The Modern Periodic Table Early Periodic Table 1

The Modern Periodic Table

Early Periodic Table • 1 st Periodic table arranged by atomic weight; simplest arrangement but not the best • Mendeleev rearranged elements by reactivity to each other and noticed that the relationship between weight was periodic (characteristics repeated every 7 th element)

Periodic Law • When elements are arranged by atomic number (# of p+) there is a repetition of chemical and physical properties
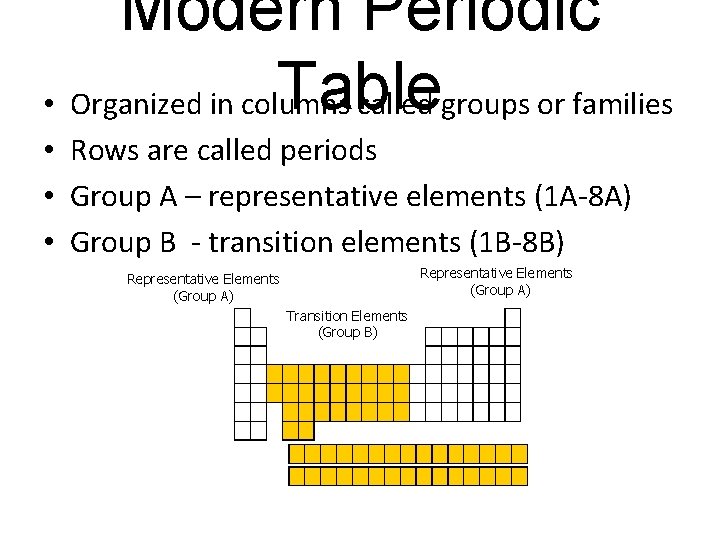
Modern Periodic Table • Organized in columns called groups or families • Rows are called periods • Group A – representative elements (1 A-8 A) • Group B - transition elements (1 B-8 B) Representative Elements (Group A) Transition Elements (Group B)

Classification of Elements • Three classifications for elements metals, metals nonmetals, nonmetals and metalloids (semimetals). semimetals

Metals • Properties – shiny, smooth, solids (except mercury) – Good conductors of heat and electricity – High densities, melting/boiling points – Malleable – bendt or pounded into sheets – Ductile – drawn into wires
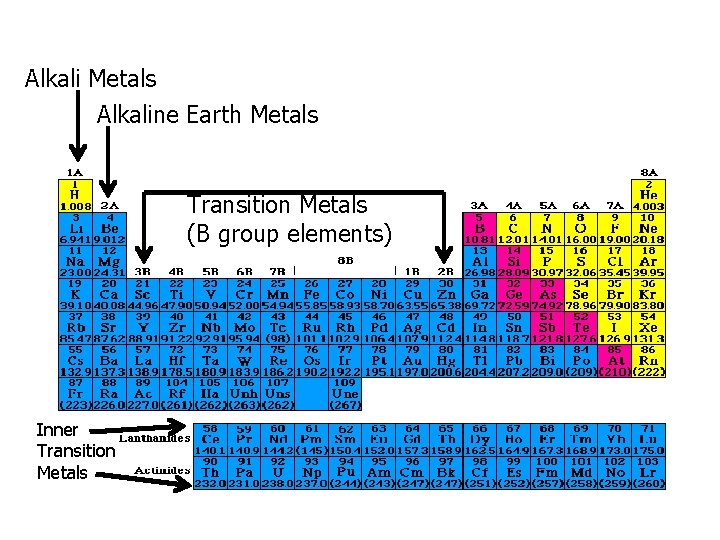
Alkali Metals Alkaline Earth Metals Transition Metals (B group elements) Inner Transition Metals

Nonmetals • Properties – Gases or brittle, dull looking solids – Poor conductors of heat and electricity – Usually have lower densities, melting point, and boiling point than metals sulfur
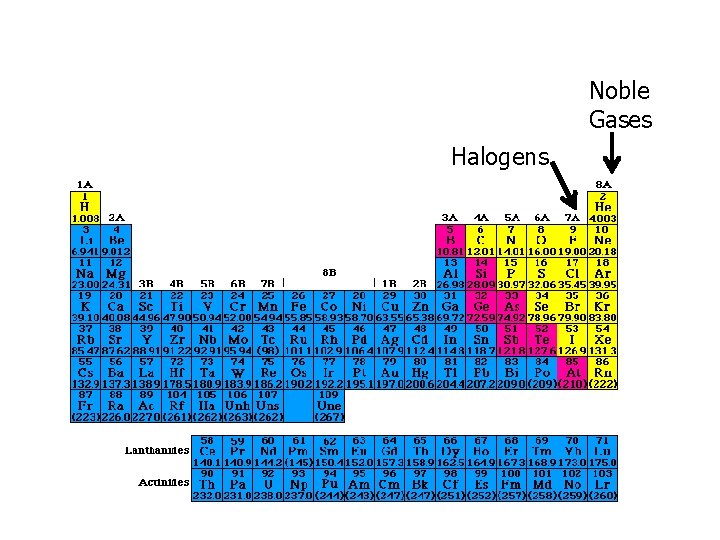
Noble Gases Halogens

Metalloids (Semimetals) • Physical and chemical properties similar to both metals and nonmetals – They are metallic-looking brittle solids – Relatively good electrical conductivity.

Electron Configurations for Alkali Metals 1 s 22 s 1 Lithium Sodium 1 s 22 p 63 s 1 Potassium 1 s 22 p 63 s 23 p 64 s 1 Rubidium 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 1 [He]2 s 1 [Ne]3 s 1 [Ar]4 s 1 [Kr]5 s 1

Electron Configurations for Alkaline Earth Metals Beryllium Magnesium Calcium Strontium 1 s 22 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 2 [He]2 s 2 [Ne]3 s 2 [Ar]4 s 2 [Kr]5 s 2

Electron Configuration for Noble Gases He – 1 s 2 Ne – 1 s 22 p 6 Ar – 1 s 22 p 63 s 23 p 6 Kr - 1 s 22 p 63 s 23 p 63 d 104 s 24 p 6
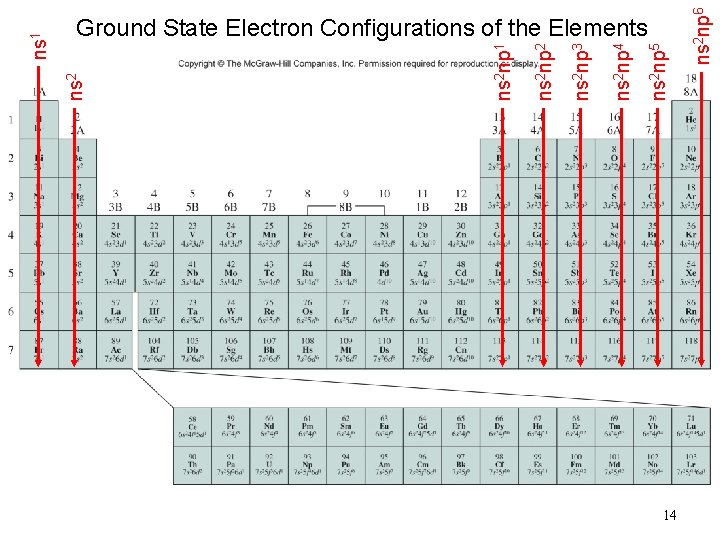
14 ns 2 np 6 ns 2 np 5 ns 2 np 4 ns 2 np 3 ns 2 np 2 ns 2 np 1 ns 2 ns 1 Ground State Electron Configurations of the Elements

• Valence Electrons in the outermost s and p orbitals (highest n shell) • These electrons participate in chemical reactions

Reactivity of Elements Atoms in the same group have similar chemical properties because they have the same number of valence electrons

Octet Rule Atoms gain, lose, or share electrons to acquire a full set of eight valence electrons (to be like a noble gas) Eight is great!!!
- Slides: 17