The Mole A mole symbol mol is the

The Mole A mole (symbol: mol) is the amount of a substance that contains as many particles as there atoms in exactly 12 g of carbon-12. 12 g Carbon-12 = 1 mole of carbon atoms

Pair (2) Dozen (12) Gross (144) Ream of paper (500) Mole (mol) of particles: (6. 022 x 1023)

A mole of copper atoms fits on this balance! One mole of copper = 63. 55 g/mol How many atoms is this?

In the 1800's, an Italian scientist by the name of Amadeo Avogadro mathematically determined… …that a mole of atoms of any element is equal to the mass of that element in grams. A mole of Carbon atoms has a mass of 12. 01 g. (Notice the periodic table)

AVOGADRO’S NUMBER 6. 022 1023 equal to the number of particles in exactly one mole of a pure substance. 6. 022 23 10 particles/mol

CFU:

LETS TRY IT! 39 39 78 94 g
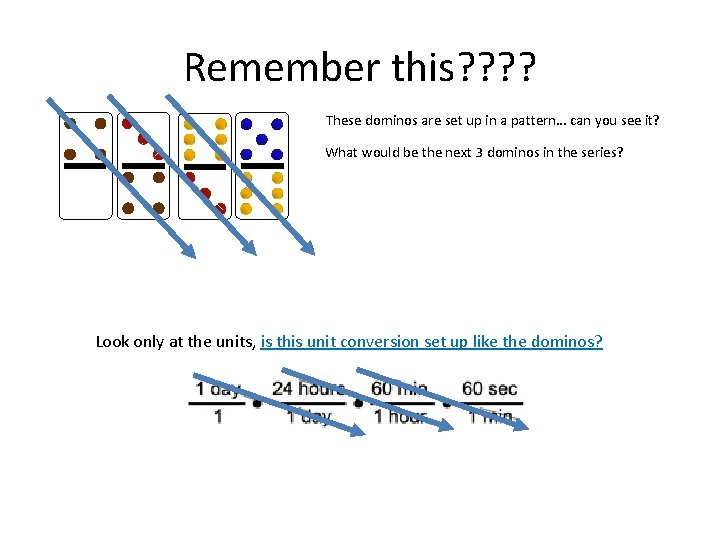
Remember this? ? These dominos are set up in a pattern… can you see it? What would be the next 3 dominos in the series? Look only at the units, is this unit conversion set up like the dominos?

Dimensional Analysisn Converting from a known unit to an unknown unit n 3 steps: 1. What do I know? (“starting” underline) 2. What do I want to know? (circle “destination”) 3. How do I get there? (*equivalence statements) Example: Sarah wants to know how many milliseconds are in 360 seconds? (equivalence statement*: 1000 milliseconds = 1 second)

Example Problem #1: You have 2. 00 grams of H 2 O, how many moles of H 2 O are present in 1 mole of this sample? Molar Mass of H 2 O = 18. 01 g 2. 00 grams H 2 O 1 1 moles H 2 O 18. 01 g/mol H 2 O

Example problem #2: • Given 4. 00 grams of nitrogen (N 2), how many moles of the compound are present? Molar Mass N 2 = 28. 00 g N 2 +2 H 2 2 NH 3 4 grams N 2 1 1 mole N 2 28. 02 grams of N 2

Calculations with Molar Mass Molar Ratio The molar ratio is made of the coefficients in front of the compounds in the balanced chemical equation. 12

Formula will use when converting from Moles of A to Moles of B: • What you see below is a basic example of the layout of this type of problem. Moles initially given in problem 1 Moles of what you want to “find” from balanced equation Moles of “given” from balanced equation This number may or may not be included in actual equation
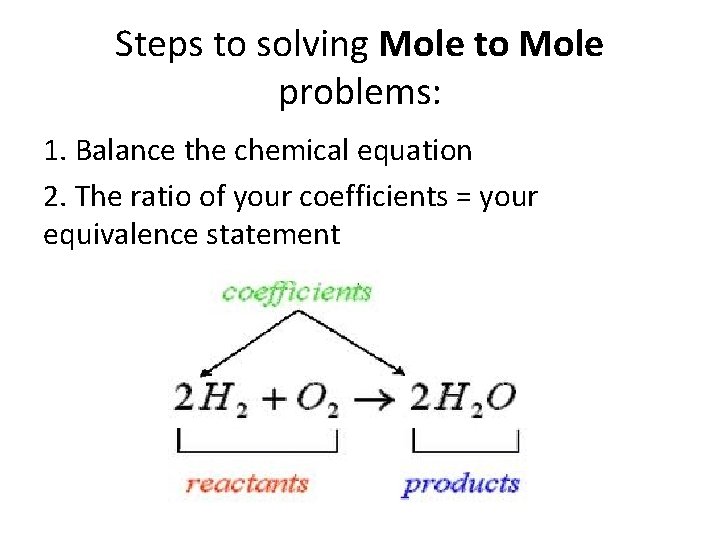
Steps to solving Mole to Mole problems: 1. Balance the chemical equation 2. The ratio of your coefficients = your equivalence statement

For every 1 mole of N 2 there are 3 moles of H 2

Example problem #1: 2 KCl. O 3 2 KCl + 3 O 2 How many moles of Oxygen are produced by the decomposition of 6 moles of KCl. O 3? 6 moles KCl. O 3 1 3 moles O 2 6 moles KCl. O 3

Example Problem #2: Zn + 2 HCl Zn. Cl 2 + H 2 How many moles of H 2 are produced from the reaction of 3 moles of Zn? 3 moles of Zn 1 mole of H 2 1 1 mole of Zn

Example Problem #3: C 3 H 8 + 5 O 2 3 CO 2 + 4 H 2 O How many moles of O 2 are necessary to react completely with 4 moles of C 3 H 8? 4 moles C 3 H 8 1 5 moles O 2 1 mole C 3 H 8
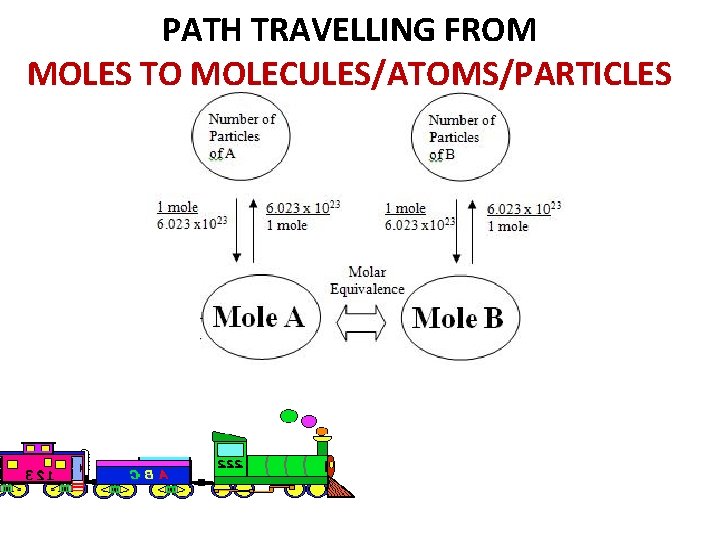
PATH TRAVELLING FROM MOLES TO MOLECULES/ATOMS/PARTICLES Molar Ratio 19

Moles initially given in problem Molecules of what you want to “find” from balanced equation 1 Moles of “given” from balanced equation This number may or may not be included in actual equation.

Example Problem #1: Convert 7. 50 mole of O 2 to molecules. 7. 50 moles O 2 1 6. 02 x 1023 moles O 2 1 mole O 2

Example Problem #2: Convert 3. 72 x 102 molecules to moles 3. 27 x 107 molecules 1 1 mole 6. 02 x 1023 molecules

Example Problem #3: How many molecules are in 3. 05 moles of NH 3? 3. 05 moles NH 3 1 6. 02 x 1023 molecules of NH 3 1 mole NH 3
- Slides: 23