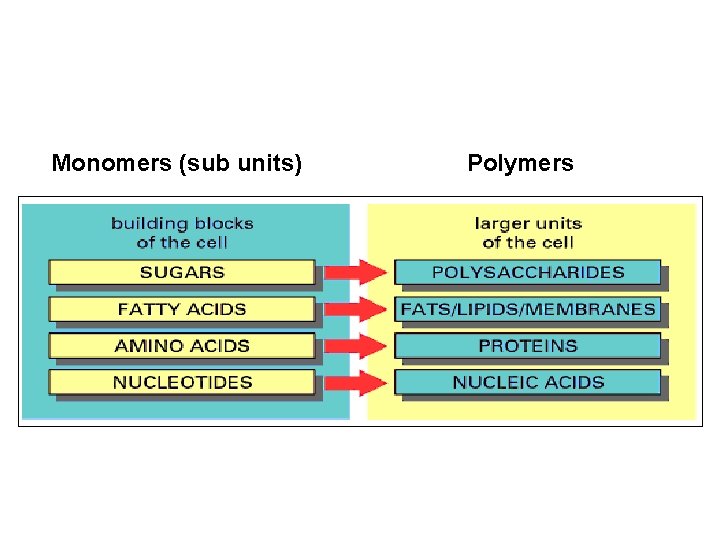
The most important biological compounds are polymers Polymers

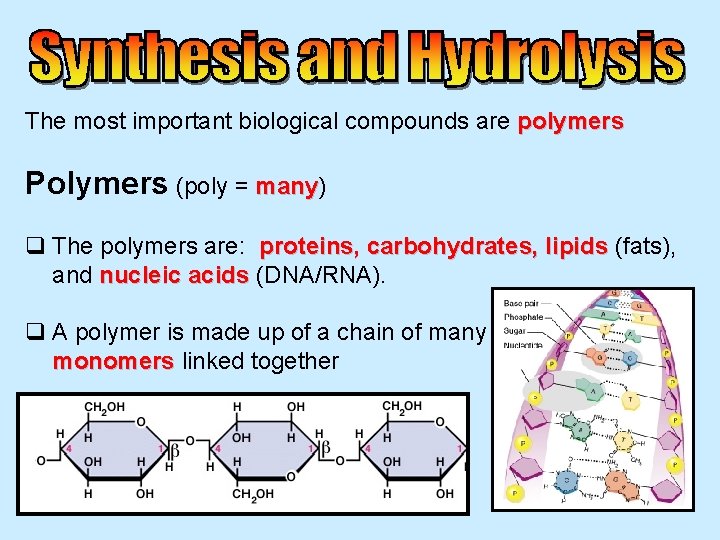
The most important biological compounds are polymers Polymers (poly = many) many q The polymers are: proteins, carbohydrates, lipids (fats), and nucleic acids (DNA/RNA). q A polymer is made up of a chain of many monomers linked together
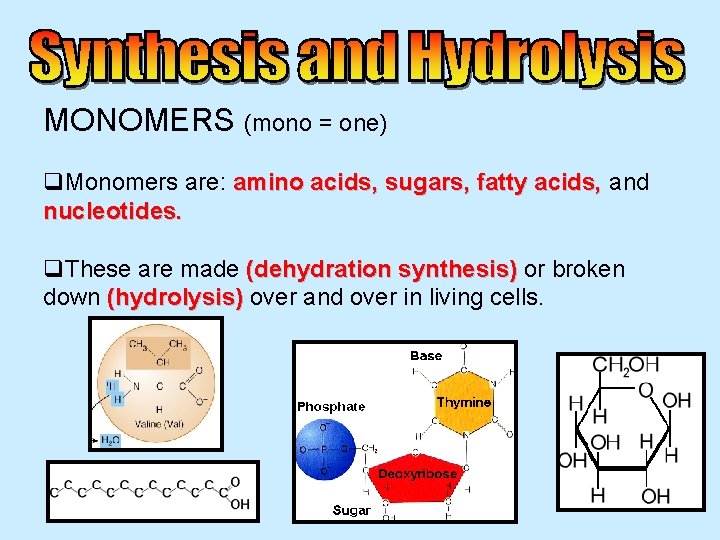
MONOMERS (mono = one) q. Monomers are: amino acids, sugars, fatty acids, and nucleotides. q. These are made (dehydration synthesis) or broken down (hydrolysis) over and over in living cells.
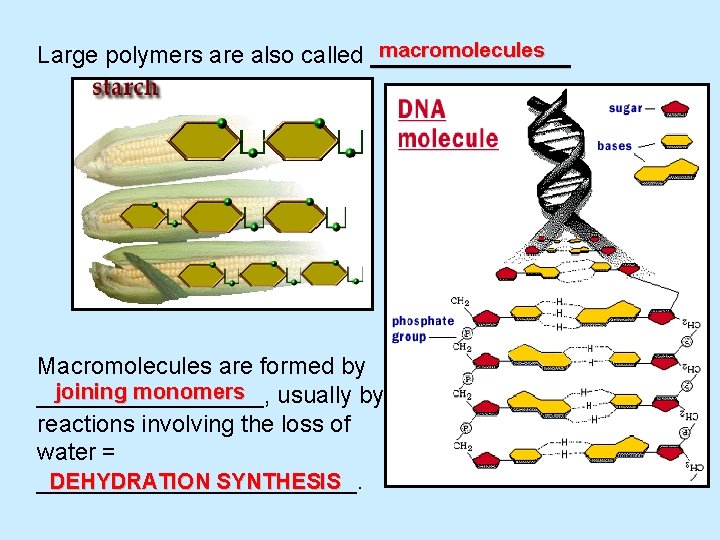
macromolecules Large polymers are also called ________ Macromolecules are formed by joining monomers usually by _________, reactions involving the loss of water = DEHYDRATION SYNTHESIS ____________.
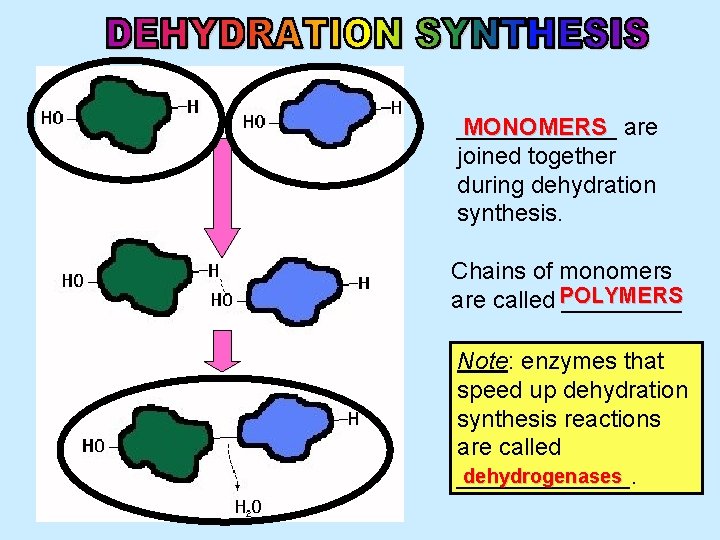
______ MONOMERS are joined together during dehydration synthesis. Chains of monomers are called POLYMERS _____ Note: enzymes that speed up dehydration synthesis reactions are called dehydrogenases _______.
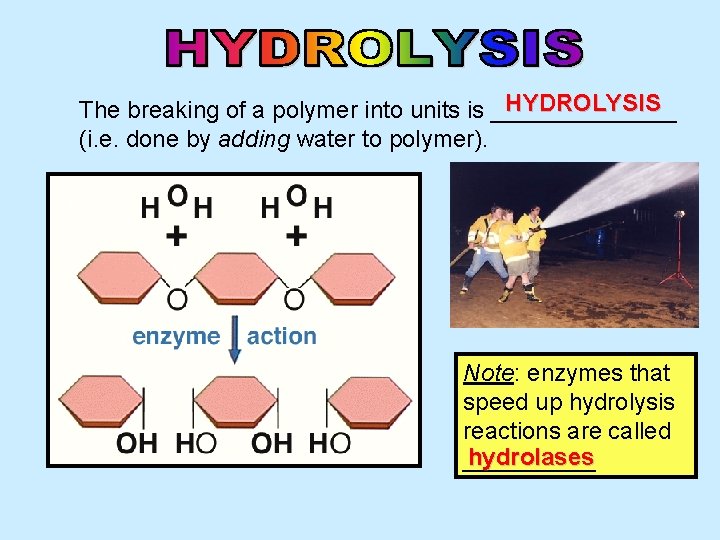
HYDROLYSIS The breaking of a polymer into units is _______ (i. e. done by adding water to polymer). Note: enzymes that speed up hydrolysis reactions are called hydrolases _____

http: //science. nhmccd. edu/biol/dehydrat. html
Monomers (sub units) Polymers

Polymers a) b) c) d)

Polymers a) Carbohydrates b) c) d)
Polymers a) Carbohydrates b) c) d) Hydrolysis
Polymers a) Carbohydrates b) c) d) Hydrolysis H 2 O & Energy
Polymers a) Carbohydrates b) c) d) Hydrolysis Monomers a) b) c) d) H 2 O & Energy
Polymers a) Carbohydrates b) c) d) Hydrolysis Monomers a) Simple sugars b) c) d) H 2 O & Energy
Polymers a) Carbohydrates b) c) d) Hydrolysis Monomers a) Simple sugars b) c) d) H 2 O & Energy
Polymers a) Carbohydrates b) c) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) c) d) H 2 O & Energy
H 2 O & Energy Polymers a) Carbohydrates b) c) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) c) d) H 2 O & Energy
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) c) d) H 2 O & Energy
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) Amino Acids c) d) H 2 O & Energy
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) Amino Acids c) d) H 2 O & Energy
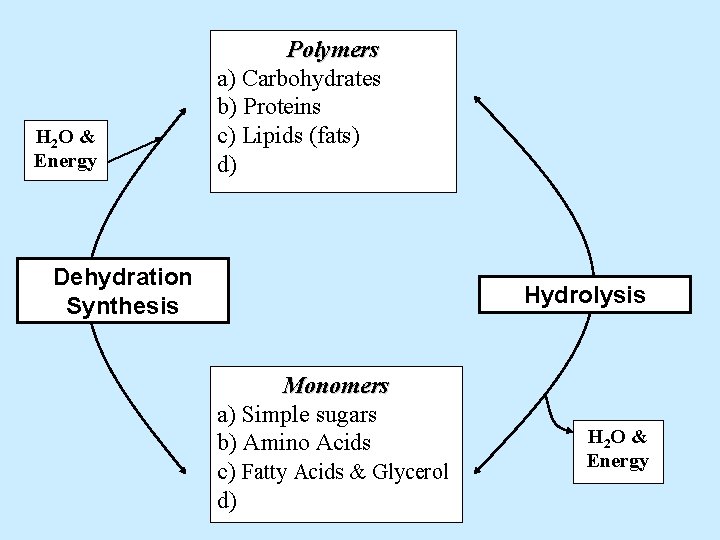
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) H 2 O & Energy
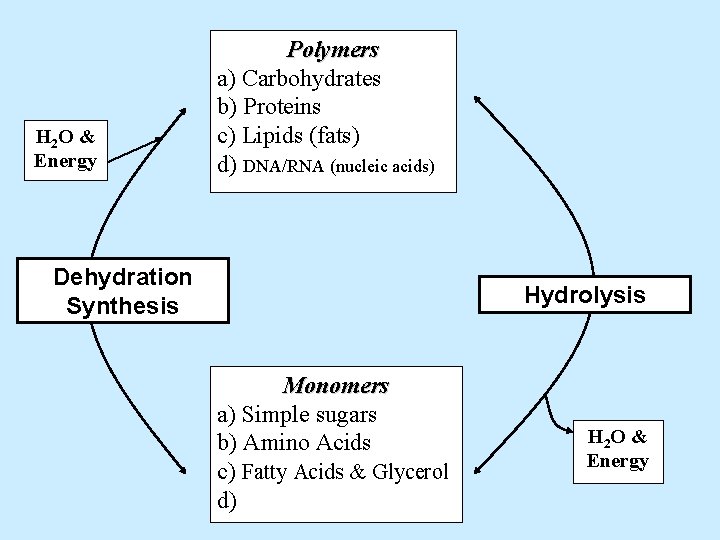
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) H 2 O & Energy
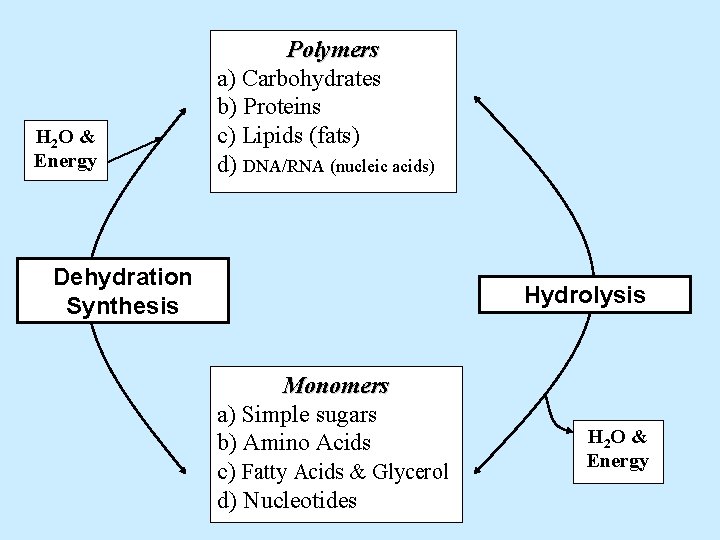
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) Dehydration Synthesis Hydrolysis Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) Nucleotides H 2 O & Energy
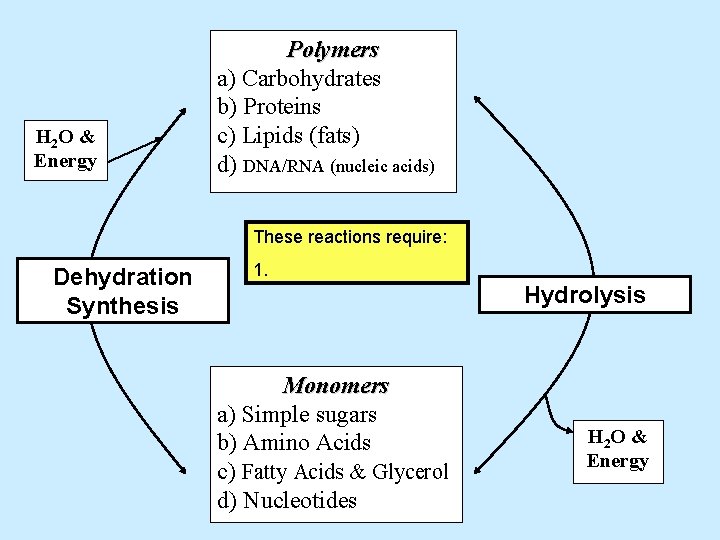
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) These reactions require: Dehydration Synthesis 1. Hydrolysis Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) Nucleotides H 2 O & Energy
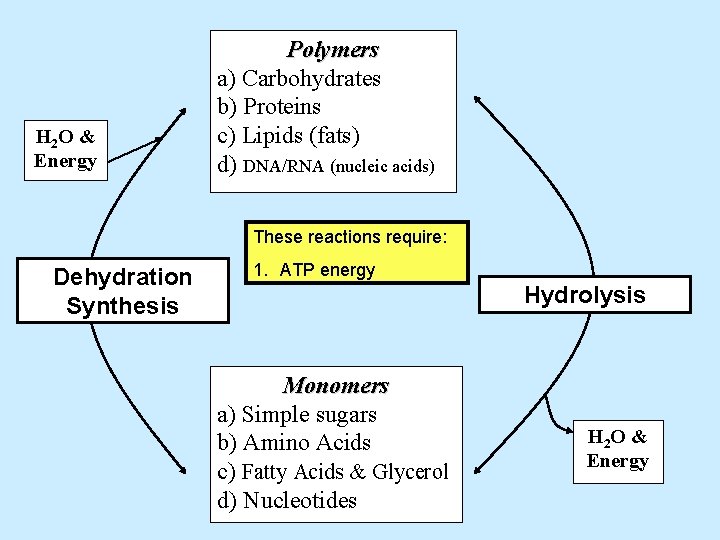
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) These reactions require: Dehydration Synthesis 1. ATP energy Hydrolysis Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) Nucleotides H 2 O & Energy
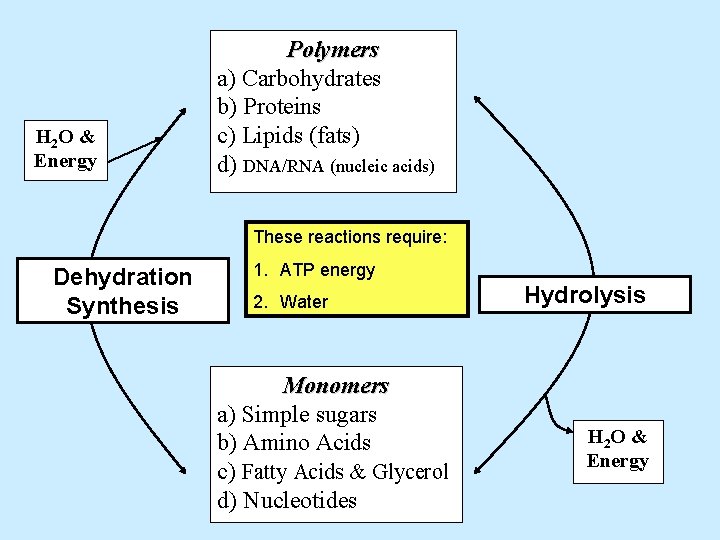
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) These reactions require: Dehydration Synthesis 1. ATP energy 2. Water Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) Nucleotides Hydrolysis H 2 O & Energy
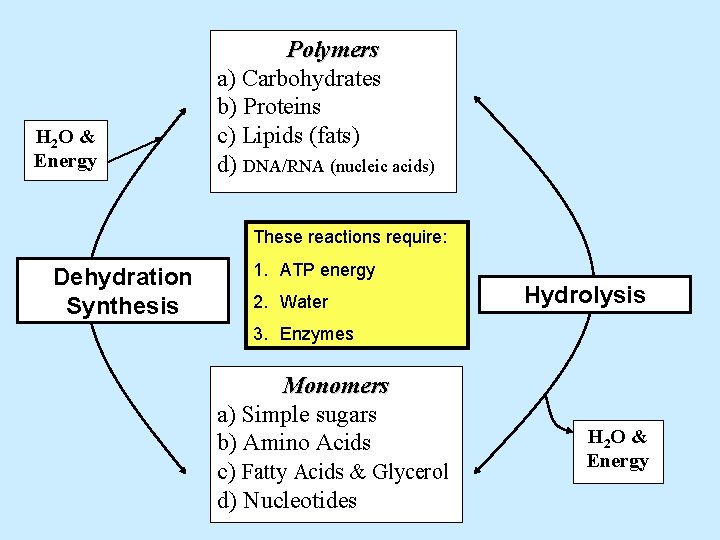
H 2 O & Energy Polymers a) Carbohydrates b) Proteins c) Lipids (fats) d) DNA/RNA (nucleic acids) These reactions require: Dehydration Synthesis 1. ATP energy 2. Water Hydrolysis 3. Enzymes Monomers a) Simple sugars b) Amino Acids c) Fatty Acids & Glycerol d) Nucleotides H 2 O & Energy

Where does the name come from? Hydrated Carbons: (CH 20)n Carbohydrates have the empirical formula of (CH 20)n where n = the # of times the chain is repeated. The carbons, hydrogens and oxygens are found in the ratio of 1: 2: 1 and are made up of a repeating chain of sugars. (CH 20)3 = C 3 H 603 (CH 20)6 = C 6 H 1206 Sugars are also known as saccarides Carbohydrates usually end in ‘ose’. ose Can you think of any examples?
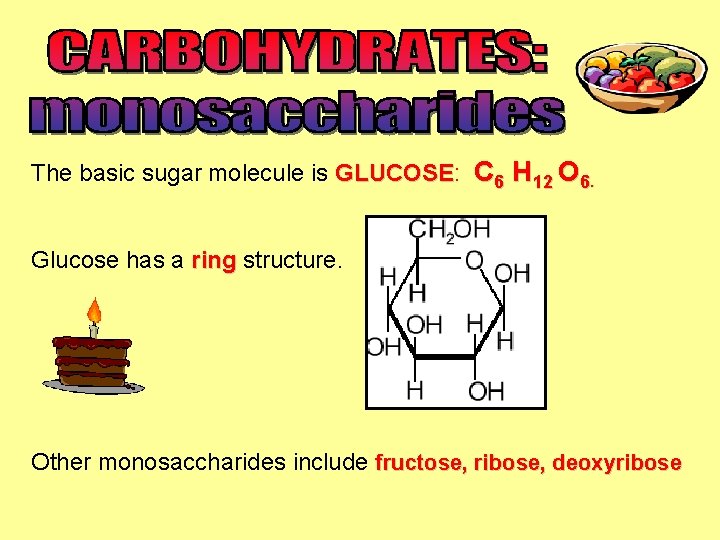
The basic sugar molecule is GLUCOSE: GLUCOSE C 6 H 12 O 6. Glucose has a ring structure. Other monosaccharides include fructose, ribose, deoxyribose
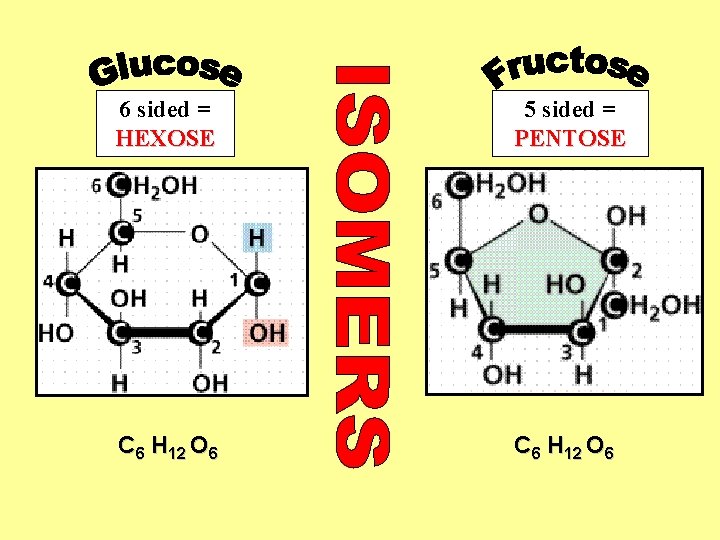
6 sided = HEXOSE 5 sided = PENTOSE C 6 H 12 O 6
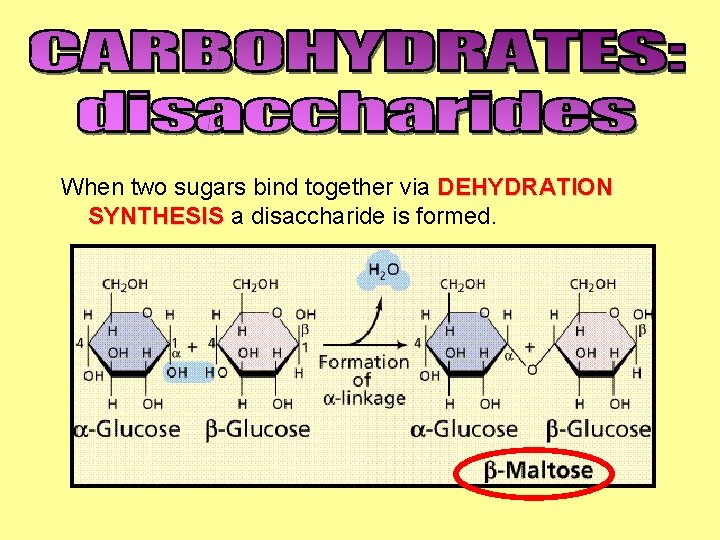
When two sugars bind together via DEHYDRATION SYNTHESIS a disaccharide is formed.
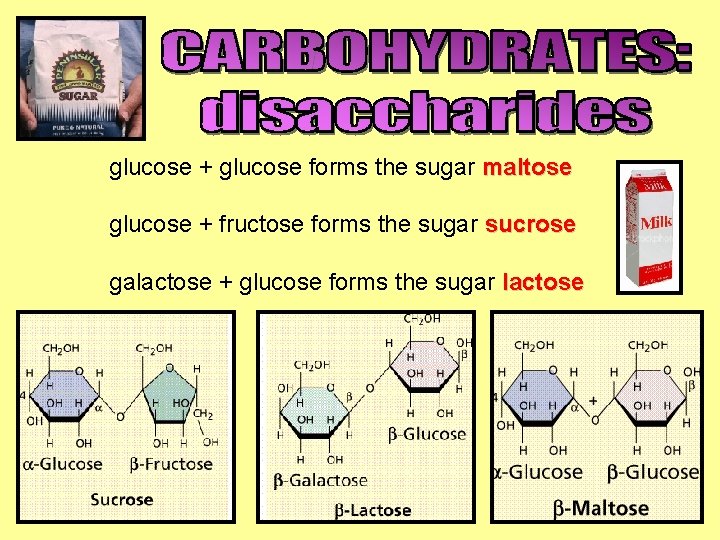
glucose + glucose forms the sugar maltose glucose + fructose forms the sugar sucrose galactose + glucose forms the sugar lactose
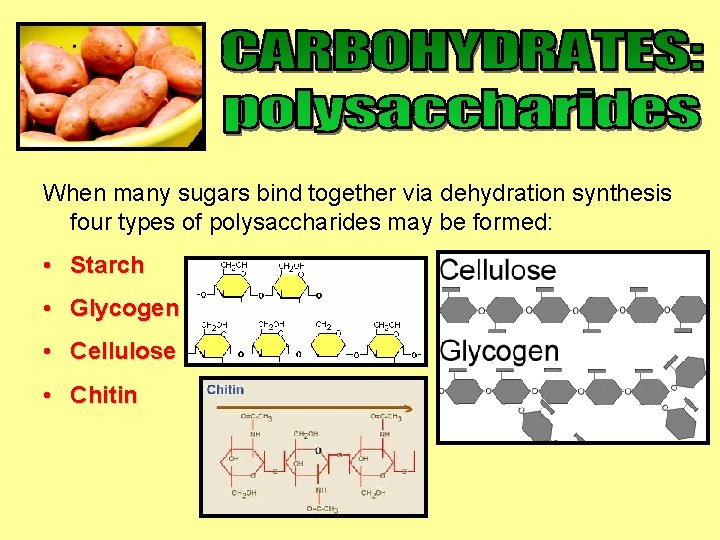
When many sugars bind together via dehydration synthesis four types of polysaccharides may be formed: • Starch • Glycogen • Cellulose • Chitin

• The cell walls of plants are made of cellulose • They are long chains of glucose molecules with no side chains • The linkage between the Carbon atoms of the sugars is different than starch and glycogen • No mammal can break this bond • 5. This is why we cannot digest cellulose = FIBRE

• Plants store their energy as starch • Starch is made up of many glucose molecules linked together • Starch has few side chains

• Animals store their energy (extra glucose) as glycogen • We store glycogen in our liver and muscles • Glycogen is made up of many glucose molecules linked together • Glycogen has many side chains
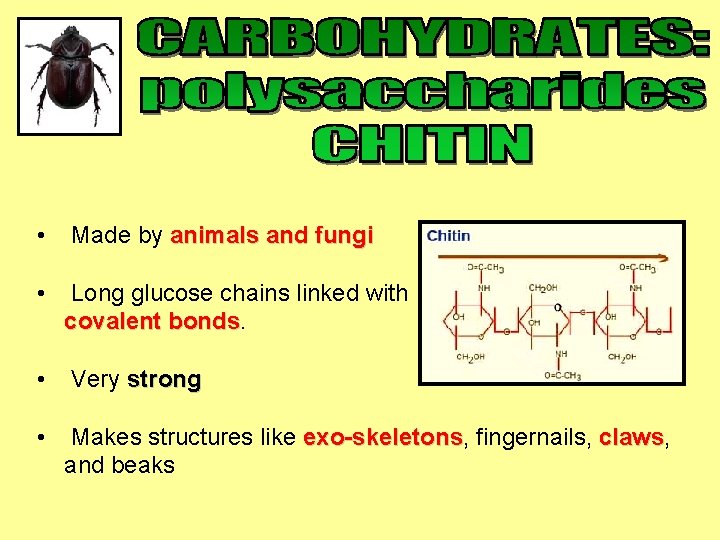
• Made by animals and fungi • Long glucose chains linked with covalent bonds • Very strong • Makes structures like exo-skeletons, exo-skeletons fingernails, claws and beaks

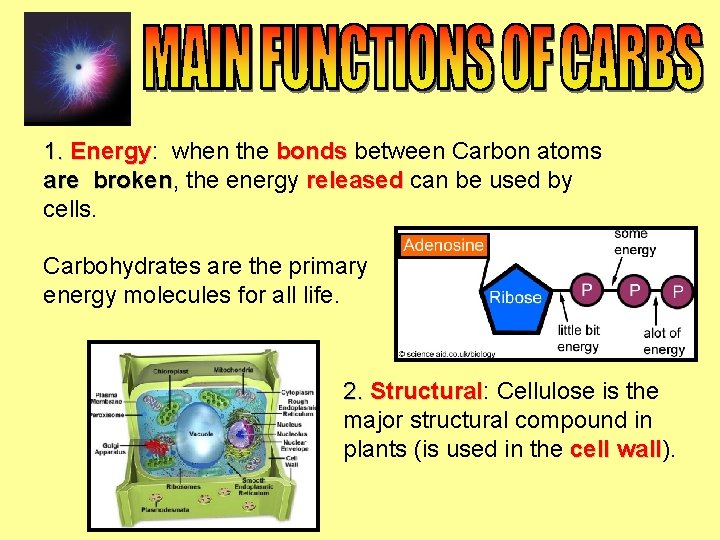
1. Energy: Energy when the bonds between Carbon atoms are broken, broken the energy released can be used by cells. Carbohydrates are the primary energy molecules for all life. 2. Structural: Structural Cellulose is the major structural compound in plants (is used in the cell wall). wall
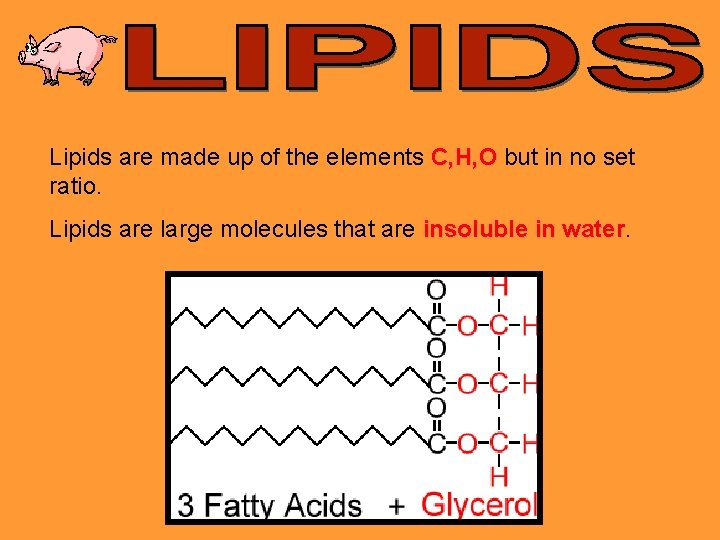
Lipids are made up of the elements C, H, O but in no set ratio. Lipids are large molecules that are insoluble in water

Synthesis of a FAT animation: http: //www 2. nl. edu/jste/lipids. htm
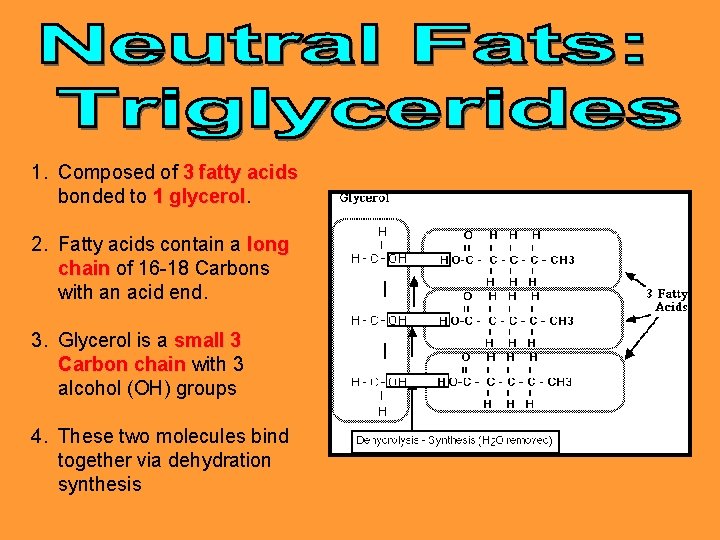
1. Composed of 3 fatty acids bonded to 1 glycerol 2. Fatty acids contain a long chain of 16 -18 Carbons with an acid end. 3. Glycerol is a small 3 Carbon chain with 3 alcohol (OH) groups 4. These two molecules bind together via dehydration synthesis

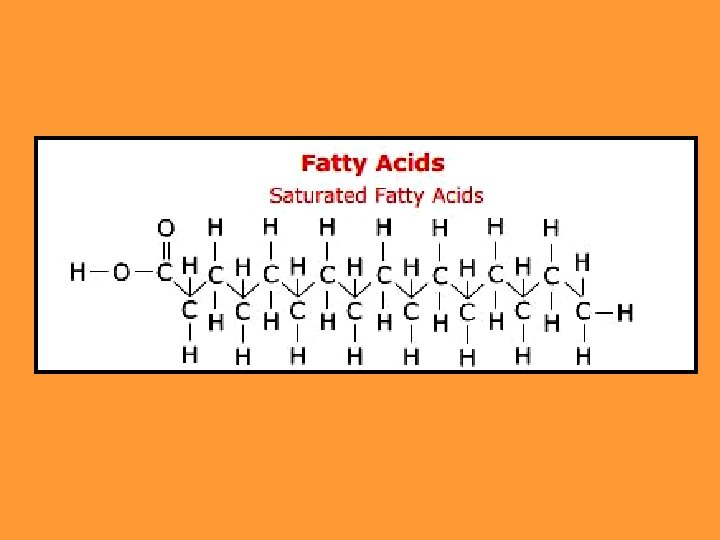
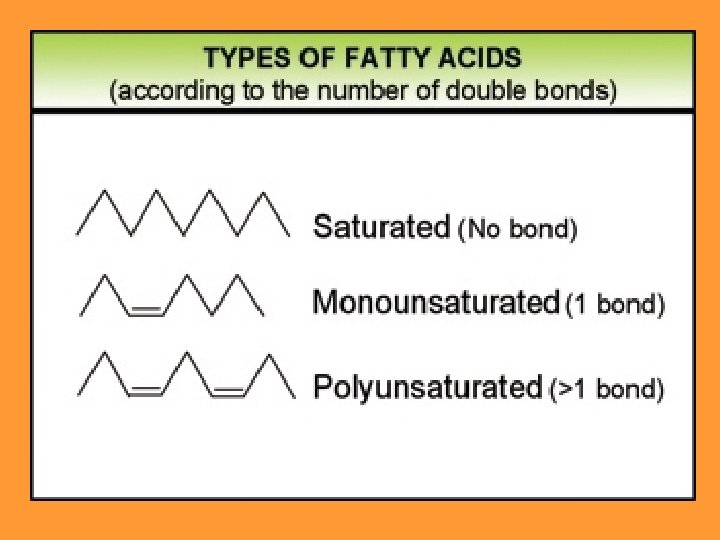
1. Saturated fats: There are no double bonds in the carbon chains of the fatty acids. The carbons are filled with hydrogens Unhealthy They mostly come from animals Become solid at room temperature. Examples: lard, butter animal fats…

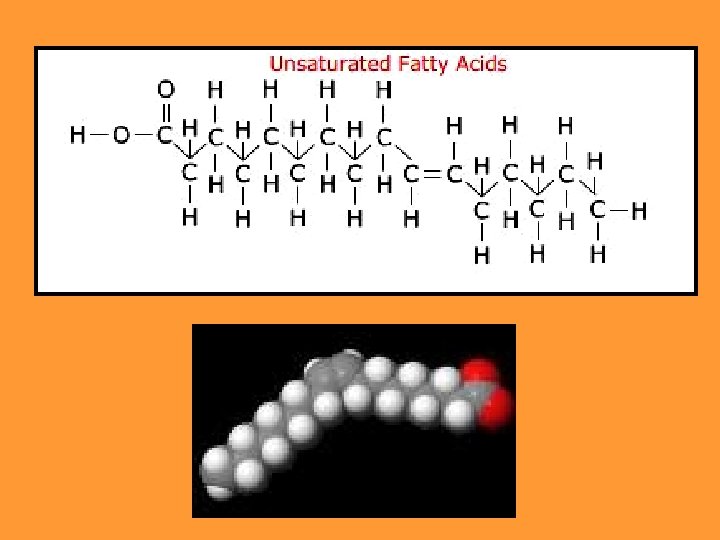
2. Unsaturated fats: There are one (monounsaturated) monounsaturated or more double bonds (polyunsaturated). polyunsaturated Mostly come from plants They are liquid at room temperature. Healthy Examples: olive oil, oil corn oil, palm oil…
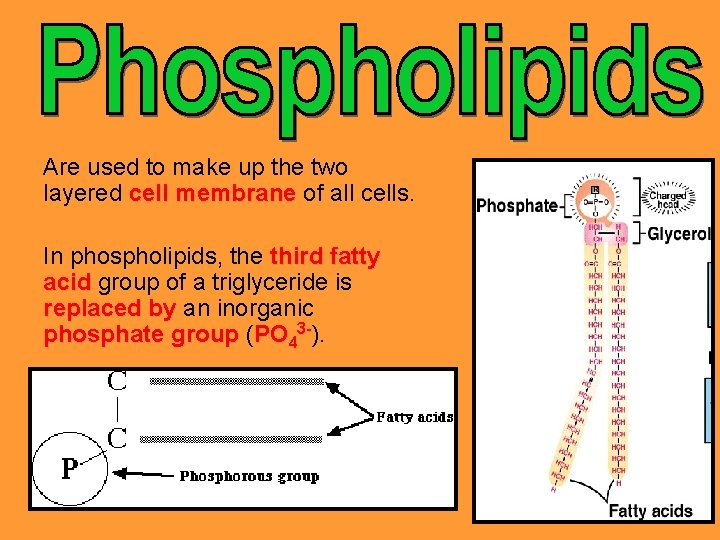
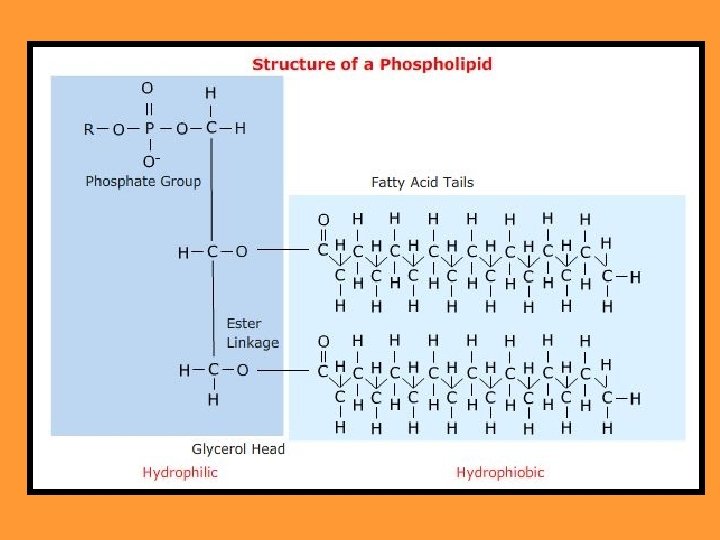
Are used to make up the two layered cell membrane of all cells. In phospholipids, the third fatty acid group of a triglyceride is replaced by an inorganic phosphate group (PO 43 -).
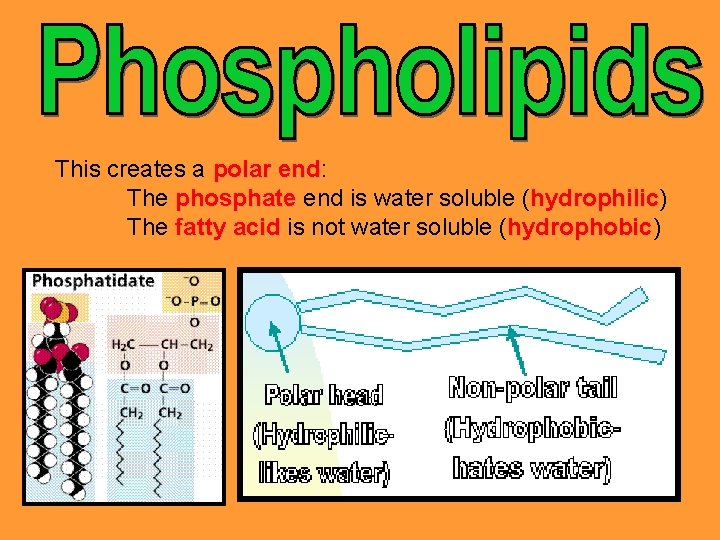
This creates a polar end: end The phosphate end is water soluble (hydrophilic) hydrophilic The fatty acid is not water soluble (hydrophobic) hydrophobic
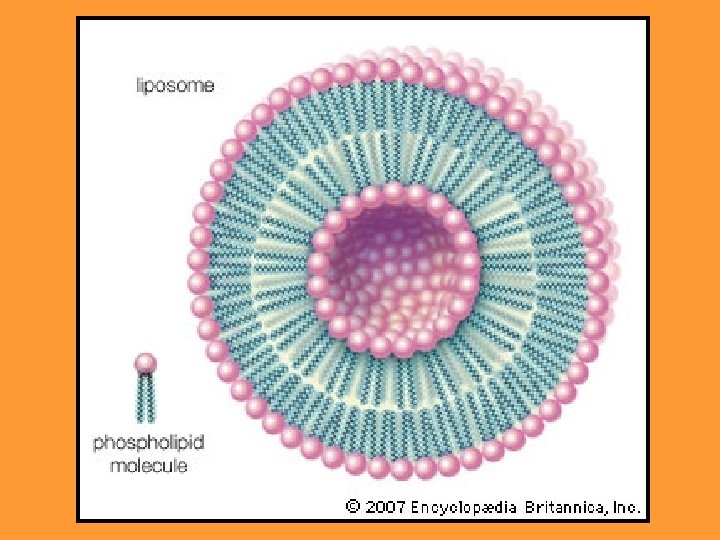
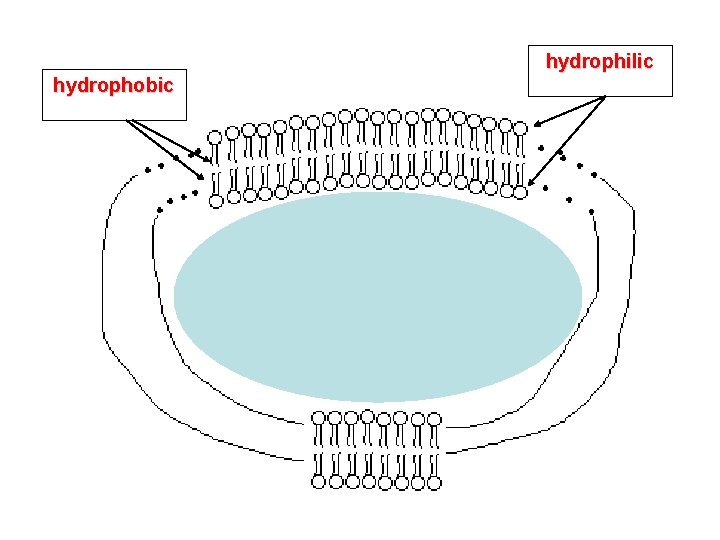
hydrophilic hydrophobic

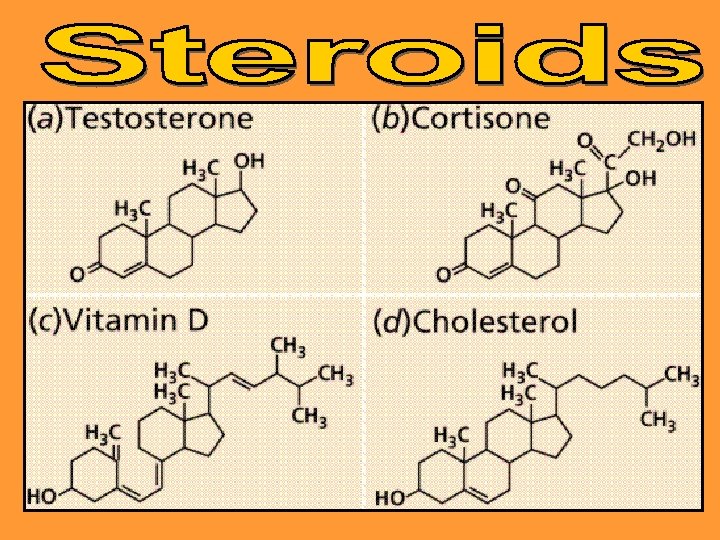
Steroids structurally look very different from lipids, but are also water insoluble They are made up of 4 Carbon ring molecules fused together. Examples: testosterone, testosterone estrogen, cholesterol and vitamin D. Used as sex hormones
1. Long term storage for energy (more efficient spacewise than glycogen or starch). 2. Insulation and protection in animals 3. Making some hormones (steroids) 4. Structure of cell membranes Without lipids, we would have no cells.

• Found in fish and leafy vegetables • Other foods are now offering omega-3’s (eggs, cereals, margarine…) • Help to reduce cancer • Helps with vision • Helps us think better

Scientific evidence has shown that dietary saturated and trans fats can increase your risk of developing heart disease.

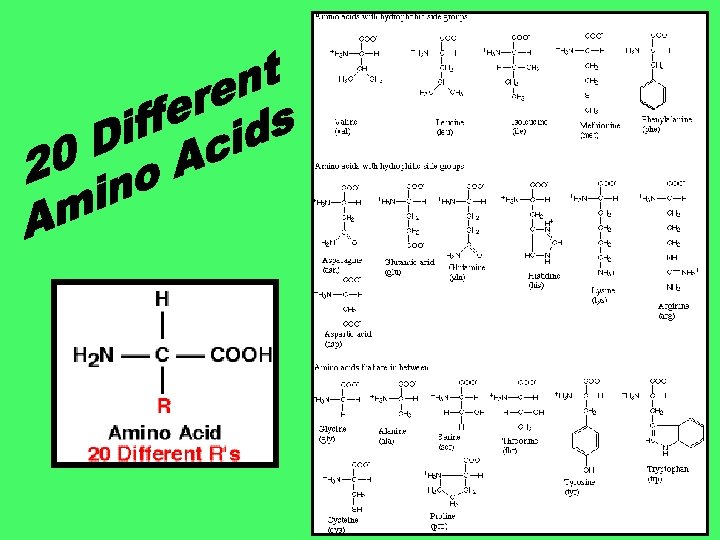
1. Proteins are made up of the elements C, H, O, and N (but in no set ratio). 2. Proteins are chains of Amino Acids (usually 75 or more) more that bond together via dehydration synthesis. 3. 40% of the average human body is made up of protein.
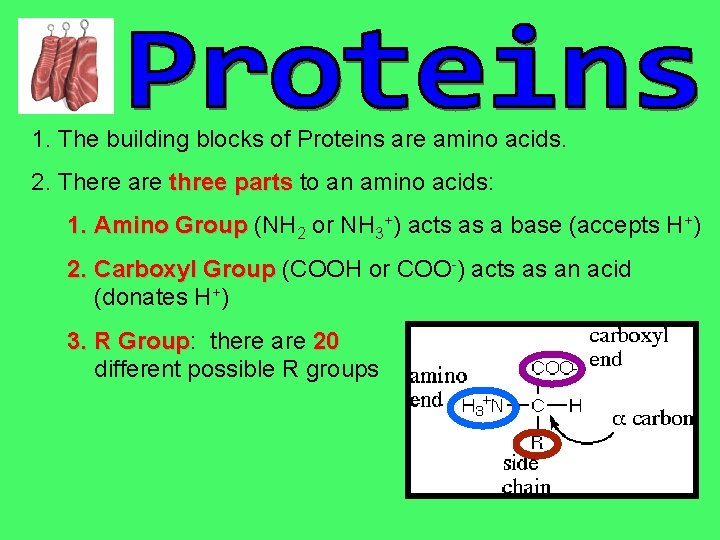
1. The building blocks of Proteins are amino acids. 2. There are three parts to an amino acids: 1. Amino Group (NH 2 or NH 3+) acts as a base (accepts H+) 2. Carboxyl Group (COOH or COO-) acts as an acid (donates H+) 3. R Group: Group there are 20 different possible R groups
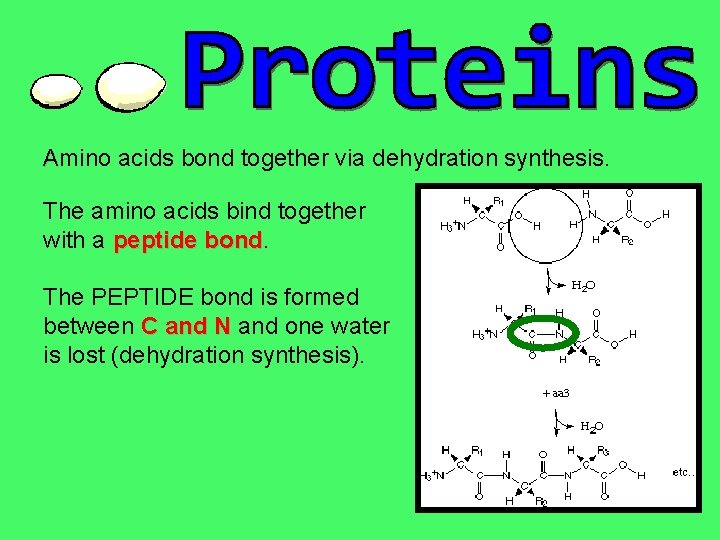
Amino acids bond together via dehydration synthesis. The amino acids bind together with a peptide bond The PEPTIDE bond is formed between C and N and one water is lost (dehydration synthesis).
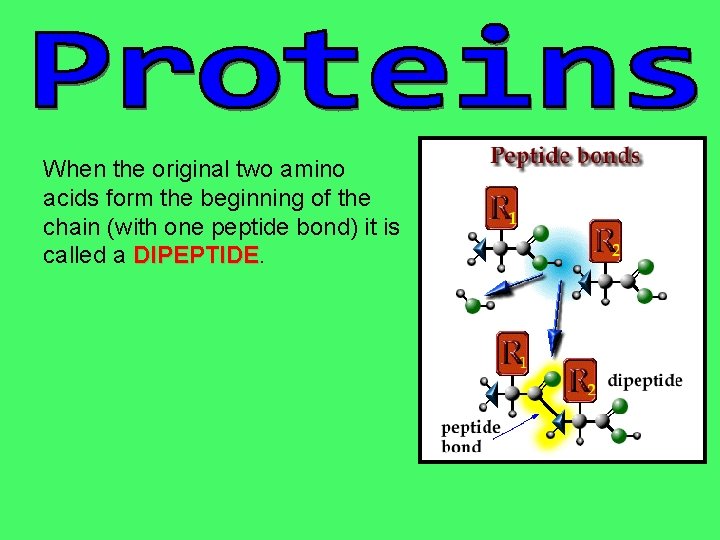
When the original two amino acids form the beginning of the chain (with one peptide bond) it is called a DIPEPTIDE
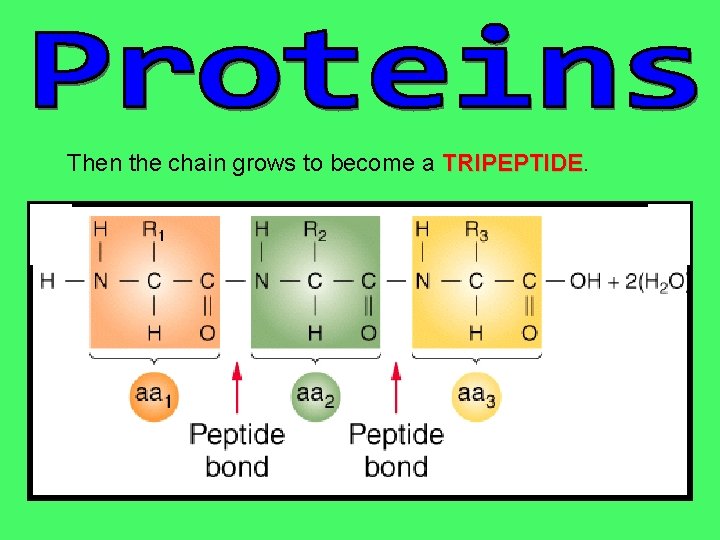
Then the chain grows to become a TRIPEPTIDE
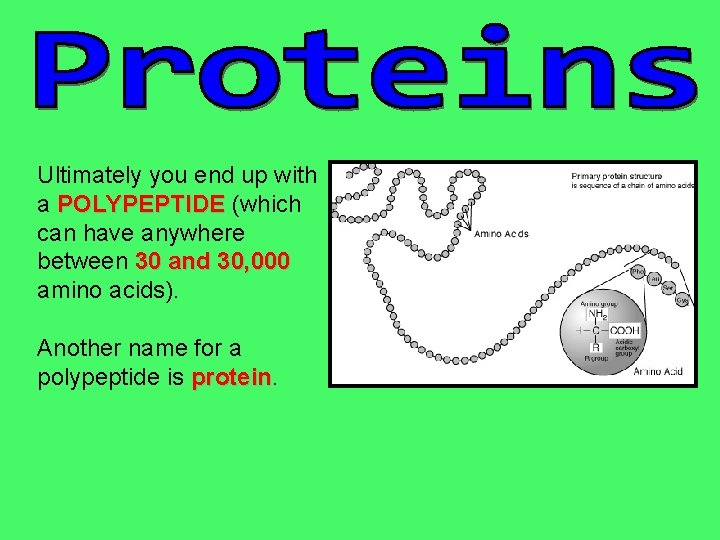
Ultimately you end up with a POLYPEPTIDE (which can have anywhere between 30 and 30, 000 amino acids). Another name for a polypeptide is protein

Every protein is different because the ORDER of amino acids is different. The chains come together differently due to the order of the different R groups and how they bond together. This structural difference also makes the polypeptides (proteins) functionally different.

This is the first level of how proteins are formed. It is simply the order of amino acids joined together with peptide bonds. It is the amino acid sequence that determines the nature and chemistry of the protein. If you change the order of amino acids, the protein may not be able to do its job

This is the second step in the formation of a protein. When a peptide bond is formed, a double bonded oxygen is left over, which is partially negative (the carboxyl group: COO-). It is attracted to the positive NH 3+ amino group from other amino acids in the chain. This attraction forms a HYDROGEN BOND This causes the chain to twist into either a spiral called an alpha helix or a beta pleated sheet
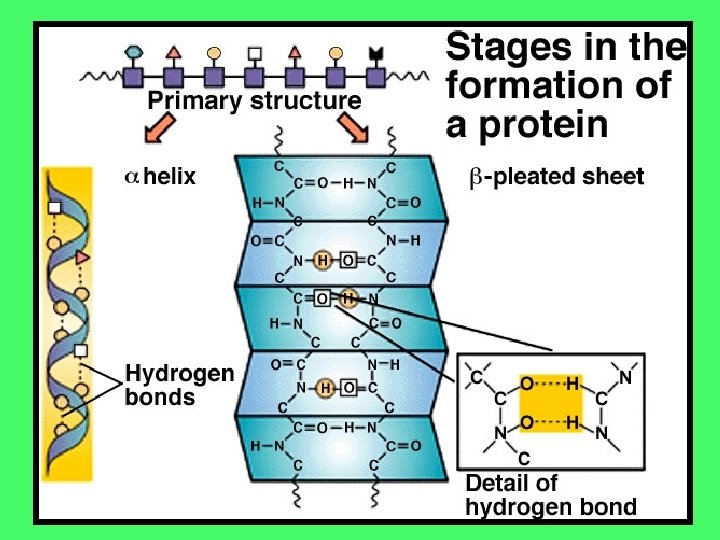

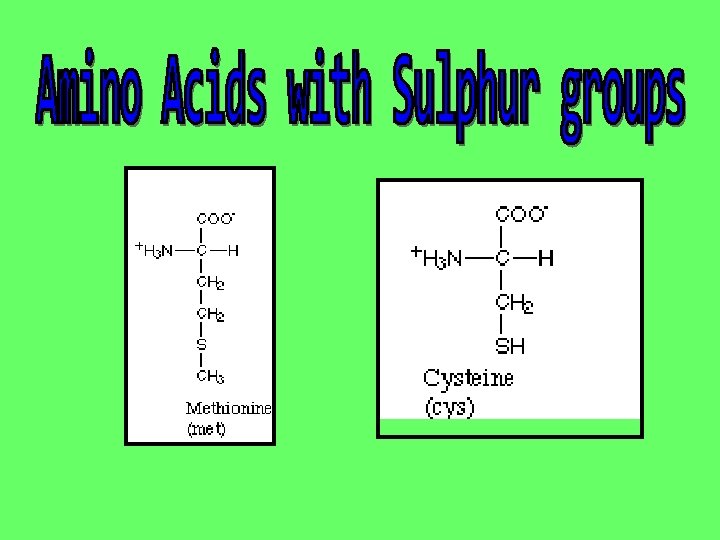
The next interactions take place between the R groups Some R groups are reactive and will interact with other reactive R groups in the chain. These are the amino acids that are either charged or that have a sulphur atom The interactions ( + and – attractions and S-S bridges) bridges will fold the molecule over into a highly specific 3 -dimensional shape It is the 3 -D shape that will determine the protein’s job or role in the body.

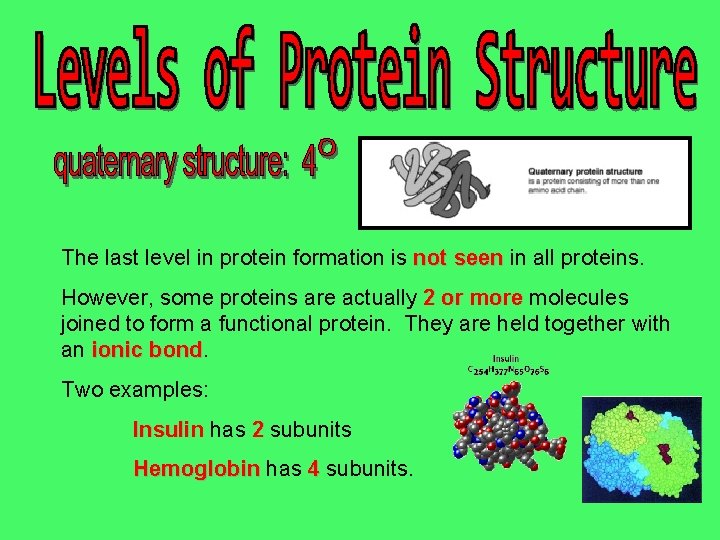
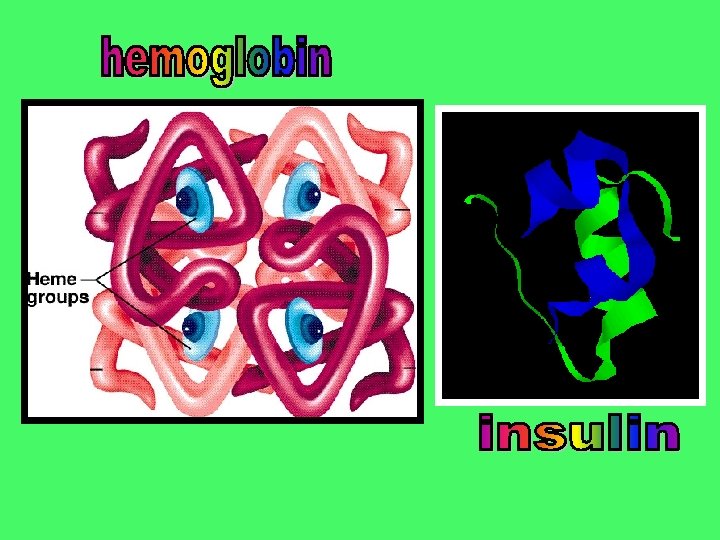
The last level in protein formation is not seen in all proteins. However, some proteins are actually 2 or more molecules joined to form a functional protein. They are held together with an ionic bond Two examples: Insulin has 2 subunits Hemoglobin has 4 subunits.
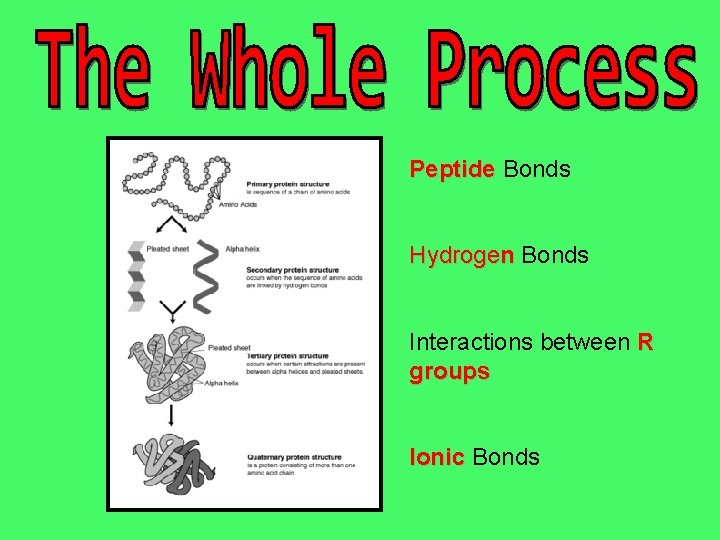
Peptide Bonds Hydrogen Bonds Interactions between R groups Ionic Bonds
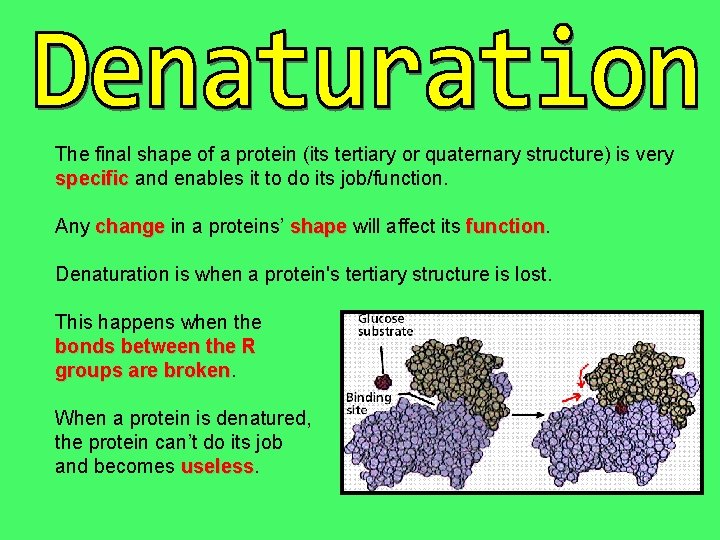
The final shape of a protein (its tertiary or quaternary structure) is very specific and enables it to do its job/function. Any change in a proteins’ shape will affect its function Denaturation is when a protein's tertiary structure is lost. This happens when the bonds between the R groups are broken When a protein is denatured, the protein can’t do its job and becomes useless

How can this happen? There are three common ways: 1. Temperature: High temperatures affect the weak Hydrogen bonds and can distort or break them, thus changing the structural shape. A slight increase in temperature an cause a reversible change (ie: fever). A high temperature increase can cause an irreversible change (ie: cooking an egg).

How can this happen? There are three common ways: 2. Chemicals: Heavy metals such as lead and mercury are large atoms that are attracted the R groups of amino acids. They bond to the R group and distort the protein’s shape. This is usually irreversible (they usually don’t want to ‘let go’).
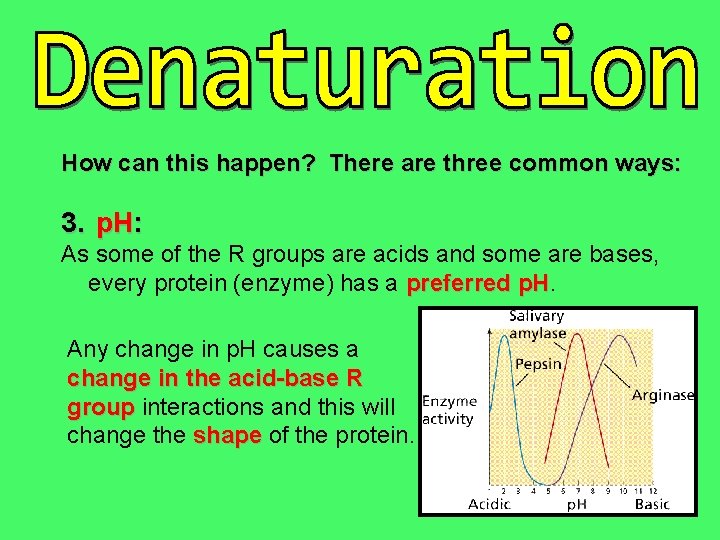
How can this happen? There are three common ways: 3. p. H: As some of the R groups are acids and some are bases, every protein (enzyme) has a preferred p. H Any change in p. H causes a change in the acid-base R group interactions and this will change the shape of the protein.
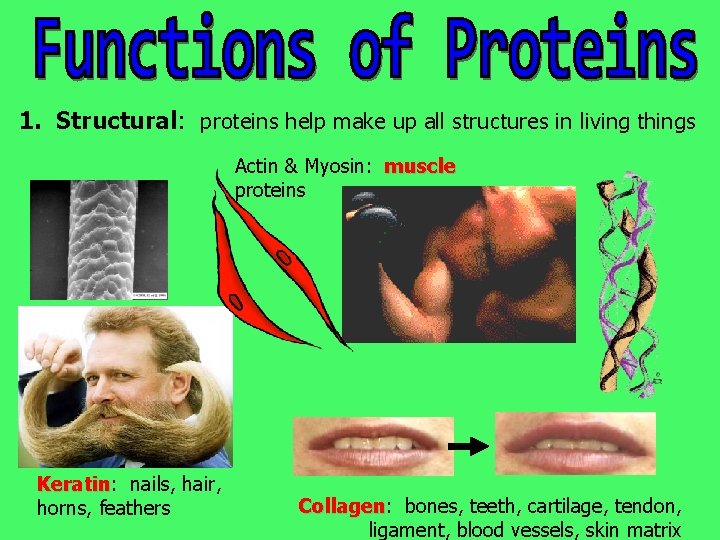
1. Structural: proteins help make up all structures in living things Actin & Myosin: muscle proteins Keratin: Keratin nails, hair, horns, feathers Collagen: Collagen bones, teeth, cartilage, tendon, ligament, blood vessels, skin matrix
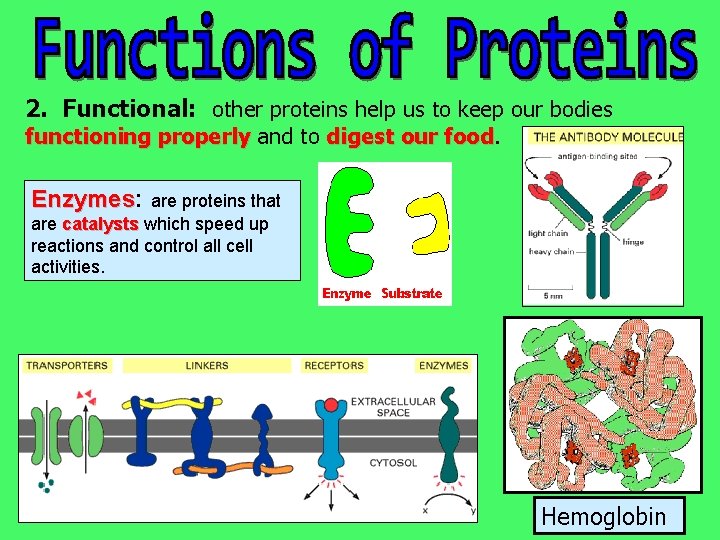
2. Functional: other proteins help us to keep our bodies functioning properly and to digest our food Enzymes: Enzymes are proteins that are catalysts which speed up reactions and control all cell activities. Hemoglobin

3. Food Source: Source once we have used up all of our carbohydrates and fats, proteins will be used for energy. Proteins are worth the least amount of energy per gram. Anorexia and Bolimia

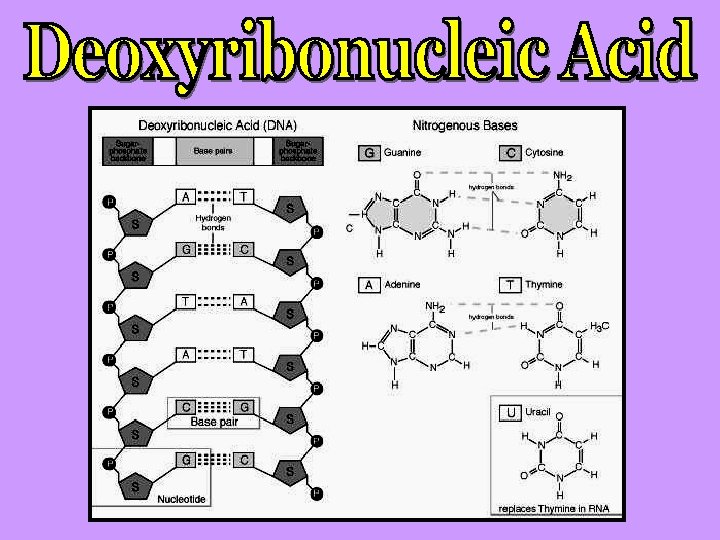
Nucleic acids are acidic molecules that are found in the nucleus of cells. There are two types, both of which are very LARGE 1. DNA: Deoxyribonucleic Acid 2. RNA: Ribonucleic Acid
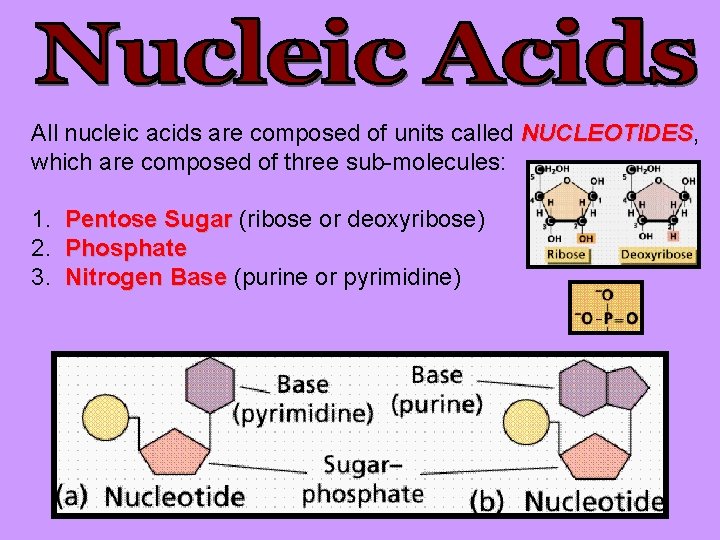
All nucleic acids are composed of units called NUCLEOTIDES, NUCLEOTIDES which are composed of three sub-molecules: 1. 2. 3. Pentose Sugar (ribose or deoxyribose) Phosphate Nitrogen Base (purine or pyrimidine)
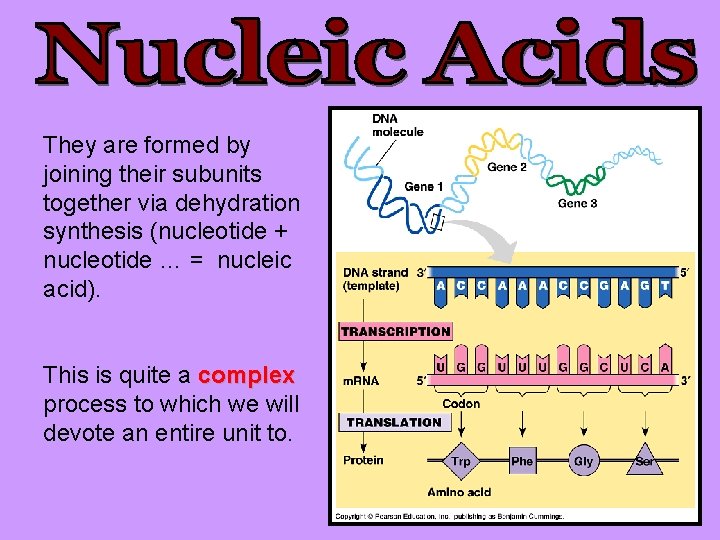
They are formed by joining their subunits together via dehydration synthesis (nucleotide + nucleotide … = nucleic acid). This is quite a complex process to which we will devote an entire unit to.
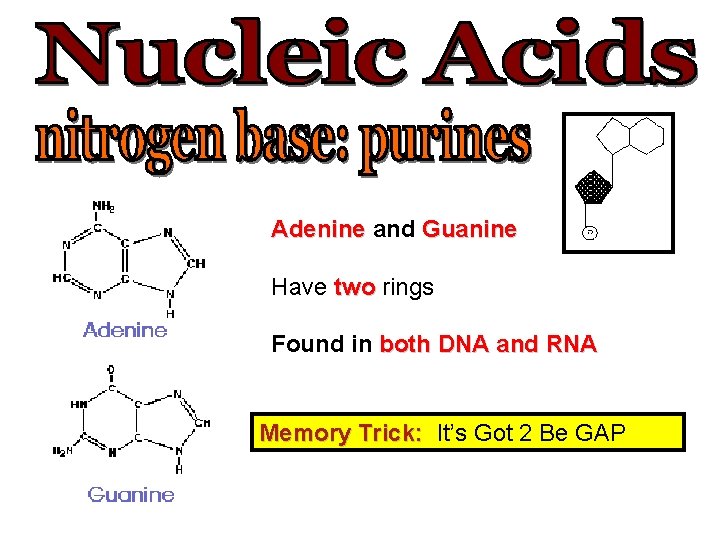
Adenine and Guanine Have two rings Found in both DNA and RNA Memory Trick: It’s Got 2 Be GAP
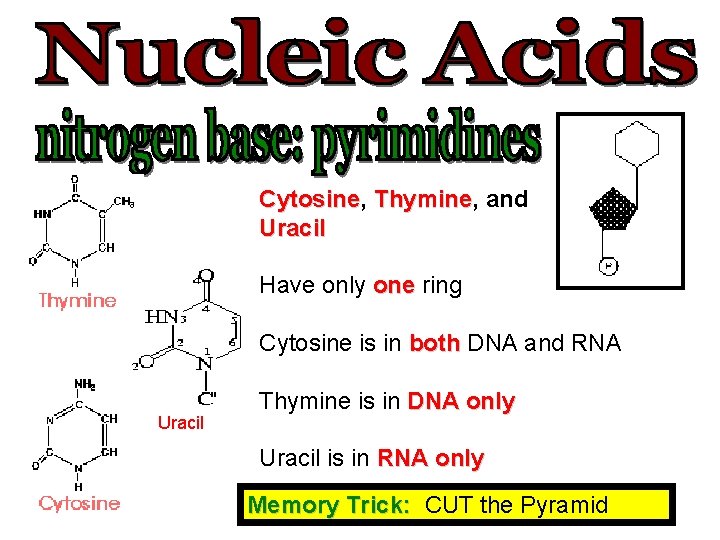
Cytosine, Cytosine Thymine, Thymine and Uracil Have only one ring Cytosine is in both DNA and RNA Uracil Thymine is in DNA only Uracil is in RNA only Memory Trick: CUT the Pyramid
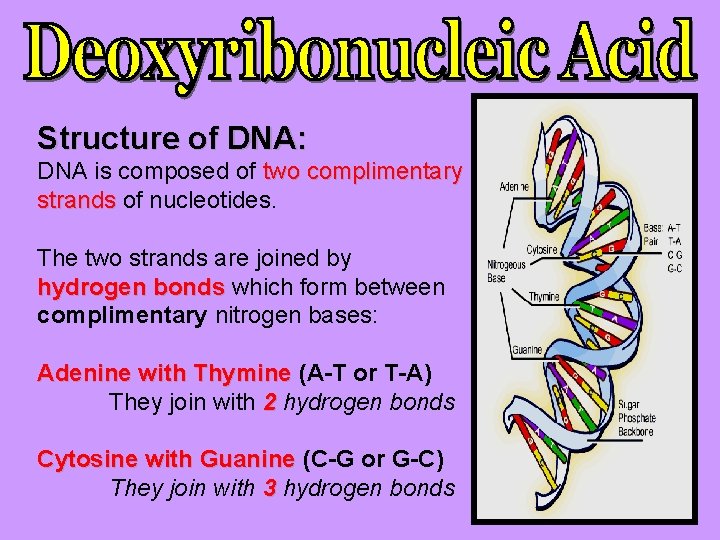
Structure of DNA: DNA is composed of two complimentary strands of nucleotides. The two strands are joined by hydrogen bonds which form between complimentary nitrogen bases: Adenine with Thymine (A-T or T-A) They join with 2 hydrogen bonds Cytosine with Guanine (C-G or G-C) They join with 3 hydrogen bonds
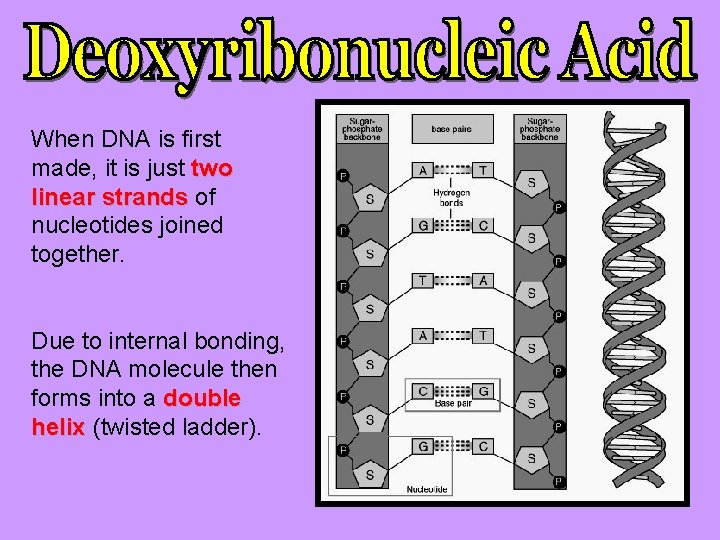
When DNA is first made, it is just two linear strands of nucleotides joined together. Due to internal bonding, the DNA molecule then forms into a double helix (twisted ladder).

a) Directs and controls all cell activities by making all of the proteins and enzymes b) Contains all of the genetic information necessary to make one complete organism of very exact specifications
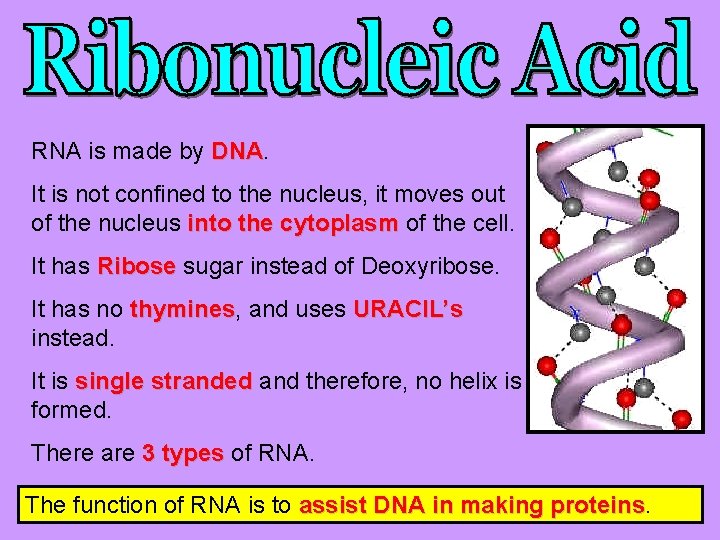
RNA is made by DNA It is not confined to the nucleus, it moves out of the nucleus into the cytoplasm of the cell. It has Ribose sugar instead of Deoxyribose. It has no thymines, thymines and uses URACIL’s instead. It is single stranded and therefore, no helix is formed. There are 3 types of RNA. The function of RNA is to assist DNA in making proteins
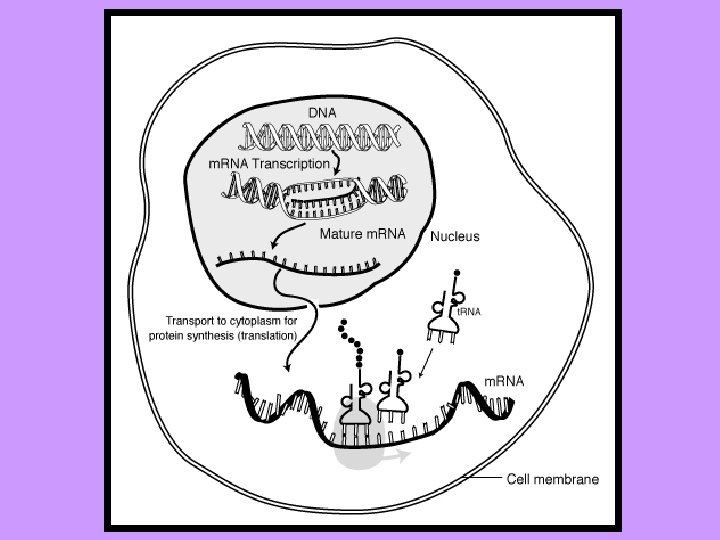
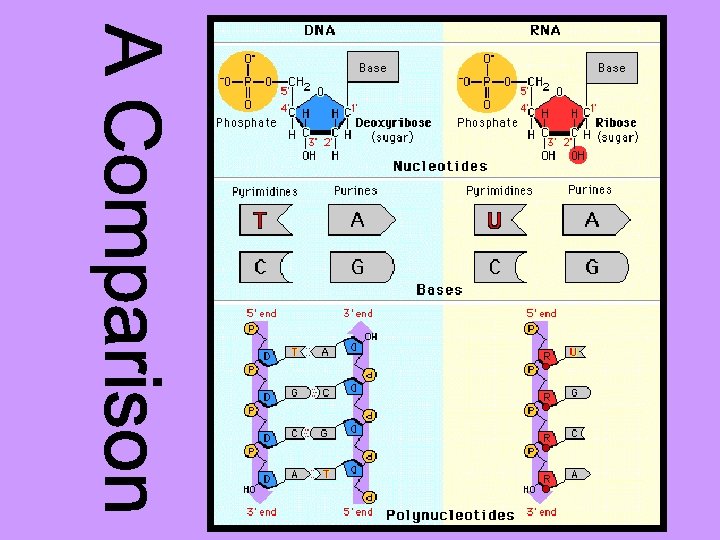
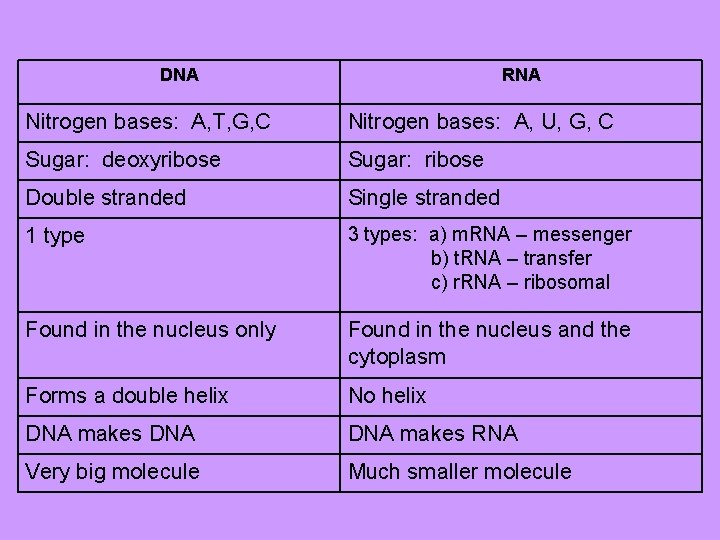
DNA RNA Nitrogen bases: A, T, G, C Nitrogen bases: A, U, G, C Sugar: deoxyribose Sugar: ribose Double stranded Single stranded 1 type 3 types: a) m. RNA – messenger b) t. RNA – transfer c) r. RNA – ribosomal Found in the nucleus only Found in the nucleus and the cytoplasm Forms a double helix No helix DNA makes RNA Very big molecule Much smaller molecule
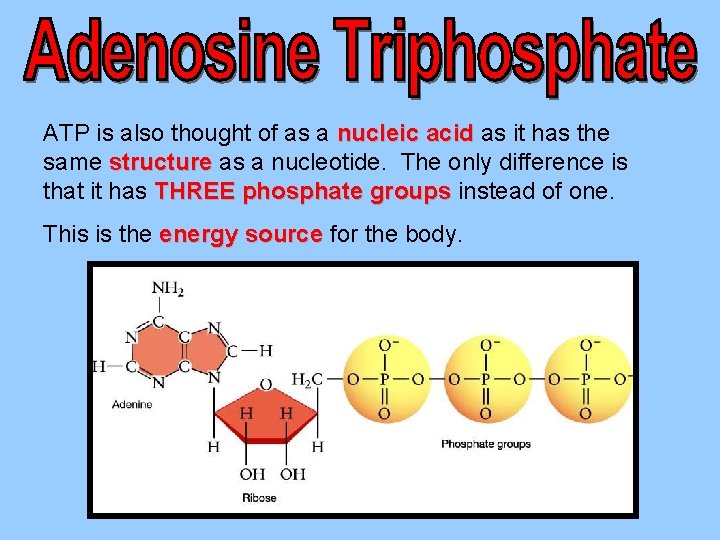
ATP is also thought of as a nucleic acid as it has the same structure as a nucleotide. The only difference is that it has THREE phosphate groups instead of one. This is the energy source for the body.
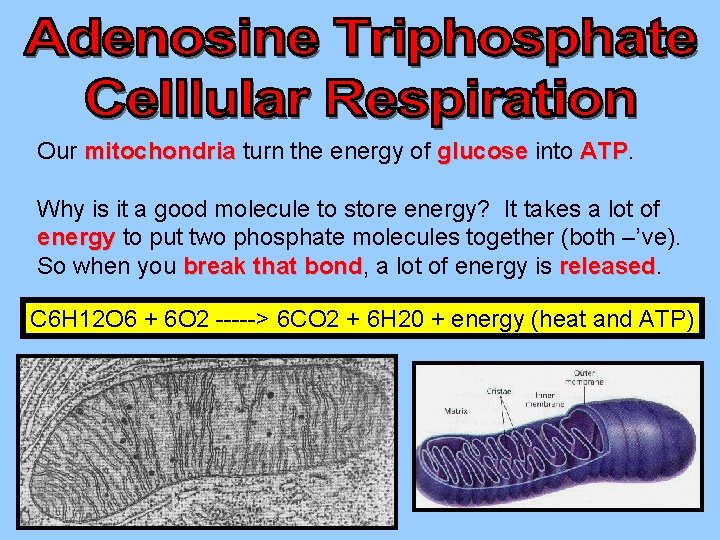
Our mitochondria turn the energy of glucose into ATP Why is it a good molecule to store energy? It takes a lot of energy to put two phosphate molecules together (both –’ve). So when you break that bond, bond a lot of energy is released C 6 H 12 O 6 + 6 O 2 -----> 6 CO 2 + 6 H 20 + energy (heat and ATP)
- Slides: 97