The Metabolism of XENOBIOTICS Reported by Group III

The Metabolism of XENOBIOTICS Reported by Group III, 1 -C 2

What are Xenobiotics? • Amay be naturalthat or synthetic foreign to the body. compound is “stranger” • may be harmful or safe EXAMPLES OF CARCINOGENIC XENOBIOTICS • Food components methyl glyoxal (coffee) Xenoaflatoxin • Food contaminants B 1 = • Cigarette smoke • Industrial (occupational)dibromoethane • Industrial (effluent) vinyl chloride • Cyanide is one example which is toxic at very low levels

Principal Classes of Xenobiotics Drugs Chemical carcinogens PCB’s Insecticides Most synthetic materials There are 200, 000 of these

How does the body handle them? For convenience, the metabolism of xenobiotics is divided into 2 phases There approximately 30 different enzymes that catalyze xenobiotic compounds

1. Phase 1 • Most common reaction: hydroxylation 2 phases • Some enzymes: monooxygenases microsomal cytochrome-P 450 s 2. Phase 2 • Most common reactions: conjugation methylation • Enzymes: transferases

Phase 1 Mainly hydroxylation reactions Enzyme: Microsomal Cytochromes P 450 s Purpose: 1. Make the toxin more water-soluble 2. Sometimes deactivates the toxin

Phase 1: Microsomal cytochrome P 450 s n n n Large number of isoforms All contains heme Has a special nomenclature: CYP Family 2 CYPs with >40% similarity Subfamily C CYPs with >55% similarity Specific 9 # CYPs with >100% (exact) similarity
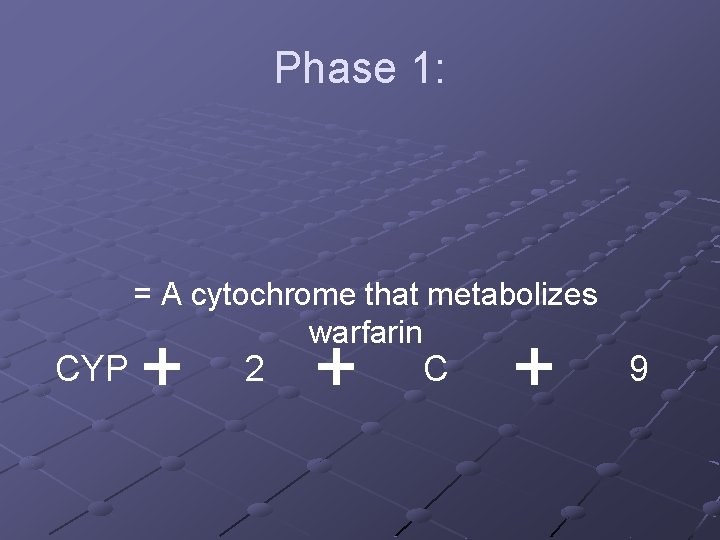
Phase 1: CYP = A cytochrome that metabolizes warfarin 2 C 9

Phase 1: Microsomal cytochrome P 450 s n n n n n Large number of isoforms All contains heme Has a special nomenclature: Plenty in liver (in the SER) Differs from the mitochondrial cytochrome P 450 Has lipid components (primarily lecithin) Inducible (CYP 2 C 9) Some exhibit polymorphism Rarely, some contribute to cancer formation

Phase 1: n Inducible (CYP 2 C 9) Microsomal cytochrome P 450 s n n n n n Large number of isoforms All contains heme Has a special nomenclature: Plenty in liver (in the SER) Differs from the mitochondrial cytochrome P 450 Has lipid components (primarily lecithin) Inducible (CYP 2 C 9) Some exhibit polymorphism Rarely, some contribute to cancer formation

Phase 1: Inducible (CYP 2 C 9) n Q: If you give phenobarbital to a patient who is dependent on warfarin, you have to adjust (higher) the dose of the latter or risk bleeding. Why? A: CYP 2 C 9 , which metabolizes (inactivates) warfarin is induced by phenobarbital. Q: Drinking along with smoking increases risk of cancer than smoking all by itself. Why? A: CYP 2 E 1, which is induced by ethanol (in liquor) is one of the cytochromes that contribute to the activation of procarcinogens found in tobacco smoke.

Phase 1: Microsomal cytochrome P 450 s n n n n n Large number of isoforms All contains heme Has a special nomenclature: Plenty in liver (in the SER) Differs from the mitochondrial cytochrome P 450 Has lipid components (primarily lecithin) Inducible (CYP 2 C 9) Some exhibit polymorphism Rarely, some contribute to cancer formation

Phase 1: CYP 1 A 1, CYP 2 E 1: ØAn isoform of cytochrome P 450 metabolize inactive PAHs (polycyclic aromatic hydrocarbons) into active carcinogens ØPAHs are abundant in cigarette smoke ØSmokers have increased levels of CYP 1 A 1 in their cells than non-smokers. ØCYP 2 E 1 is induced by ethanol. n Rarely, some contribute to cancer formation

Phase 1: Microsomal cytochrome P 450 s n n n n n Large number of isoforms All contains heme Has a special nomenclature: Plenty in liver (in the SER) Differs from the mitochondrial cytochrome P 450 Has lipid components (primarily lecithin) Inducible (CYP 2 C 9) Some exhibit polymorphism Rarely, some contribute to cancer formation

Phase 1: CYP 2 D 6 ØCYP 2 D 6 is involved in the metabolism of debrisoquin (antihypertensive drug) and sparteine (antiarrhythmic and oxytocic drug) ØPolymorphisms (many different forms of CYP 2 D 6 in the same medium) contribute to the lower the overall activity of the enzyme. ØThis is because some of the “variant forms” have low catalytic activity which pulls the overall activity down. n ØThe poor catalysis of debrisoquin and sparteine allows them to stay and accumulate in the body and cause toxicity. Some exhibit polymorphism ØPolymorphisms and differences between enzyme structure between individuals is genetic

Phase 2 Conjugation reactions Enzyme: Transferases Purpose: Make the toxin further watersoluble for excretion
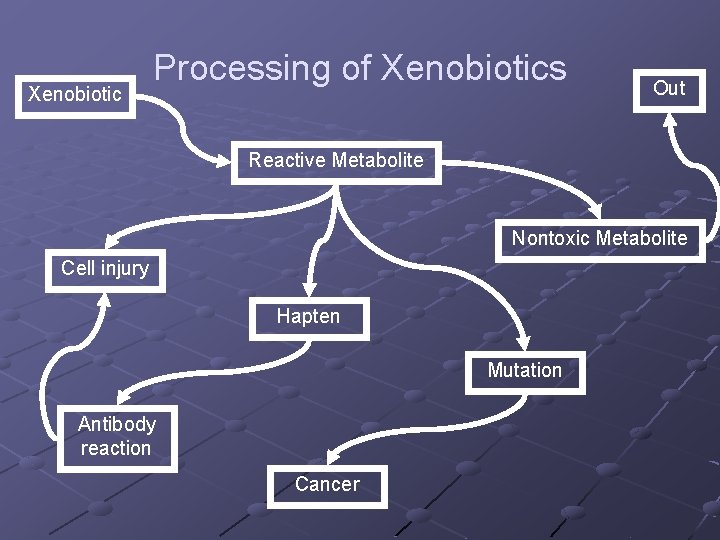
Xenobiotic Processing of Xenobiotics Out Reactive Metabolite Nontoxic Metabolite Cell injury Hapten Mutation Antibody reaction Cancer

Phase 2: Conjugation Rxn Donor Examples of Xenobiotic Targets Glucoronidation Glucoronosyl(most frequently transferases used by the body) UDPglucoronic acid 2 -acetylaminoflourene, benzoic acid, aniline, phenols Sulfation --- PAPS (active sulfate) Alcohols, arylamines, phenols Conj. w/ Glutathione --- G-SH form Electrophilic xenobiotics (R) R + G-SH R-S-G Methylation Acetylation Enzyme Methyltransferases SAM Acetyl-Co. A isoniazid
- Slides: 18