The NCI Central IRB Initiative Jacquelyn L Goldberg

The NCI Central IRB Initiative Jacquelyn L. Goldberg, J. D. VA IRB Chair Training April 8, 2004

The NCI CIRB Initiative Purpose: To establish a Central IRB for NCI Phase 3 multi-center trials – To eliminate the significant administrative burdens on local investigators and IRBs while maintaining a high level of human subjects protection – To enhance the protection of research participants by providing consistent expert IRB review at the national level before the protocol is distributed to local investigators

Background l l l Armitage Report NCI holds an FWA NCI Director appoints diverse Board Meeting monthly since January 2001 Menu includes all Phase 3 Adult Cooperative Group protocols (20 -30 new per year), some Phase 2, plans for peds Daily administrative operations managed by contractor
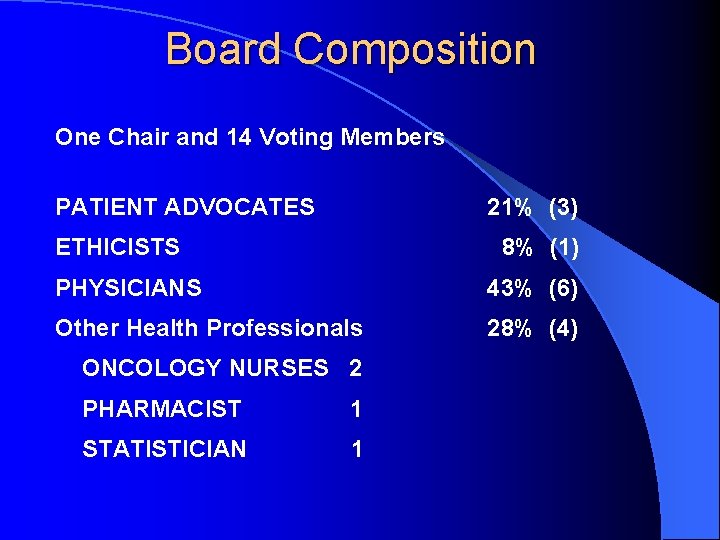
Board Composition One Chair and 14 Voting Members PATIENT ADVOCATES 21% (3) ETHICISTS 8% (1) PHYSICIANS 43% (6) Other Health Professionals 28% (4) ONCOLOGY NURSES 2 PHARMACIST 1 STATISTICIAN 1

Selecting a CIRB Model l OHRP (OPRR) allows for different centralized IRB models l See Guidance of August 27, 1998 (updated July 21, 2000) entitled “Knowledge of Local Research Context” http: //ohrp. osophs. dhhs. gov/humansubjects/guid ance/local. htm

l Model A– Central IRB review only – Appropriate where no local IRB exists – Understanding of local context obtained via site visits, audits, teleconferences – Large expenditure in time and money to understand local context

l Model B– Central IRB review with some local IRB review – More appropriate where local IRB already present – Value of this model: Can utilize LIRB for understanding of local context and forego expense of site visits, etc.

l NCI chose Model B for practical reasons – Unlike many other CIRBs, the NCI CIRB does not exist in lieu of a local IRB – Local IRBs already exist and NCI must interface with them – Who better to understand local research context than local IRB chair/members?

Division of Responsibilities l CIRB and LIRB share regulatory responsibilities l The CIRB’s primary function is initial and continuing review of protocols l The local institution’s primary function is consideration of local context and oversight of local performance

How does the NCI CIRB model work? l CIRB approves protocol l Local investigator is notified of protocol via – Routine Group activation announcement – CIRB e-mail

l If the local investigator decides to open protocol, – OPTION 1: Investigator or CRA downloads the completed IRB application, protocol and consent from the CIRB website and submits documents to local IRB; LIRB downloads remaining documents – OPTION 2: PI/CRA downloads all the documents and submits them to the LIRB

Local IRB chair or a subcommittee reviews for local concerns and decides whether to approve (“facilitated review”) l If LIRB accepts, they notify CIRB. l The CIRB becomes the IRB of record. It handles amendments, continuing reviews, adverse events etc. l If LIRB does not accept, LIRB can decide to review the protocol themselves as per their own local procedures. l

Adverse Event Reporting l Local site/PI does not report AEs to CIRB (local PI must still report local event to LIRB) l CIRB accesses NCI’s adverse event reporting system – CIRB notified of AEs reported to FDA l CIRB decisions posted on Web for local IRB use

Informed Consent Local boilerplate additions to the informed consent dealing with state and local law, institutional requirements, or IRB policies l Minor word changes which do not alter the meaning of the CIRB approved contents l Revisions/changes to the local consent form other than those described above require local full board review and facilitated review may not be used. l

Current Status of Initiative Number of Participating LIRBS l Target of 100 LIRBs l 159 LIRBs have joined l Represents 254 research sites (potential of 1500 -2000)

Use of Facilitated Review l 101 of the 159 LIRBs have used facilitated review at least once l Estimated lag time of six months between joining and first facilitated review – – High turnover of IRB coordinators Attention elsewhere (HIPAA) Not yet developed internal SOPs IRB culture

l NCI goal for 2003 to show use of facilitated review in 70 -80 LIRBs l NCI goal for 2004 is expansion to 500 sites

Evaluation Plan Measure local utilization of facilitated review l Quantify CIRB effect on local site time frames l Assess the CIRB experience: l – Local IRB Chair – LIRB Coordinator – Site Principal Investigator for protocol – CIRB members Evaluate the quality of CIRB reviews l Demonstrate CIRB compliance with federal regulations l

Benefits to Investigators l No advance preparation for IRB review at the local site – no IRB application to complete, just download the IRB packet from the CIRB website l No waiting for the next meeting of the full board – investigators can enroll patients in trials much faster potentially as quickly as 24 -48 hours – greater menu of clinical trials available to patients – improves patient access to NCI sponsored trials l No need to submit continuing reviews, amendments or AE reports (except for AEs at local site) to the LIRB or the CIRB - CIRB becomes IRB of record

6 Easy Steps l Modify institution’s FWA to include the CIRB l Sign authorization agreement l Create a local IRB SOP for utilizing the CIRB – Determine the composition of the LIRB subcommittee that will conduct facilitated review l Notify local investigators of the new process l Copy the CIRB office on the notification (which will show that the process is complete) l Provide CIRB office with LIRB and PI site contact information

www. ncicirb. org
- Slides: 21