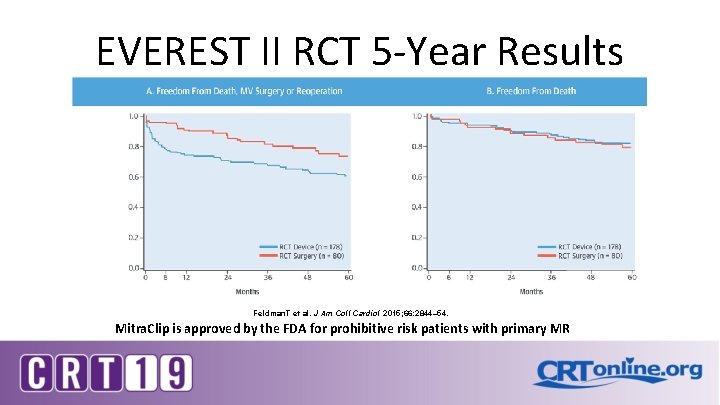
EVEREST II RCT 5 -Year Results Feldman. T et al. J Am Coll Cardiol 2015; 66: 2844– 54. Mitra. Clip is approved by the FDA for prohibitive risk patients with primary MR

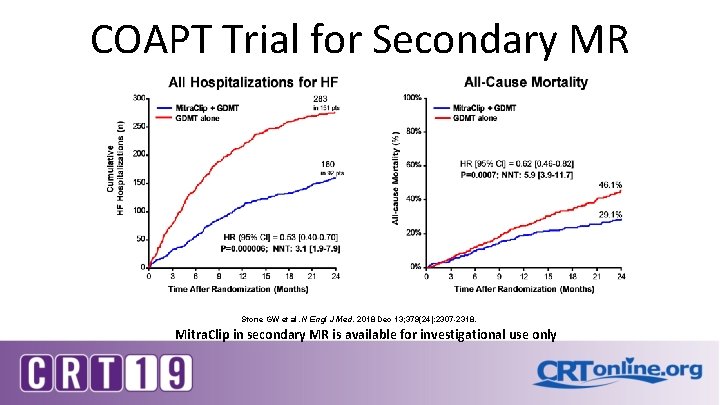
COAPT Trial for Secondary MR Stone GW et al. N Engl J Med. 2018 Dec 13; 379(24): 2307 -2318. Mitra. Clip in secondary MR is available for investigational use only

Transcatheter Mitral Valve Therapies* Replacement • Transapical • Transseptal *Source: Maisano, EHJ April 2015 Tendyne

Tendyne (Replacement) • Tri-leaflet, bioprosthetic valve • Outer frame contoured to mitral annulus – Multiple sizes – Conforms to broad range of dimensions – Isolates function of sealing and stability • Valve stabilized by tether and apical pad • Fully retrievable, repositionable intraoperatively • No requirement for CPB or rapid ventricular pacing Note: Tendyne is available for investigational use only

Clinical Evidence for Tendyne • Over 200 subjects studied through ongoing global clinical trials • Longest follow-up: 3 years Tendyne is available for investigational use only

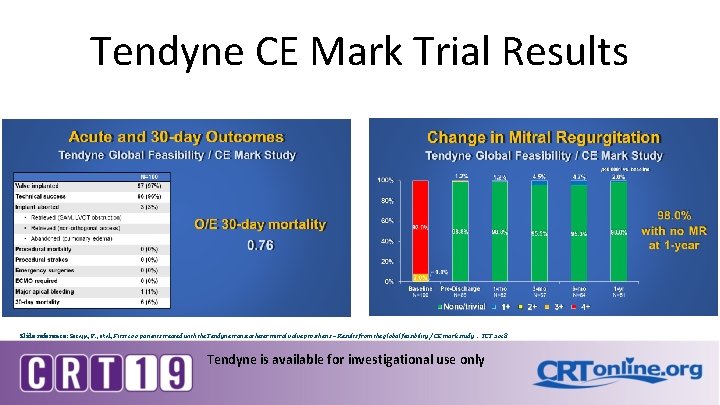
Tendyne CE Mark Trial Results Slide reference: Sorajja, P. , et al, First 100 patients treated with the Tendyne transcatheter mitral valve prosthesis – Results from the global feasibility / CE mark study. TCT 2018 Tendyne is available for investigational use only

What’s Next in MR Therapies? Improvements in deliverability and safety Improvement in MR reduction Minimize invasiveness Lower profile Tailored treatment based on individual anatomies, combination therapies (toolbox) • Evolution of imaging and related operator’s skills • Clinical studies to determine which patients are appropriate for repair vs. replacement • • •