The Molecular Orbital Model Molecular Orbital Model The

The Molecular Orbital Model

Molecular Orbital Model The localized electron model works very well with the prediction of structure and bonding of molecules, but the electron correlation problem still exists. Since we do not know the details of the electron movements, we cannot deal with the electron-electron interactions in a specific way The Molecular Orbital model helps us to deal with the molecular problem.

Molecular Orbitals Molecular orbitals (MOs) have many of the same characteristics as atomic orbitals. Two of the most important are: MOs can hold two electrons with opposite spins. The square of the MO’s wave function indicates electron probability.

MOs For simplicity we will first look at the H 2 molecule. The combination of hydrogen 1 s atomic orbitals results in 2 molecular orbitals. The wave phases of the atomic orbitals combine/overlap. Since electrons move in wave functions, this causes constructive and destructive interference in the wave pattern. When the orbitals are added, the matching phases produce constructive interference and the opposite phases produce destructive interference.

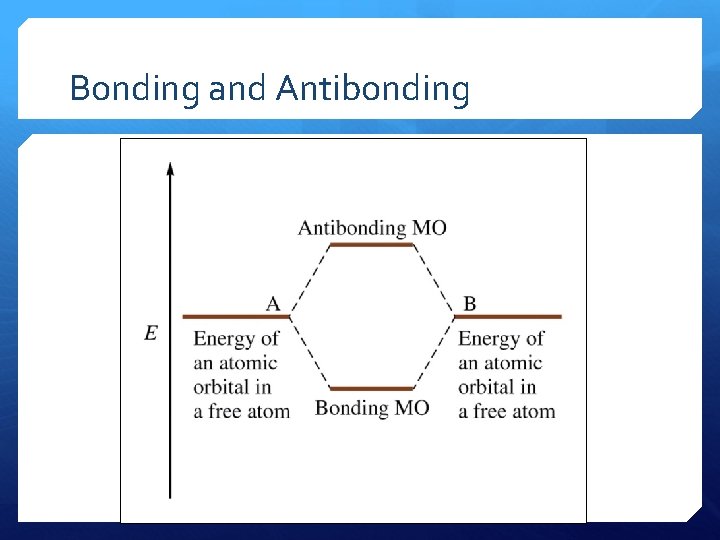
MOs A constructive combination gives a bonding MO. This gives an enhanced electron probability between the nuclei. The destructive combination gives an antibonding MO. This interference produces a node between the nuclei.

MOs Two MOs exist for H 2: MO 1= 1 s. H 1 + 1 s. H 2 MO 1 is constructive and therefore a bonding MO 1 is lower energy MO 2 = 1 s. H 1 – 1 s. H 2 MO 2 is destructive and therefore an antibonding MO 2 is higher energy

MOs The type of electron distribution described in these MOs is called sigma as in the localized electron model. MO 1 and MO 2 are sigma (σ) molecular orbitals. In this molecule only the molecular orbitals are available for occupation by electrons. The 1 s atomic orbitals of the hydrogen atoms no longer exist, because the H 2 molecule – a new entity – has its own set of new orbitals.
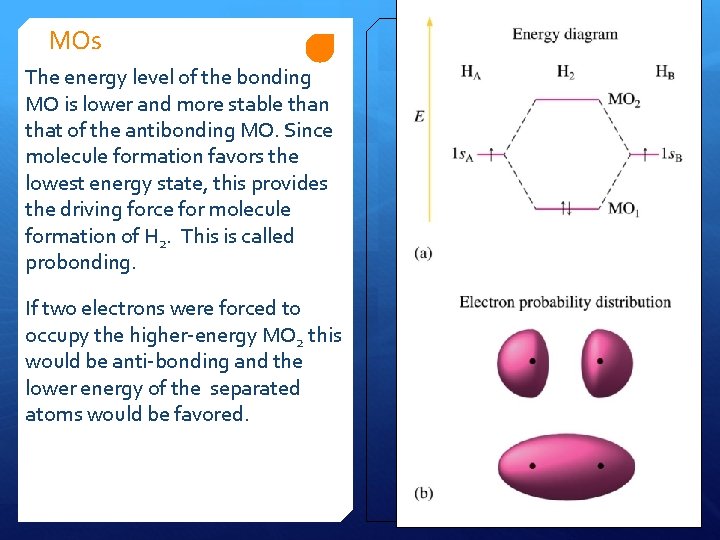
MOs The energy level of the bonding MO is lower and more stable than that of the antibonding MO. Since molecule formation favors the lowest energy state, this provides the driving force for molecule formation of H 2. This is called probonding. If two electrons were forced to occupy the higher-energy MO 2 this would be anti-bonding and the lower energy of the separated atoms would be favored.
Bonding and Antibonding

MOs Labels are given to MOs indicate their symmetry (shape), the parent atomic orbitals, and whether they are bonding or antibonding. Antibonding character is indicated by an asterisk. Subscripts indicate parent orbitals σ and π indicate shape. H 2 has the following MOs: MO 1 = σ1 s MO 2 = σ1 s*

MOs Molecular electron configurations can be written in much the same way as atomic (electron) configurations. Since the H 2 molecule has two electrons in the σ1 s molecular orbital, the electron configuration is: σ1 s 2 Each molecular orbital can hold two electrons, but the spins must be opposite. Orbitals are conserved. The number of molecular orbitals will always be the same as the number of atomic orbitals used to construct them.

MOs From this molecular electron configuration, we can determine a molecules stability. Would H 2 - be stable? (σ1 s )2 ( σ1 s*) 1 The key idea is that H 2 - would exist if it were a lower energy than its separated parts. Two electrons are in bonding and one is in antibonding. Since more electrons favor bonding H 2 - is formed. This also is a good indicator of bond strength. H 2 has a stronger bond than H 2 -. The net lowering of the bonding electrons by one is a direct relationship to bond strength. H 2 is twice as strong.

Bond Order To indicate bond strength, we use the concept of bond order. Example: H 2 has a bond order of 1 H 2 - has a bond order of ½

Bond Order Bond order is an indication of bond strength because it reflects the difference between the number of bonding electrons and the number of antibonding electrons. Larger bond order means greater bond strength. Bond order of 0 gives us a molecule that doesn’t exist.
https: //www. youtube. com/watch? v=tmqyl. JAEd. DE
- Slides: 15