The MOLE The mole is one of the


















- Slides: 19

The MOLE!!!

The mole is one of the big concepts of Chemistry. The mole is a counting number, just like 1 dozen = 12 things 1 score = 20 things 1 ream = 500 things 1 mole = 6. 022 x 1023 things or 1 mole = 602, 200, 000, 000 things We use moles in Chemistry b/c actually counting things would take too long.

st 1 Definition of a mole 6. 022 x 1023 = Avogadro’s number just talks about numbers of particles like atoms (elements), molecules (covalent compounds) and formula units (ionic compounds).

1 mole C = 6. 022 x 1023 atoms of C 1 mole H 2 O = 6. 022 x 1023 molecules of H 2 O 1 mole Al. Cl 3 = 6. 022 x 1023 formula units of Al. Cl 3

1 mole H 2 O = 6. 022 x 1023 molecules of H 2 O 1 mole H 2 O = ? atoms 1 molecule of water has 3 atoms (6. 022 x 1023 x 3) 1 mole H 2 O = 1. 807 x 1024 atoms You can also use dimensional analysis

1 mole Al. Cl 3 = 6. 022 x 1023 formula units of Al. Cl 3 1 mole Al. Cl 3 = ? atoms Try it!

1 mole Al. Cl 3 = 6. 022 x 1023 formula units of Al. Cl 3 1 mole Al. Cl 3 = ? atoms Try it! (1 formula unit of Al. Cl 3 has 4 atoms) 1 mole Al. Cl 3 = 2. 409 x 1024 atoms

nd 2 definition – Molar mass However, elements have different masses, So 1 mole of everything is also going to have different masses. Ex. 5 grapes vs. 5 grapefruits Same # of fruit, but very different masses

Molar Mass – nd 2 definition of a mole The decimal numbers on the Periodic Table tell you the average atomic mass of an element in amu. By definition that is also the mass of that element in grams of one mole of that element. Ex. 1 atom Ca = 40. 078 amu Ca 1 mole Ca = 40. 078 grams Ca 1 mol Na = 22. 990 g Na

Molar Mass – nd 2 definition of a mole 1 mol Cl 2 2 Cl = 70. 906 g Cl 2 1 mol Al(Cl. O 2)3 1 Al + 3 Cl + 6 O = 229. 335 g Al(Cl. O 2)3 1 mol P 2 O 3 2 P + 3 O= 109. 945 g P 2 O 3 these are known as molar masses (the mass of one mole) Molar is the adjective form of mole

Molar Volume 3 rd definition of a mole 1 mole of a gas = 22. 4 Liters of a gas (for gases only at STP, standard temperature and pressure) 1 mol O 2 = 22. 4 L O 2 1 mol CO 2 = 22. 4 L CO 2

Molar Conversions 1 mole = molar mass (g) (grams from the Periodic Table) 1 mole = volume (L) (22. 4 L of a gas at STP) 1 mole = # of things (atoms) (6. 022 x 1023 atoms)

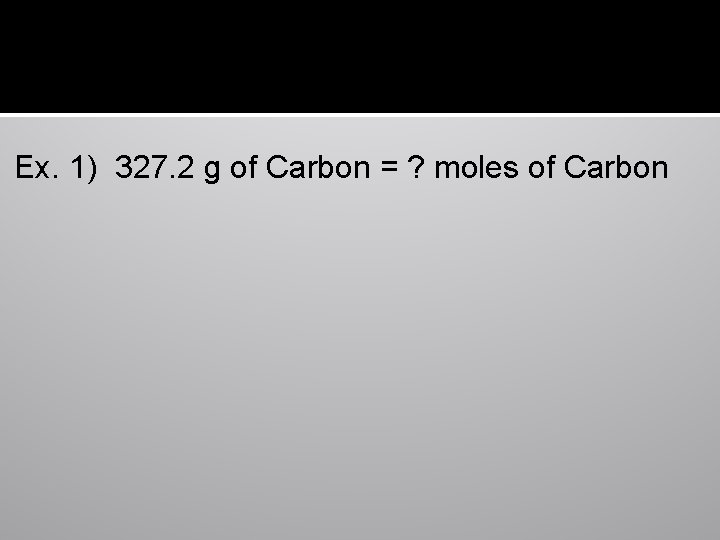
Ex. 1) 327. 2 g of Carbon = ? moles of Carbon

Ex. 2) How many grams are in 3. 295 x 1020 atoms of Copper?

Ex. 3) If you capture 17 grams of Chlorine gas how many liters do you have?

Ex. 4) Determine the amount of grams in 0. 250 moles of sodium phosphate.

Ex. 5) Find the number of sodium atoms in 0. 77 moles of sodium phosphate.

Michael Offutt - A Mole is a Unit Music Video �http: //www. youtube. com/watch? v=Qg 0 Lajw ew 3 A

Mole day videos Happy Mole Day to you! http: //www. youtube. com/watch? v=Re. Me 348 Im 2 w&list=FLn. Khde. Ju. C 7 b 73 gw. Wnb. WEr 8 g&index=17&feature=plpp_video AVOGADRO'S BACK http: //www. youtube. com/watch? v=ZLO 47 JPHa. F 0&feature=BFa&list=FLn. Khde. Ju. C 7 b 73 gw. Wnb. WEr 8 g Tom Lehrer CHEMISTRY element song http: //www. youtube. com/watch? v=DYW 50 F 42 ss 8&feature=BFa&list=FLn. Khde. Ju. C 7 b 73 gw. Wnb. WEr 8 g Daniel Radcliffe sings "The Elements“ http: //www. youtube. com/watch? v=r. SAai. YKF 0 cs&feature=BFa&list=FLn. Khde. Ju. C 7 b 73 gw. Wnb. WEr 8 g AP Chem Rap http: //www. youtube. com/watch? v=d. Ggz. B 7 xi. F 5 U&feature=BFa&list=FLn. Khde. Ju. C 7 b 73 gw. Wnb. WEr 8 g