The Model of the Atom II The Atom






















- Slides: 36

The Model of the Atom II

The Atom Review • Dalton’s Atomic Theory • All matter made up of atoms • Atoms are indivisible • Atoms of same element are exactly alike Dalton Was Wrong—But Not His Fault

The Atom Review • Dalton’s Atomic Theory • Atoms ARE made of smaller particles • Neutrons – Discovery of isotopes Carbon-12, Carbon-14 • Protons – Rutherford’s Gold Foil experiment • Electrons – Thomson’s cathode ray experiment

The Atom Electrons Most Important • Why? • Responsible for most chemical and physical properties • Responsible for chemical bonding – Covalent (sharing) & Ionic (transferring) • Understanding electron arrangement allows for understanding of chemical reactions

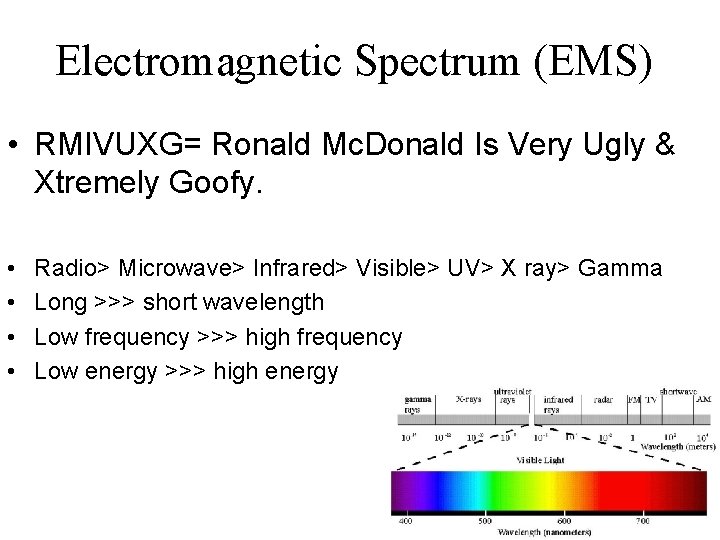
Electromagnetic Spectrum (EMS) • RMIVUXG= Ronald Mc. Donald Is Very Ugly & Xtremely Goofy. • • Radio> Microwave> Infrared> Visible> UV> X ray> Gamma Long >>> short wavelength Low frequency >>> high frequency Low energy >>> high energy

Electromagnetic Spectrum (EMS) • Visible = ROYGBIV • Red>Orange>Yellow>Green>Blue>Indigo>Violet • Every element emits a different set of wavelengths, we can thus ID an element by its EMS.

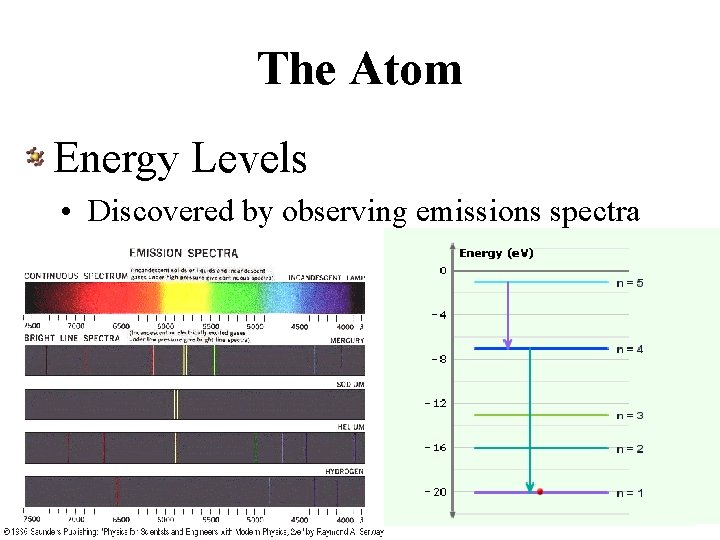
The Atom Energy Levels • Discovered by observing emissions spectra

The Atom Energy Sublevels • Scientists Seeing Unexpected Results With Emissions Spectra • Hypothetical: only 3 energy levels, how many possible colors could be seen? 3 2 1 3

The Atom Energy Sublevels • Scientists Seeing Unexpected Results With Emissions Spectra • They were seeing a lot more spectral lines than what they expected • Only explanation: sublevels 3 2 1

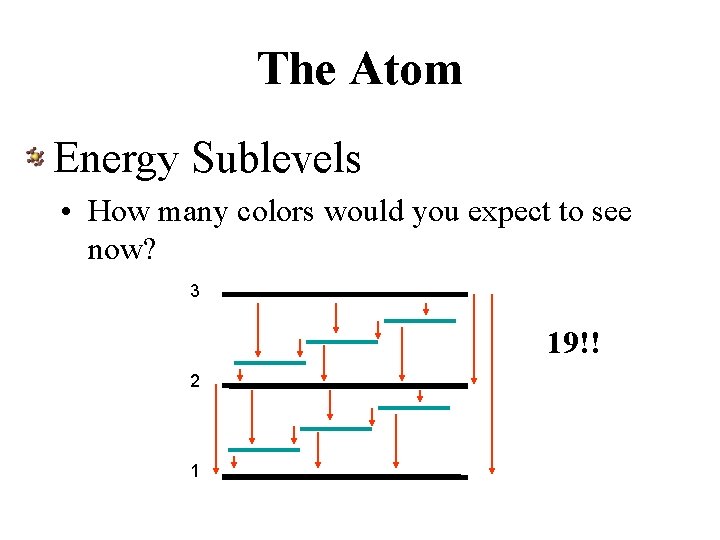
The Atom Energy Sublevels • How many colors would you expect to see now? 3 19!! 2 1

The Atom Energy Sublevels • Given Abbreviations • From lowest energy to highest energy: – s, p, d, f • Each energy level has a corresponding number of sublevels Energy Level 1 1 Sublevel , 1 s Energy Level 2 2 Sublevels, 2 p Energy Level 3 3 Sublevels, 3 p, 3 d Energy Level 4 4 Sublevels, 4 p, 4 d, 4 f

The Atom Energy Sublevels • Each sublevel has a specific number of orbitals: s p d f 2 (1 orbital) 6 (3 orbitals) 10 (5 orbitals) 14 (7 orbitals)

The Atom Energy Sublevels • Each orbital can hold only TWO electrons: s p d f 2 (1 orbital-2 total electrons) 6 (3 orbitals-6 total electrons) 10 (5 orbitals-10 total electrons) 14 (7 orbitals-14 total electrons)

The Atom Energy Levels • Each energy level then has a set number of sublevels and maximum electrons: 1 – only s 2 electrons max 2 - s and p sublevels, 8 electrons max 3 - s, p, and d sublevels, 18 electrons max 4 - s, p, d, and f sublevels, 32 electrons max

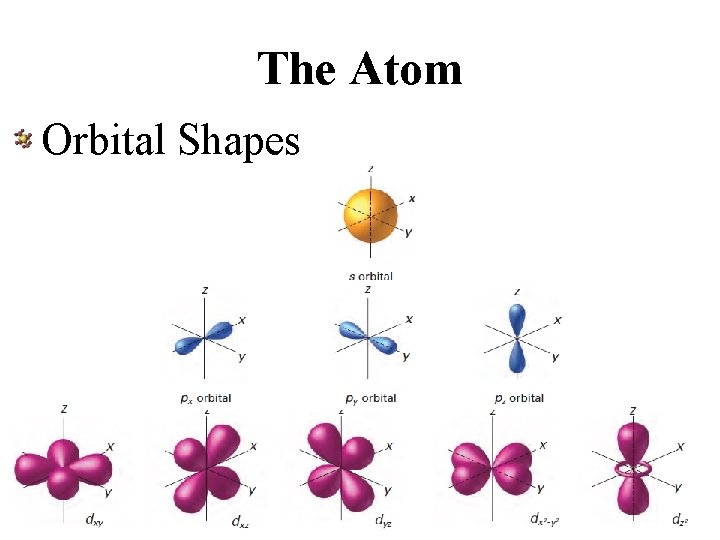
The Atom Electron Orbitals • Regions in space where electrons are likely to be found • Follows electron cloud model – Electrons do not travel in a specific orbit • Each orbital holds maximum of 2 electrons • Electron configuration – When all electrons are in most “comfortable” orbital and energy level


The Atom Orbital Shapes

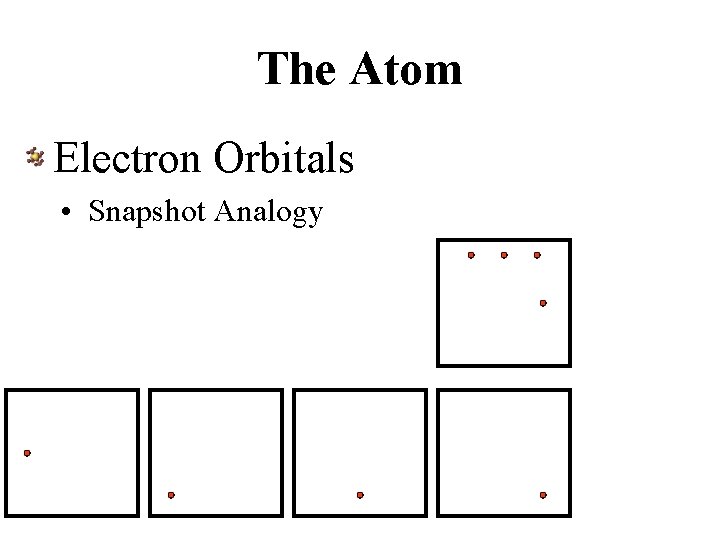
The Atom Electron Orbitals • Snapshot Analogy

The Atom Electron Orbitals • Snapshot Analogy

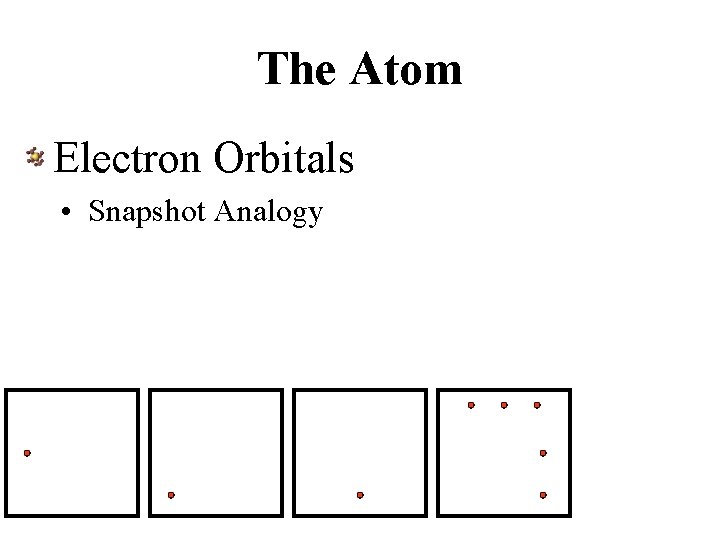
The Atom Electron Orbitals • Snapshot Analogy

The Atom Electron Orbitals • Snapshot Analogy

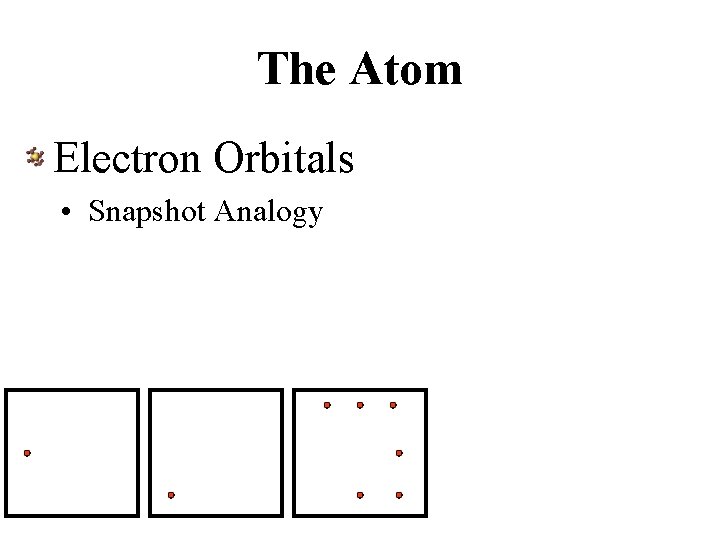
The Atom Electron Orbitals • Snapshot Analogy

The Atom Electron Orbitals • Snapshot Analogy

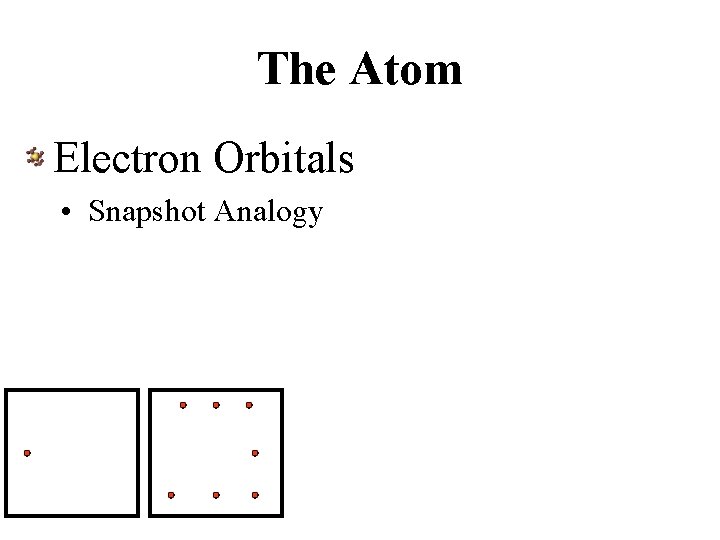
The Atom Electron Orbitals • Snapshot Analogy

The Atom Electron Orbitals • Snapshot Analogy

The Atom Electron Orbitals • Snapshot Analogy Orbital Shape

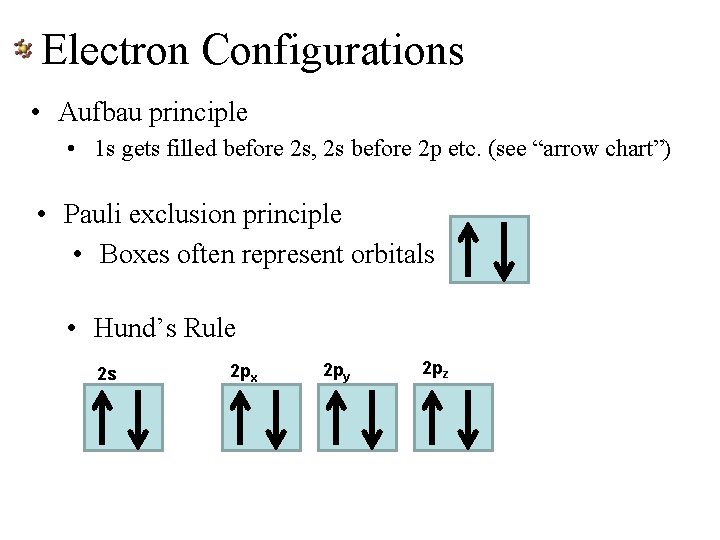
The Atom Electron Configurations • Rules to tell you in what order the orbitals get filled • Aufbau principle • LOWEST energy orbitals get filled FIRST • Pauli exclusion principle • Each orbital can only hold TWO electrons— each with opposite “spin” • Hund’s Rule • Each orbital must have ONE electron before any has TWO

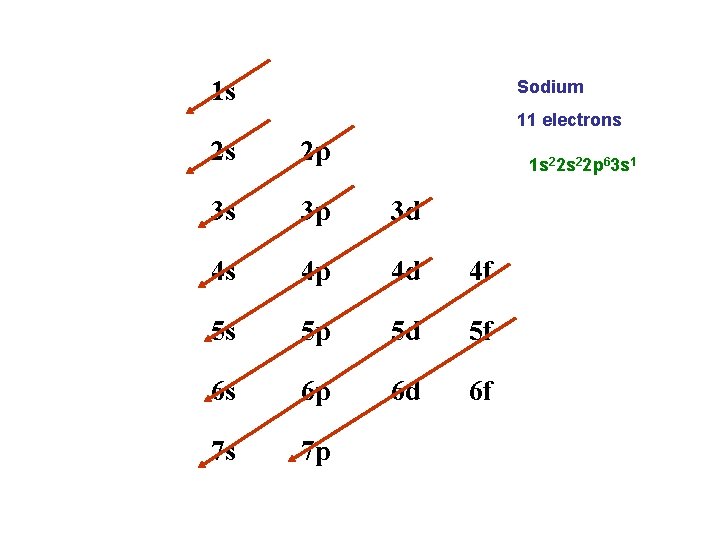
Electron Configurations • Aufbau principle • 1 s gets filled before 2 s, 2 s before 2 p etc. (see “arrow chart”) • Pauli exclusion principle • Boxes often represent orbitals • Hund’s Rule 2 s 2 px 2 py 2 pz

Writing Electron Configurations • The electron configuration for H is 1 s 1 • The e- configuration for He is 1 s 2 • Li = 1 s 22 s 1 • Na = 1 s 22 p 63 s 1 • Notice that the superscripts should add up to the total number of electrons!

1 s Sodium 11 electrons 2 s 2 p 3 s 3 p 3 d 4 s 4 p 4 d 4 f 5 s 5 p 5 d 5 f 6 s 6 p 6 d 6 f 7 s 7 p 1 s 22 p 63 s 1

Electron Configurations EXCEPTIONS • Some elements do NOT follow all the rules • Copper • Expected: Cu- 1 s 22 p 63 s 23 p 64 s 23 d 9 4 s 3 dxy 3 dxz 3 dyz 3 dx 2 -y 1 3 dz 2

Electron Configurations EXCEPTIONS • Some elements do NOT follow all the rules • Copper • Expected: Cu- 1 s 22 p 63 s 23 p 64 s 23 d 9 • Actual: Cu- 1 s 22 p 63 s 23 p 64 s 13 d 10 4 s 3 dxy 3 dxz 3 dyz 3 dx 2 -y 1 3 dz 2 Partially-filled 3 d sublevel, with a filled 4 s is less stable than a filled 3 d and a half-filled 4 s

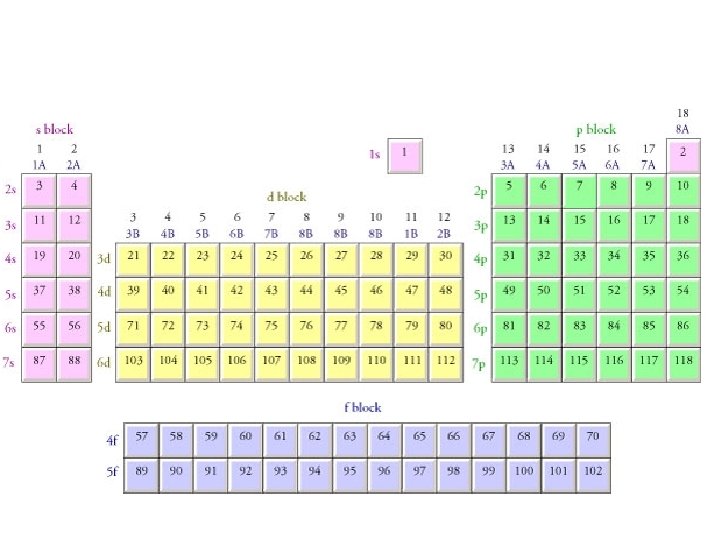
Kernel Structures Electron Configurations • Can get very long as you go down the PT • Kernel structures are used as abbreviations • The “kernel” is ALWAYS a Noble Gas (group 18) • They represent all inner electrons

Kernel Structures Electron Configurations Iodine 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 5 Tellurium 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 4 Antimony 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 3 Tin 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 2

Kernel Structures Electron Configurations Iodine 1 s 22 p 63 s 23 p 64 s 23 d 104 p 65 s 24 d 105 p 5 Find the noble gas that comes BEFORE the element Krypton [Kr]5 s 24 d 105 p 5

The End