The Modern Periodic Table Early Periodic Table Atomic

The Modern Periodic Table

Early Periodic Table – Atomic Number • In 1913 Henry Mosley discovered that each element contained a unique number of protons in the nuclei • Arranged elements in order of atomic number. • Resulted in a clear periodic pattern of properties.

Periodic Law • There is a periodic repetition of chemical and physical properties of elements when arranged in increasing atomic number (increasing number of protons) called the periodic law

Modern Periodic Table • • Organized in columns called groups or families Rows are called periods Group A – representative elements (1 A-8 A) Group B - transition elements (1 B-8 B) Representative Elements (Group A) Transition Elements (Group B)
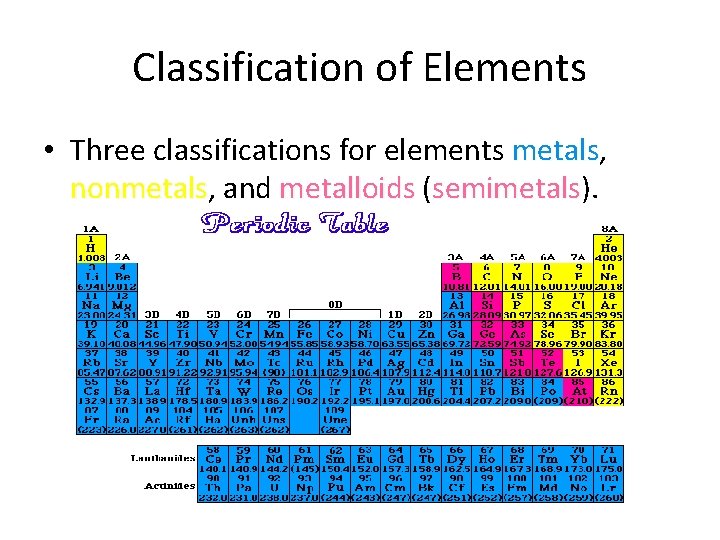
Classification of Elements • Three classifications for elements metals, metals nonmetals, nonmetals and metalloids (semimetals). semimetals

Metals • Properties of Metals – shiny, smooth, solids (except mercury) – Good conductors of heat and electricity – High densities – High melting and boiling points – Malleable – bended or pounded into sheets – Ductile – drawn into wires

Groups of Metals • Alkali metals – group 1 A except H • Alkaline earth metals – group 2 A – Alkali metals and alkaline earth metals are chemically reactive • Transition metals – group B elements • Inner transition metals – Lanthanide – Actinide
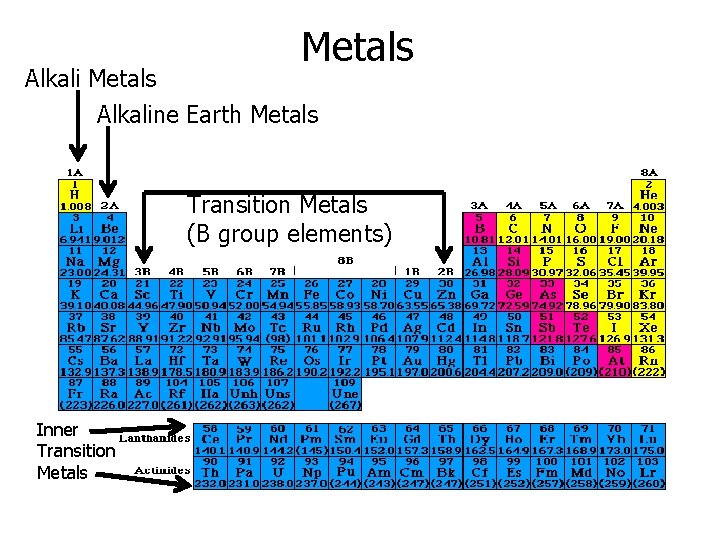
Alkali Metals Alkaline Earth Metals Transition Metals (B group elements) Inner Transition Metals

Organizing by Electron Configuration • Group number for group A elements represents the number of valence electrons • Atoms in the same group have similar chemical properties because they have the same number of valence electrons
![Alkali Metals Electron configurations for alkali metals Lithium 1 s 22 s 1 [He]2 Alkali Metals Electron configurations for alkali metals Lithium 1 s 22 s 1 [He]2](http://slidetodoc.com/presentation_image/0dce869495a211e528b57eab8e25ddb8/image-10.jpg)
Alkali Metals Electron configurations for alkali metals Lithium 1 s 22 s 1 [He]2 s 1 Sodium 1 s 22 p 63 s 1 [Ne]3 s 1 Potassium 1 s 22 p 63 s 23 p 64 s 1 [Ar]4 s 1 Rubidium 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 1[Kr]5 s 1 What do the four configurations have in common? • They have a single electron in their outermost energy level • They all have one valence electron, thus similar chemical properties • • •

Alkaline Earth Metals • Electron configuration for alkaline earth metals • • Beryllium Magnesium Calcium Strontium [He]2 s 2 [Ne]3 s 2 [Ar]4 s 2 [Kr]5 s 2 • All alkaline earth metals have two valence electrons, thus similar chemical properties.

Question Time What are representative elements? What are Group B elements called? What are three classifications for elements? What are the characteristics of metals? Where are the alkali metals and alkaline earth metals located? • Where are transition metals located? • What are the inner transition metals? • How many valence electrons do alkali metals and alkaline earth metals have? • • •

Nonmetals • Gases or brittle, dull looking solids • Poor conductors of heat and electricity • Usually have lower densities, melting point, and boiling point than metals. sulfur • Groups of nonmetals – Halogens 7 A – Noble gases 8 A
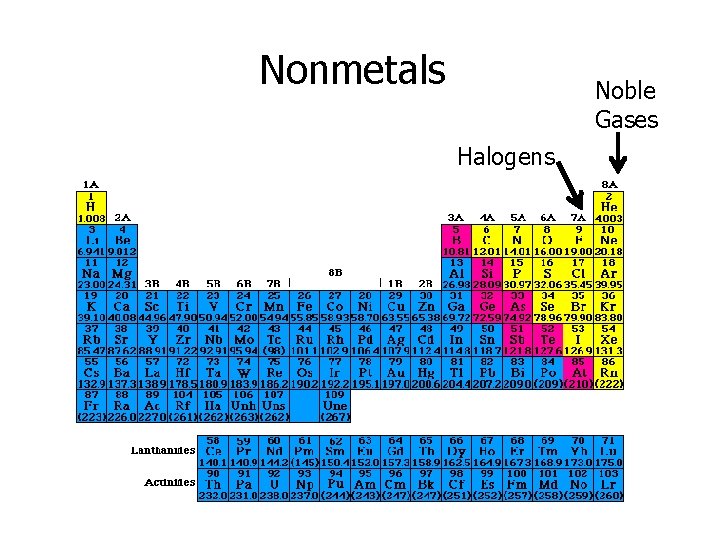
Nonmetals Noble Gases Halogens

Noble Gases • Noble gases – Group 8 A • Called inert gases because they rarely take part in a reaction He – 1 s 2 Ne – 1 s 22 p 6 Ar – 1 s 22 p 63 s 23 p 6 Kr - 1 s 22 p 63 s 23 p 63 d 104 s 24 p 6 • Because noble gases have completely filled s and p sublevels, they do not react with other elements

Metalloids (Semimetals) • Physical and chemical properties similar to both metals and nonmetals – They are metallic-looking brittle solids – Relatively good electrical conductivity. • Used in glasses, alloys, and semiconductors • The six elements commonly recognized as metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. Polonium and astatine are sometimes classified as metalloids
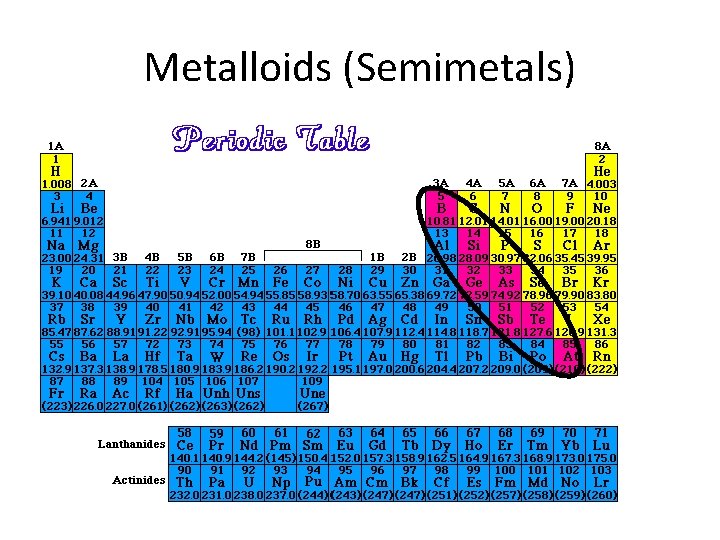
Metalloids (Semimetals)

Question Time What are the properties of nonmetals? Where are halogens and noble gases found? Why are noble gases unreactive (inert)? What are the properties of metalloids (semimetals)? • Which six elements are most commonly known as metalloids (semimetals)? • •
- Slides: 18