The Molecules of Cells Carbon Organic Compound Contain
The Molecules of Cells
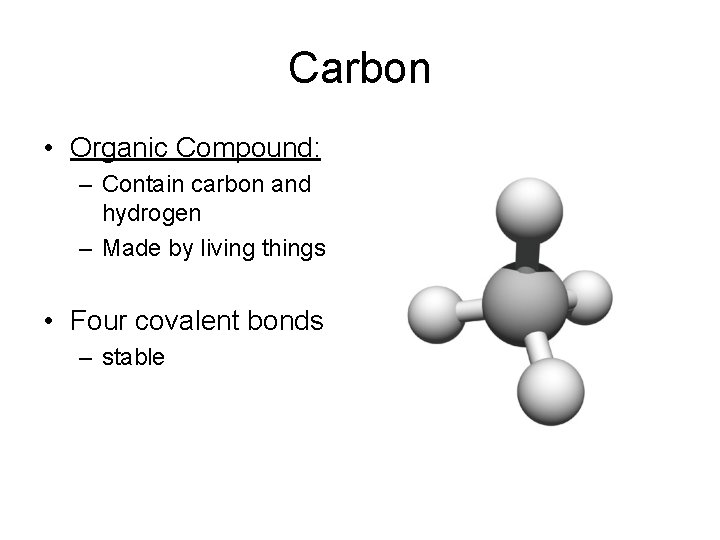
Carbon • Organic Compound: – Contain carbon and hydrogen – Made by living things • Four covalent bonds – stable
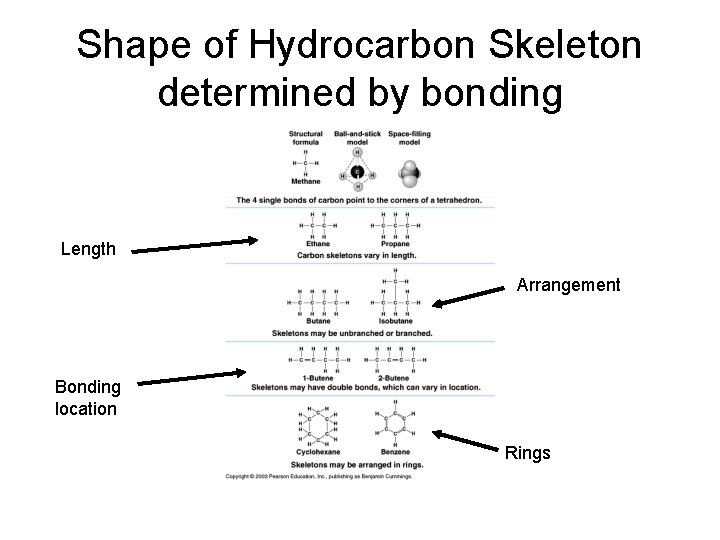
Shape of Hydrocarbon Skeleton determined by bonding Length Arrangement Bonding location Rings

Functional Groups • Attach to hydrocarbon skeleton • Chemically reactive • Usually polar • Question: is the molecule soluble or insoluble in water? Why?
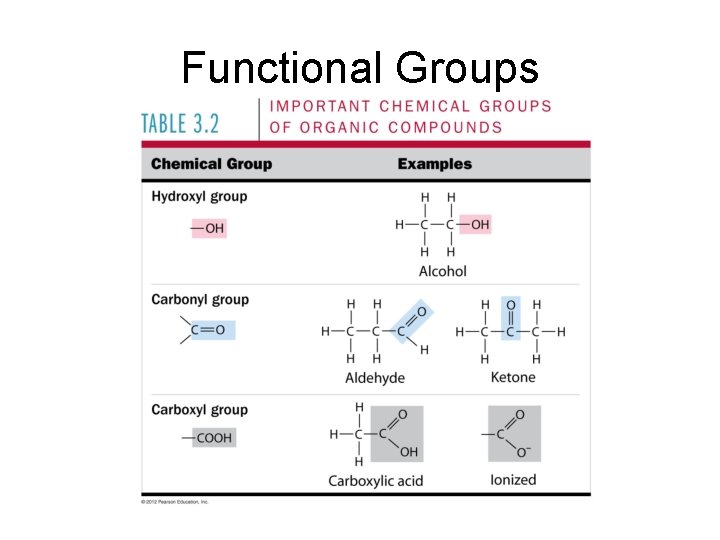
Functional Groups
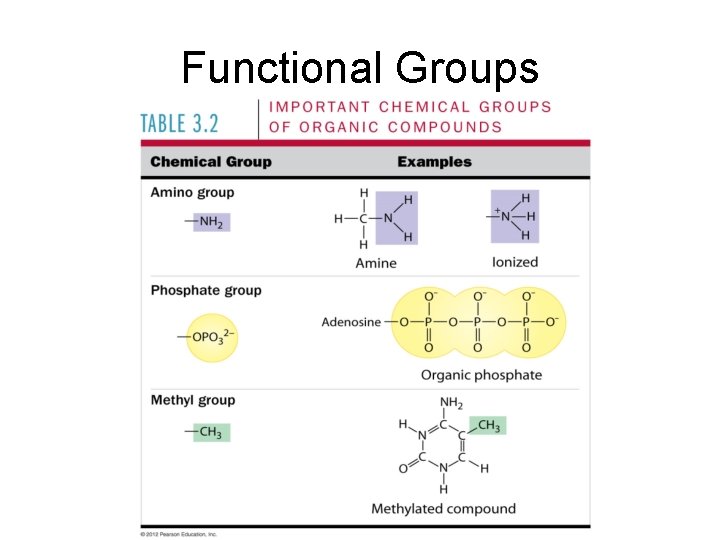
Functional Groups

Carbohydrates • C, H, O – H&O – always 2: 1 ratio • Sugars & Starches • Used for – Cellular energy – Raw material for amino acids – Plant support and structure • Monomer - monosaccaride
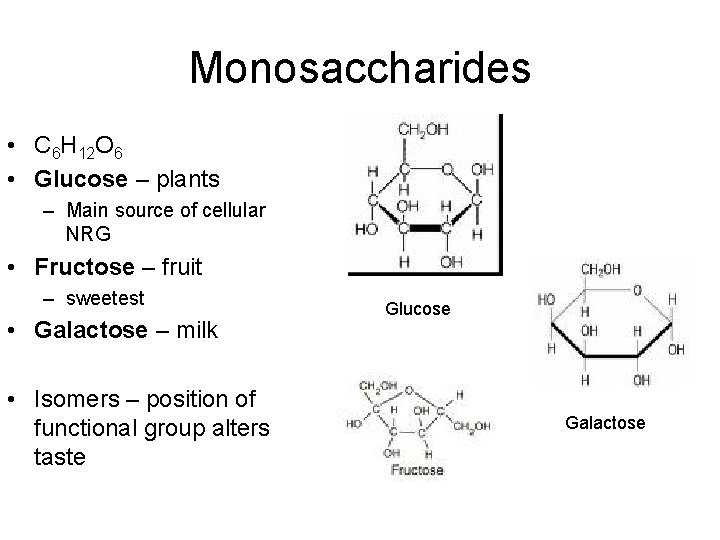
Monosaccharides • C 6 H 12 O 6 • Glucose – plants – Main source of cellular NRG • Fructose – fruit – sweetest • Galactose – milk • Isomers – position of functional group alters taste Glucose Galactose
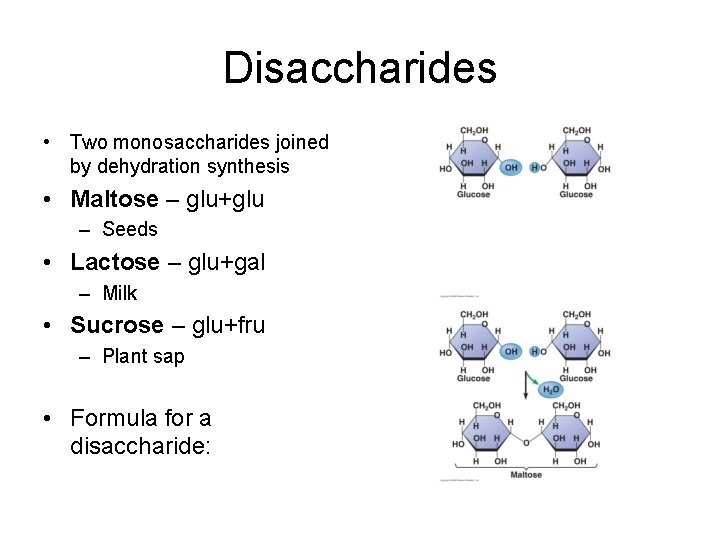
Disaccharides • Two monosaccharides joined by dehydration synthesis • Maltose – glu+glu – Seeds • Lactose – glu+gal – Milk • Sucrose – glu+fru – Plant sap • Formula for a disaccharide:
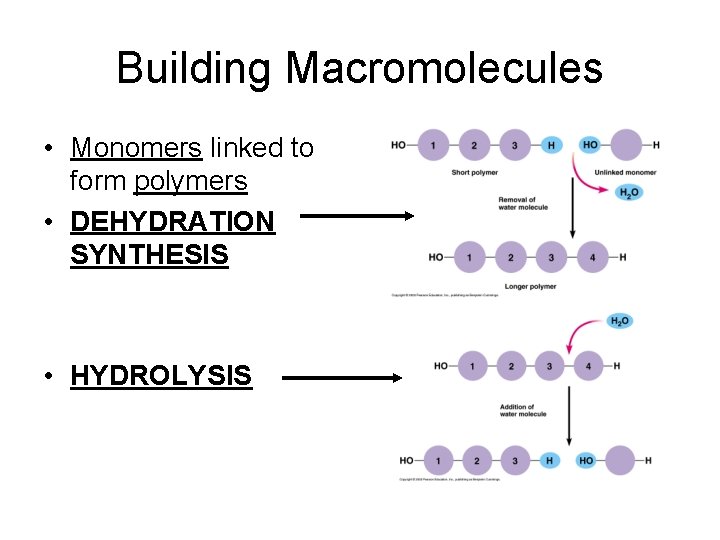
Building Macromolecules • Monomers linked to form polymers • DEHYDRATION SYNTHESIS • HYDROLYSIS

Dehydration Synthesis • Enjoy the cheesy video!!
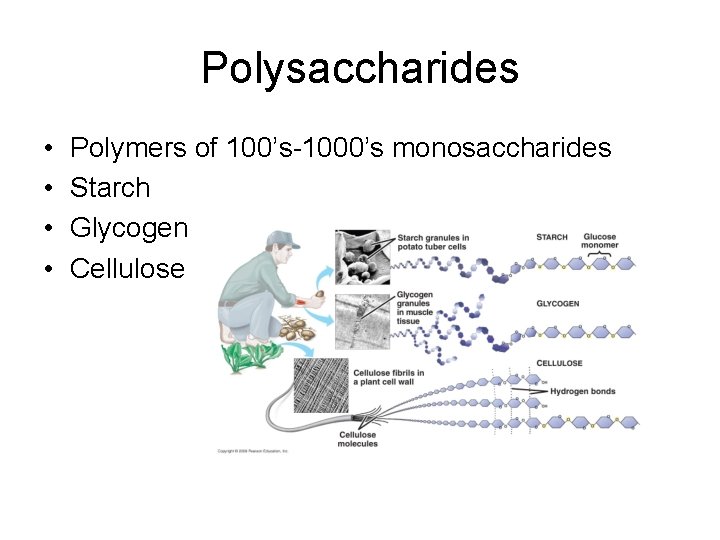
Polysaccharides • • Polymers of 100’s-1000’s monosaccharides Starch Glycogen Cellulose
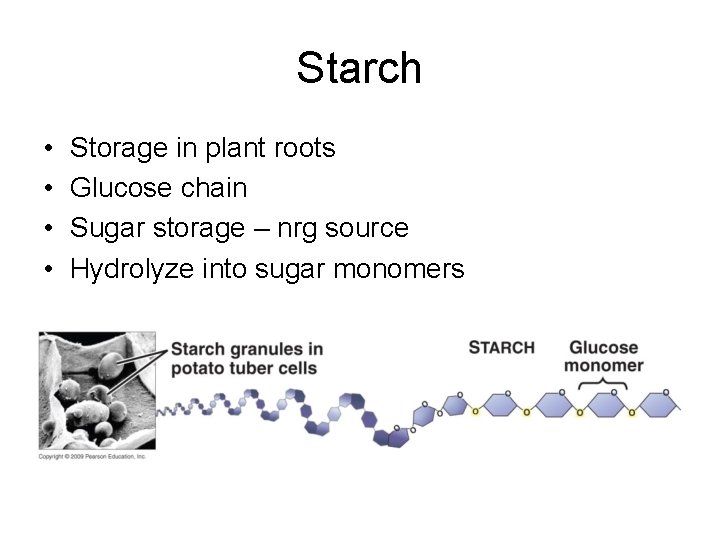
Starch • • Storage in plant roots Glucose chain Sugar storage – nrg source Hydrolyze into sugar monomers

Glycogen • Identical to starch except more branched • Stored in animal liver and muscles • Hydrolyzed to get more sugar/nrg
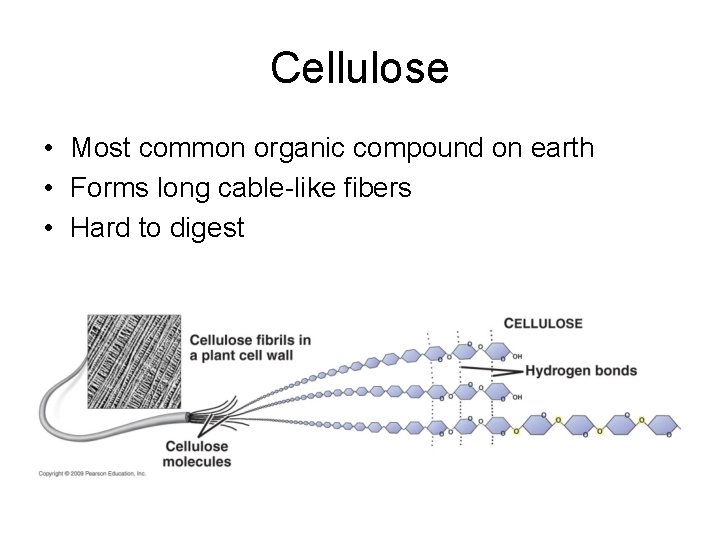
Cellulose • Most common organic compound on earth • Forms long cable-like fibers • Hard to digest

High Fructose Corn Syrup What’s the fuss over HFCS?

Lipids • Mostly C, H very little O • Non-polar – hydrophobic • Fats, Oils, Waxes, Phospholipids, Steroids Chicken fat
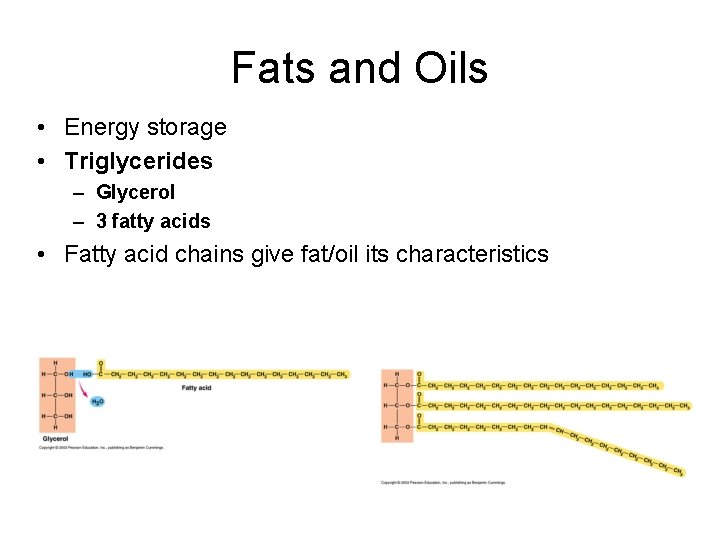
Fats and Oils • Energy storage • Triglycerides – Glycerol – 3 fatty acids • Fatty acid chains give fat/oil its characteristics

Cheesy Video!! • The making of a triglyceride
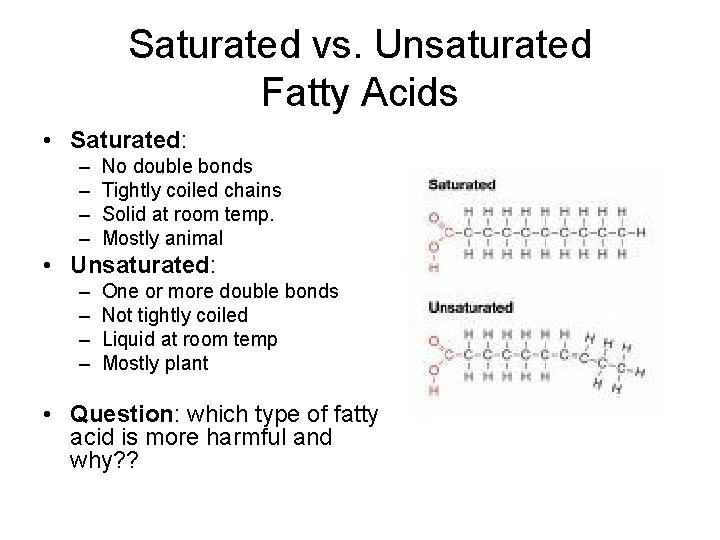
Saturated vs. Unsaturated Fatty Acids • Saturated: – – No double bonds Tightly coiled chains Solid at room temp. Mostly animal • Unsaturated: – – One or more double bonds Not tightly coiled Liquid at room temp Mostly plant • Question: which type of fatty acid is more harmful and why? ?

Problems & Solutions • Atherosclerosis – saturated fats collecting in arteries • Trans Fats – hydrogenating unsat. Fatty acids to make them solid • Why is this a problem? margarine Clogged artery
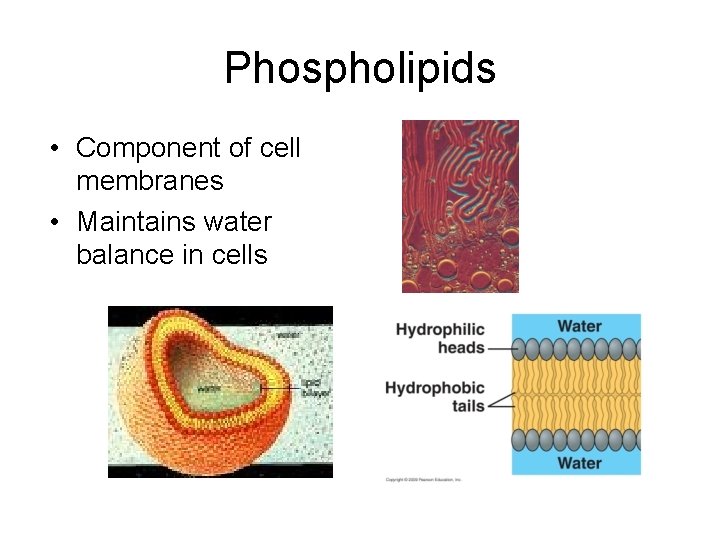
Phospholipids • Component of cell membranes • Maintains water balance in cells

Waxes • Most hydrophobic • Natural waterproofing – Fruit skin – Insect exoskeletons – What else? ?
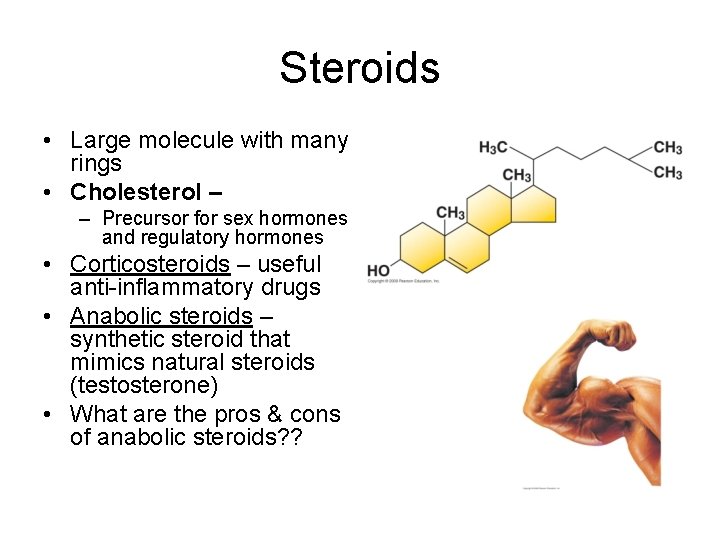
Steroids • Large molecule with many rings • Cholesterol – – Precursor for sex hormones and regulatory hormones • Corticosteroids – useful anti-inflammatory drugs • Anabolic steroids – synthetic steroid that mimics natural steroids (testosterone) • What are the pros & cons of anabolic steroids? ?
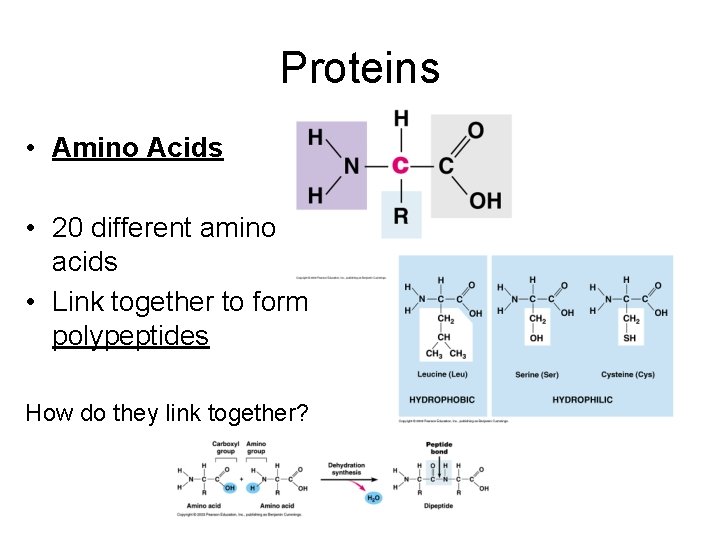
Proteins • Amino Acids • 20 different amino acids • Link together to form polypeptides How do they link together?

Classes of Proteins • • Structural Contractile Storage Defense Transport Signal Enzyme
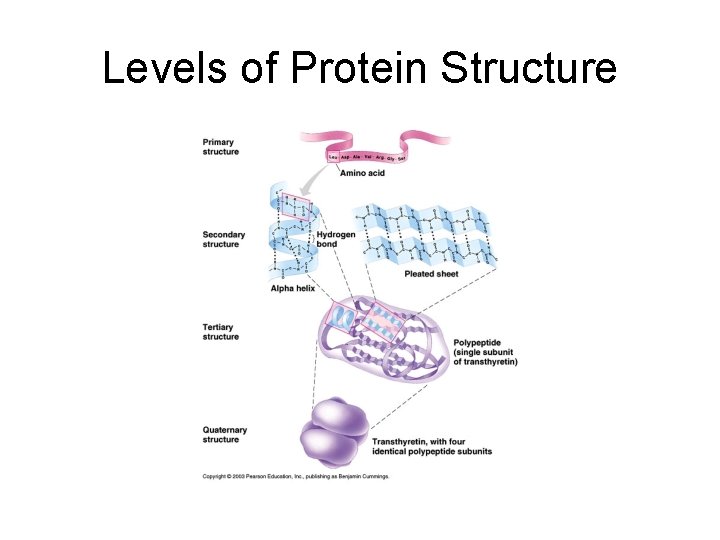
Levels of Protein Structure
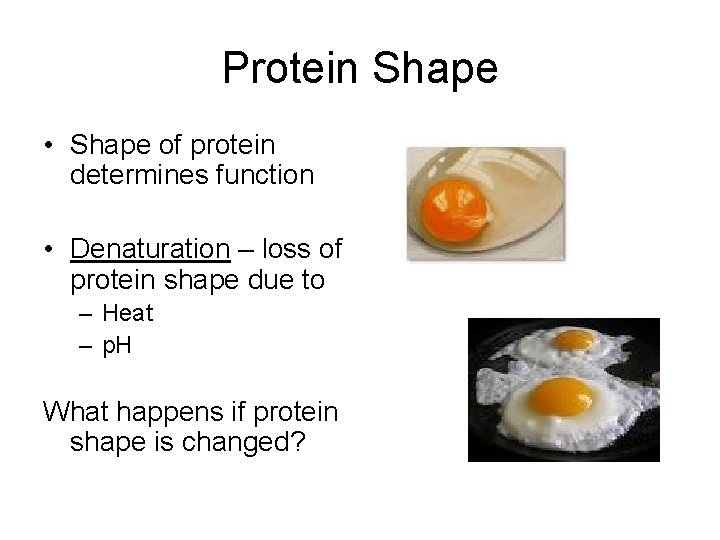
Protein Shape • Shape of protein determines function • Denaturation – loss of protein shape due to – Heat – p. H What happens if protein shape is changed?
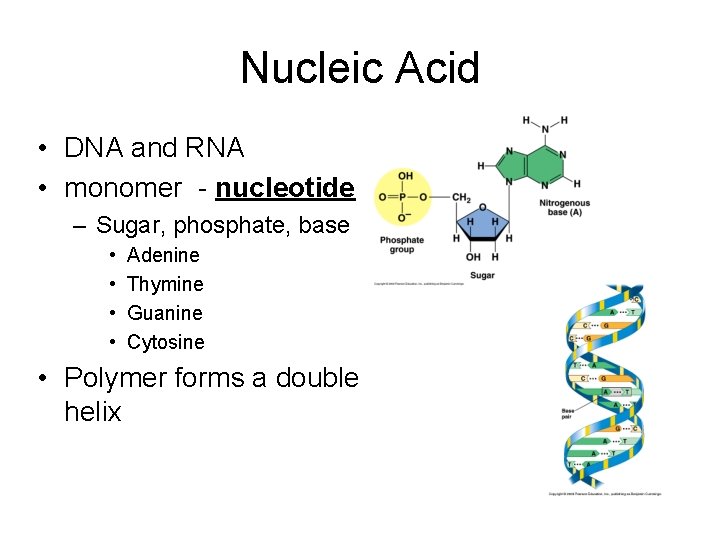
Nucleic Acid • DNA and RNA • monomer - nucleotide – Sugar, phosphate, base • • Adenine Thymine Guanine Cytosine • Polymer forms a double helix
- Slides: 29