The most common acidbase indicators are complex molecules

• The most common acid-base indicators are complex molecules that are themselves weak acids (represented by HIn). • They exhibit one color when the proton is attached to the molecule and another color when the proton is absent. • Indicators are commonly used for titration to mark the end point of a titration (color change). • The end point should be very close to the equivalence point (to reduce error).

For example, phenolphthalein is colorless in its HIn form and pink in its In- form.

• In solution, the indicator is involved in typical acid-base equilibrium: HIn(aq) ⇌ H+(aq) + In-(aq) Acid form (one color) Base form (another color) • The corresponding equilibrium expression, Kin, is given by • In a strongly acidic solution, when the H+ concentration is high, this equilibrium shifts to the left and most of the indicator exists in its “acid form”. • We observe the color for HIn.

• If the solution is made basic, the H+ concentration drops, and the equilibrium shifts to the right, toward In-, and the color we observe is that of the “base form” of the indicator. • The observed change in color for an indicator actually occurs over a range of p. H values. • How much In- must be present for the human eye to detect that the color is different from the original? • For most indicators, about a tenth of the initial form must be converted to the other form before a new color is apparent. • In a titration of an acid with a base, the color change will occur at a p. H where,

• In a typical acid-base titration, there is a sudden and large p. H change around the equivalence point. • The large swing in p. H causes a sudden shift in the equilibrium position for the indicator. • We go from the HIn form to the I- form and hence a color change. • To pick an indicator best suited for a titration assumes that the midpoint of the color change for the indicator corresponds to the p. H at the eq. point. • In other words, we want an indicator end point (where the color changes) and the titration equivalence point to be as close as possible.

• At this midpoint there will be equal amounts of HIn and In-. • This leads to • So Kin = [H+], and taking the negative log of both sides gives us p. KIn = p. H (at eq. point). • The titration curve of HCl and Na. OH (strong acid/strong base). • Note the endpoints of phenolphthalein and methyl red occur at virtually the same amount of added Na. OH. • Either would be a suitable choice for indicator.
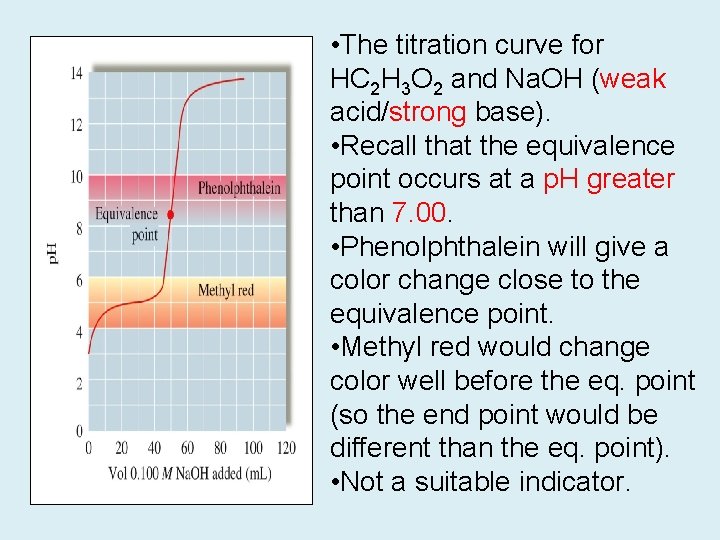
• The titration curve for HC 2 H 3 O 2 and Na. OH (weak acid/strong base). • Recall that the equivalence point occurs at a p. H greater than 7. 00. • Phenolphthalein will give a color change close to the equivalence point. • Methyl red would change color well before the eq. point (so the end point would be different than the eq. point). • Not a suitable indicator.
- Slides: 7