The Metric System The Metric System The metric












- Slides: 26

The Metric System

The Metric System • The metric system (International System of Units) or SI System, is a universal system of measurement used by scientists all over the world. – Based on multiples of 10 • Base Unit = 1 – Length = meter (1. 0 m) – Mass = gram (1. 0 g) – Volume = liter (1. 0 L)

Prefixes and Suffixes • Many words can be broken down into smaller parts that can help you to better understand the meaning of the word. – Prefixes – terms attached to the beginning of a word. • Preheat – Pre means before » To heat the oven before you cook something. – Suffixes – terms attached to the end of a word. • Laryngitis – itis means inflammation » A swelling or inflammation of the larynx or vocal cords.

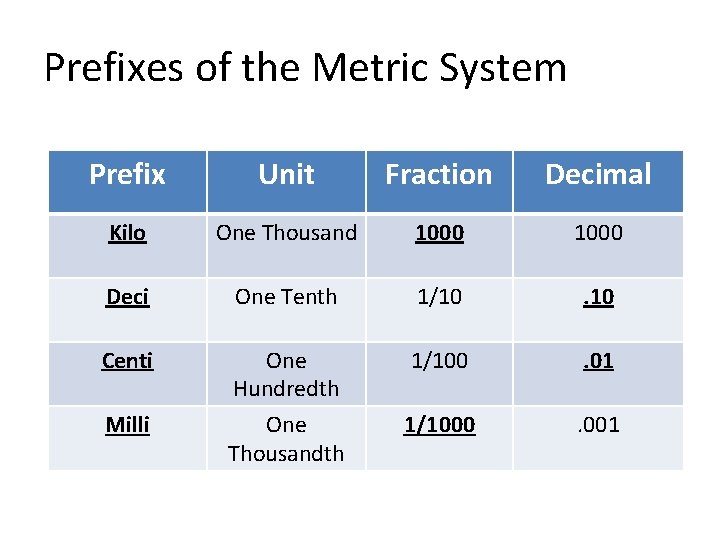
Prefixes of the Metric System Prefix Unit Fraction Decimal Kilo One Thousand 1000 Deci One Tenth 1/10 . 10 Centi One Hundredth 1/100 . 01 Milli One Thousandth 1/1000 . 001

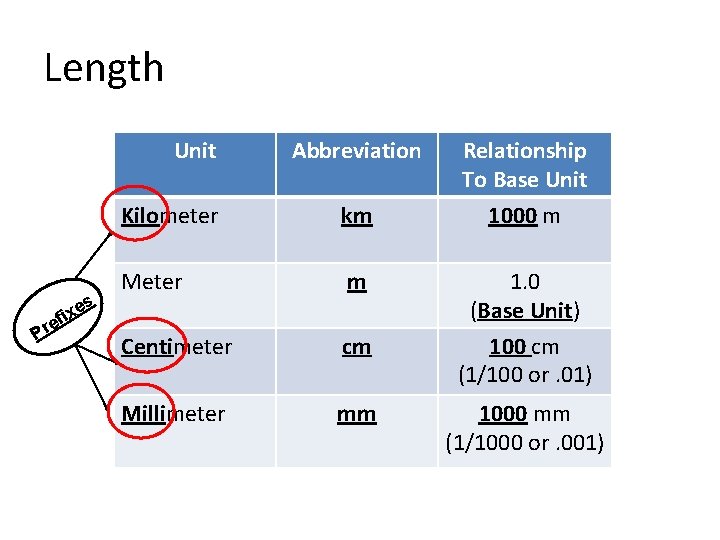
Length Unit Pr es x i ef Abbreviation Kilometer km Meter m Centimeter cm Millimeter mm Relationship To Base Unit 1000 m 1. 0 (Base Unit) 100 cm (1/100 or. 01) 1000 mm (1/1000 or. 001)

Length • Length - How long something is. – Instrument = metric ruler – Base Unit = Meter (m) (Base Unit) (. 1 or 1/10) (. 01 or 1/100) (. 001 or 1/1000)

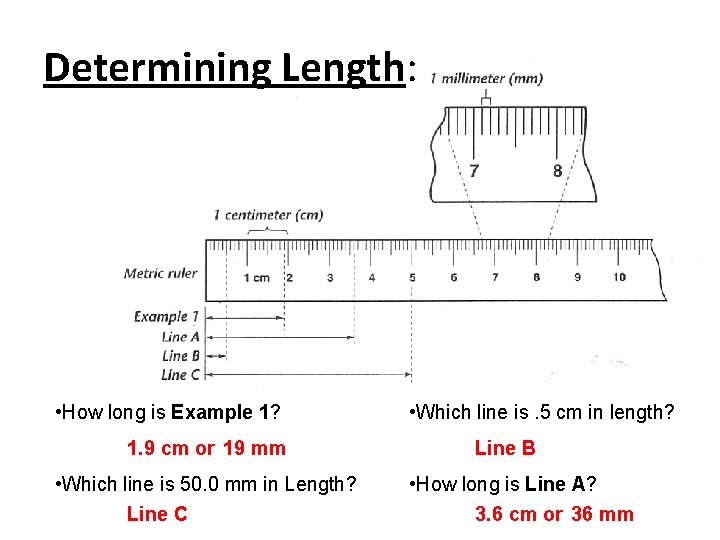
Determining Length: • How long is Example 1? 1. 9 cm or 19 mm • Which line is 50. 0 mm in Length? Line C • Which line is. 5 cm in length? Line B • How long is Line A? 3. 6 cm or 36 mm

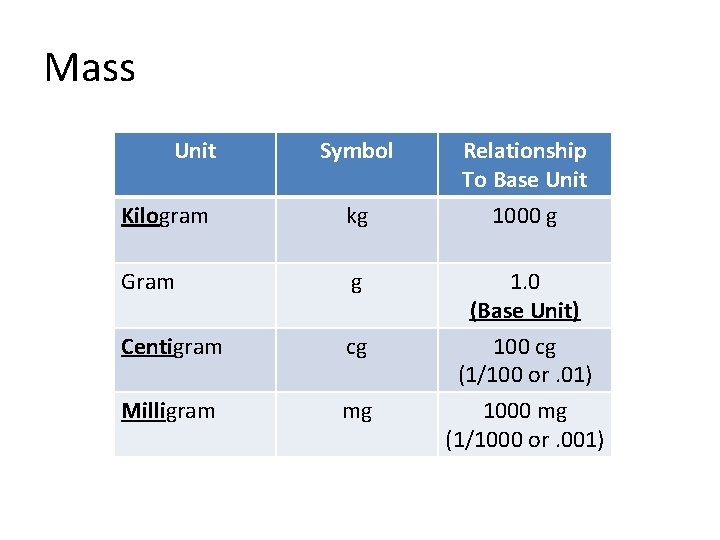
Mass Unit Symbol Kilogram kg Gram g Centigram cg Milligram mg Relationship To Base Unit 1000 g 1. 0 (Base Unit) 100 cg (1/100 or. 01) 1000 mg (1/1000 or. 001)

Mass • Mass - The amount of matter in an object – Instrument = Triple Beam Balance – Base Unit = Gram (g)

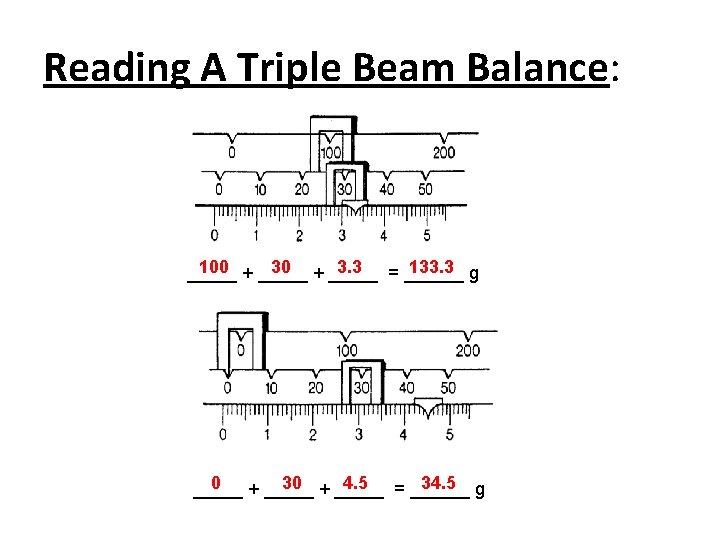
Reading A Triple Beam Balance: Start with the largest beam, 100 s, then add 10 s and finish with the smallest beam, 1 s. Solve the Problem: 200 + _____ 70 + _____ 6. 5 = ______ 276. 5 g _____

Reading A Triple Beam Balance: 100 + _____ 3. 3 = ______ 133. 3 g _____ 0 30 + _____ 4. 5 = ______ 34. 5 g _____ + _____

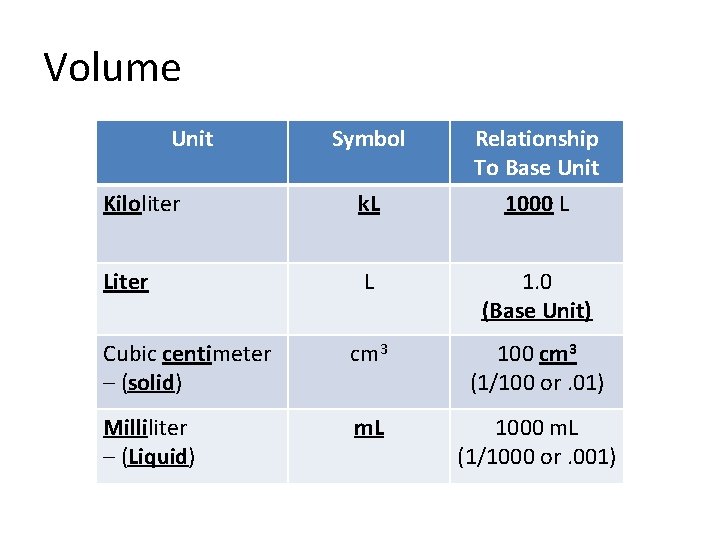
Volume Unit Symbol Relationship To Base Unit 1000 L Kiloliter k. L Liter L 1. 0 (Base Unit) Cubic centimeter – (solid) cm 3 100 cm 3 (1/100 or. 01) Milliliter – (Liquid) m. L 1000 m. L (1/1000 or. 001)

Volume • Volume - The amount of space an object takes up. • 2 Types of Volume measurements: – Solid Volume • An object that has regular measurable sides. – Liquid Volume • An object that is a liquid or has irregular, nonmeasurable sides.

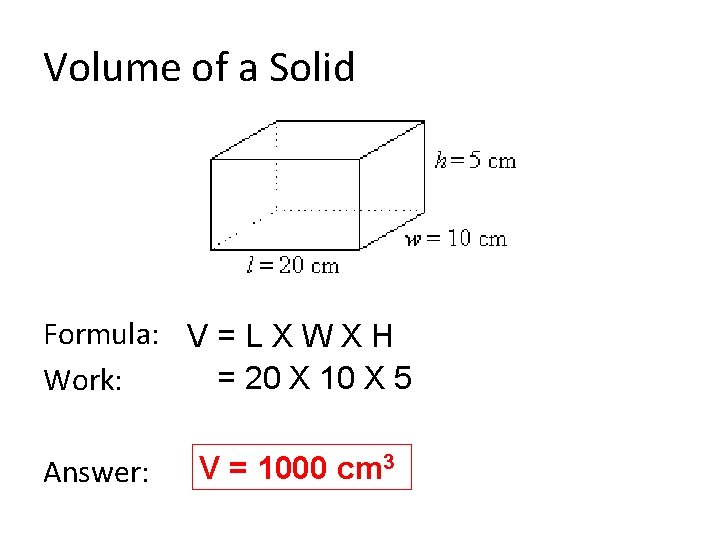
Solid Volume • Instrument – metric ruler • Base Unit – cubic meters (m 3) • Formula: Volume = Length X Width X Height • There are 3 measurements so you must have a superscript of 3

Volume of a Solid Formula: V = L X W X H = 20 X 10 X 5 Work: Answer: V = 1000 cm 3

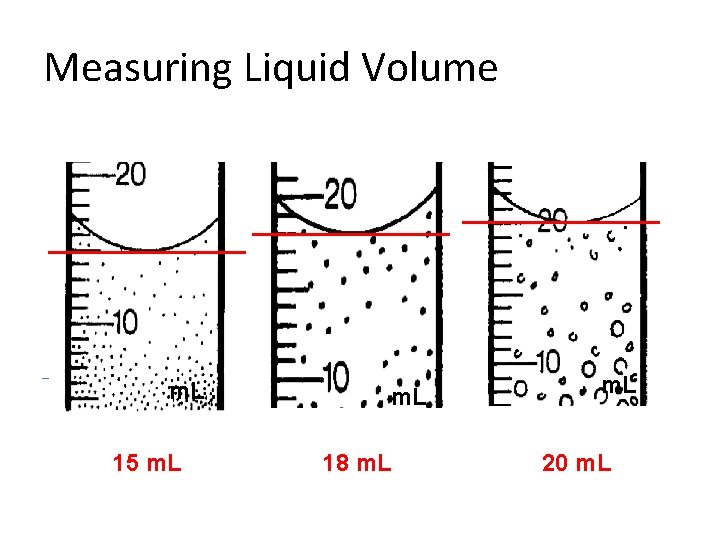
Liquid Volume • Instrument – Graduated Cylinder • Base Unit – liter (L) • Reading a Graduated Cylinder: – Always take the reading from eye level! • Water in a container has a curved surface or meniscus. – Take the reading from the bottom of the meniscus.

Measuring Liquid Volume m. L 15 m. L 18 m. L 20 m. L

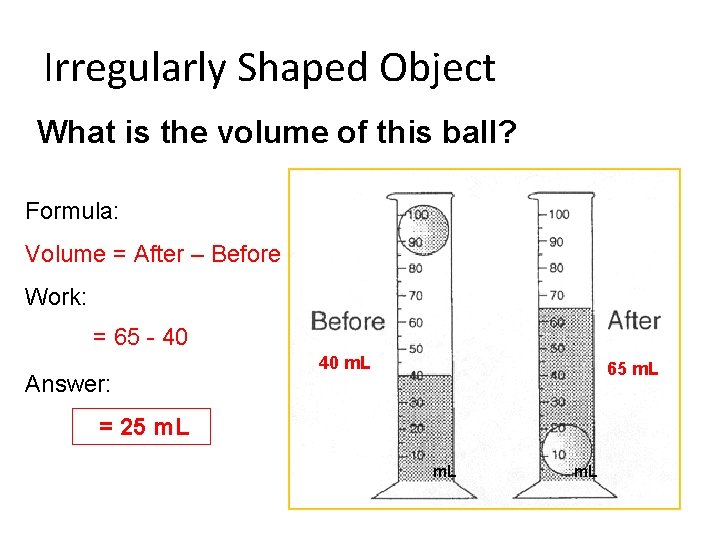
Irregularly Shaped Object What is the volume of this ball? Formula: Volume = After – Before Work: = 65 - 40 Answer: 40 m. L 65 m. L = 25 m. L

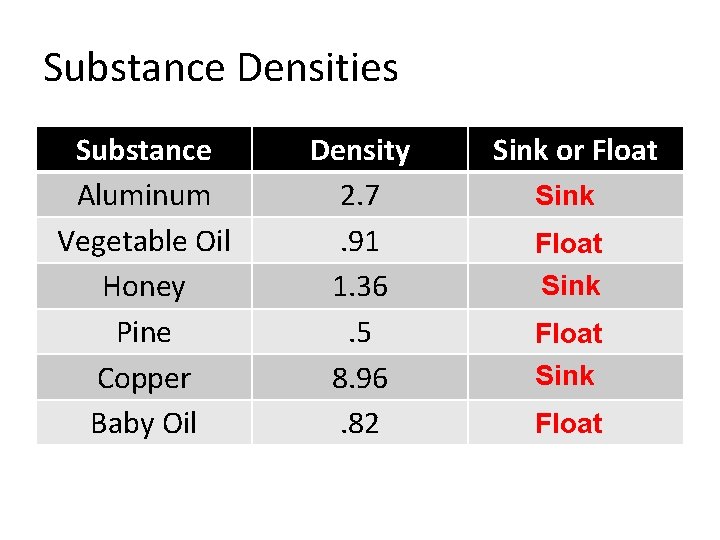
Density • Density - The measurement of how much mass is contained in a given volume of an object. – Density is unique to each individual substance on Earth. • Water = 1 g/cm 3 or 1 g/m. L – Used to compare all other substances to: » Anything greater than 1 g/cm 3 will sink » Anything less than 1 g/cm 3 will float Formula: Density = Mass Volume

Substance Densities Substance Aluminum Vegetable Oil Honey Pine Copper Baby Oil Density 2. 7. 91 1. 36. 5 8. 96. 82 Sink or Float Sink Float

Finding Solid Density: (D = M/V) 1. A block of metal has a volume of 15 cm 3 and has a mass of 10 g. What is its density? Formula: Density = M/V Work: = 10 g / 15 cm 3 Answer: Density =. 6 g/cm 3 Would this substance sink or float? Float, less dense than water

Finding Solid Density: (D = M/V) 2. A block of metal measures 4 cm X 4 cm and has a mass of 75 g. What is its density? Volume = l x w x h =4 X 4 X 4 = 64 cm 3 4 cm 4 cm Formula: Density = M/V Work: = 75 g / 64 cm 3 Answer: 3 = 1. 2 g/cm Would this substance sink or float? Sink, more dense Than water

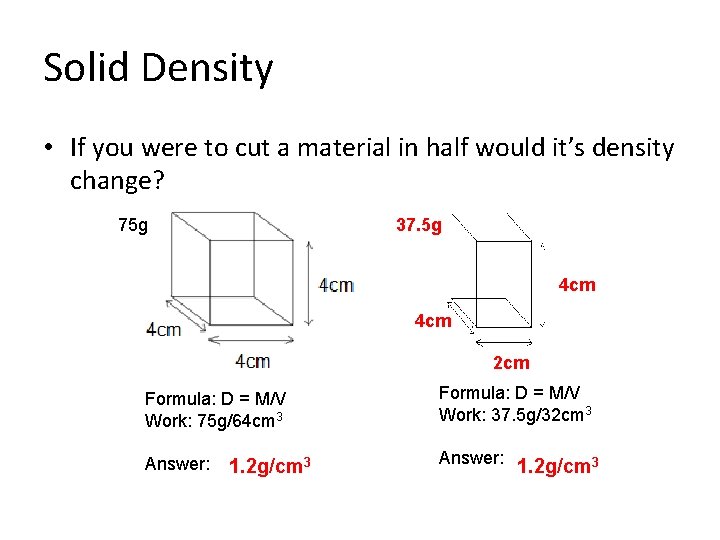
Solid Density • If you were to cut a material in half would it’s density change? 75 g 37. 5 g 4 cm 2 cm Formula: D = M/V Work: 75 g/64 cm 3 Formula: D = M/V Work: 37. 5 g/32 cm 3 Answer: 1. 2 g/cm 3

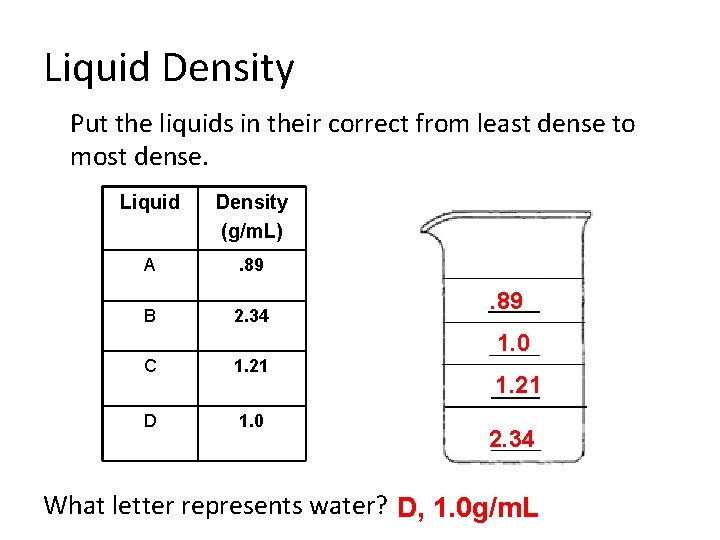
Liquid Density Put the liquids in their correct from least dense to most dense. Liquid Density (g/m. L) A . 89 B 2. 34 C 1. 21 D 1. 0 . 89 1. 0 1. 21 2. 34 What letter represents water? D, 1. 0 g/m. L

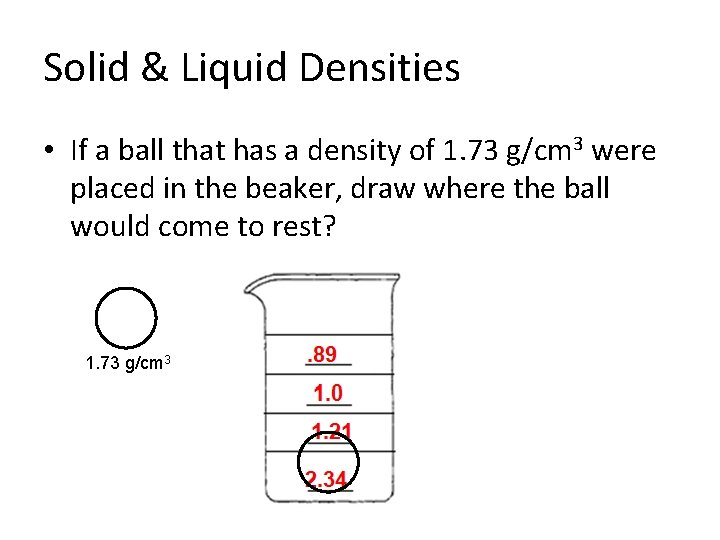
Solid & Liquid Densities • If a ball that has a density of 1. 73 g/cm 3 were placed in the beaker, draw where the ball would come to rest? 1. 73 g/cm 3

Solid & Liquid Densities • If a block of wood has a density of. 7 g/cm 3 where would the block of wood be located in this beaker of water? . 7 g/cm 3