The Medicare Part D Prescription Drug Benefit Understanding

The Medicare Part D Prescription Drug Benefit Understanding the Formulary Requirements and Related Implications Michael Sharp, R. Ph, Pharmacy Consultant Office of Medicaid Policy and Planning, State of Indiana Michael. Sharp@fssa. in. gov

Areas of Focus Basic benefit principles, fundamental formulary requirements and CMS review processes n Exceptions/Appeals overview n Formulary implications for dual-eligibles and the Indiana Medicaid approach n Implementation considerations, timeline and recommended resources n

Medicare Coverages Part A Hospital insurance for inpatient stays, some skilled nursing facility care, hospice care and home health care Part B Medical insurance for physician services, outpatient hospital care, durable medical equipment, some medical supplies and selected drugs Medicare Advantage (MA-PD) for benefits through private health plans – old Medicare+Choice Prescription drug benefit for persons eligible for Part A or enrolled in Part B Part C Part D

Medicare Prescription Drug Benefit, 2006 and Beyond n Beginning in 2006, beneficiaries have choice of: n n n Traditional Medicare, with access to private drug-only plans (PDPs) Medicare Advantage (MA-PD) plans for Medicare benefits and Rx drugs New plans provide “standard” prescription drug benefit or its “actuarial equivalent” n Plans have some flexibility to determine which drugs are covered and cost-sharing requirements, subject to certain constraints n Premium and cost-sharing subsidies for low-income beneficiaries with incomes up to 150% poverty and modest assets n Medicaid will no longer pay for Medicare D covered drugs after December 31, 2005

Medicare Prescription Drug Plans n n n Must offer basic drug benefit n Standard benefit May offer supplemental benefits n Alternative Benefit n Enhanced benefit Can be flexible in benefit design n May look different than standard benefit n May have different co-pay or co-insurance n Cannot change actuarial equivalence

Part D Sponsors – Risk-Bearing Entities n n Prescription Drug Plans (PDPs) n Pharmacy Benefit Managers n Private Insurance Companies Medicare Advantage-Prescription Drug (MA-PDs) n Must offer at least 1 option for Rx coverage n May offer plans with no drug coverage for beneficiaries who decline Part D coverage n May offer Special Needs Plans, focusing on Duals & selected diagnoses

Formulary Coverage Fundamentals n CMS says “clinically appropriate medications, at lowest possible cost” n Formularies must not discriminate against: n Individuals with HIV/AIDS, mental health and other cognitive disorders n The Dual eligibles n CMS utilizes the USP formulary classification model as the minimum benchmark formulary appropriateness n USP model consists of 146 therapeutic classifications and related pharmacologic categories n Plans must accommodate all medically necessary medications at all levels of care

Medicare Prescription Covered Drugs Prescription drugs, biologicals and insulin n Medical supplies associated with injection of insulin (syringes/swabs/etc) n Cases where a drug is not FDA approved for an indication but it has clinical literature to support its use n Vaccines not covered by Part B n Viagra, Levitra and Cialis n Brand name and generic drugs will be included in each formulary* *Less for generics or preferred Rx, more for brands. Multisource brand name products can be excluded. n

Formulary Requirements n n Plan formulary must be developed by a Pharmacy and Therapeutics Committee Formulary must include at least 2 drugs in each therapeutic category and pharmacologic class of covered Part D drugs and in certain categories, must contain “all or substantially all” of the following medications: • Antidepressants • Antipsychotics • Anticonvulsants • Antiretrovirals • Antineoplastics • Immunosuppressants

Part D Drug Exclusions Drugs for n Anorexia, weight loss, or weight gain n Fertility n Cosmetic purposes or hair growth n Symptomatic relief of cough and colds n Prescription vitamins and mineral products n Except prenatal vitamins and fluoride preparations n Non-prescription (OTC) drugs*, with the exception of OTC insulin n Barbiturates n Benzodiazepines n Outpatient drugs for which the manufacturer seeks to require that associated tests or monitoring services be purchased exclusively from the manufacturer or its designee as a condition of sale *Plans may choose to pay for OTC products as an administrative cost, with the member not incurring a co-pay, these products do not count towards formulary requirements. n

Part D Drug Exclusions (cont) n Part A Prescriptions – In skilled nursing homes – up to 100 day stay n n Related to the terminal illness for hospice patients Part B Outpatient Drugs n Durable Medical Equipment Drugs (e. g. , inhalation therapy, insulin w/pumps & some chemotherapeutics) n Immunosuppressive Drugs n Hemophilia Clotting Factors n Selected Oral Anti-Cancer Drugs n Selected Oral Anti-Emetic Drugs, up to 48 hrs after chemotherapy administration n Erythropoietin for persons on dialysis n Intravenous Immune Globulin, provided in the home

P&T Committee: Requirements n Membership includes the following: n n The majority are practicing physicians and pharmacists. Various clinical specialties that reflect the needs of the plan beneficiaries. At least one practicing physician and pharmacist who are experts in the care of the disabled or elderly. CMS provides extensive guidance on the expectations surrounding the composition and activities of the P&T committee

Formulary Review: Rationale n Medicare Modernization Act requires CMS to review formularies and related processes to ensure: Beneficiaries have access to a broad range of medically appropriate drugs to treat all disease states, and n Formulary design does not discriminate or substantially discourage enrollment of certain groups n

Formulary Review: CMS Validations Checks for appropriate utilization management strategies n Checks for two drugs per USP category and class n Checks for Key Drug Types as defined by USP n Checks for the most common drugs used in the LTC population n Checks “all or substantially all” requirement n

Formulary Considerations Safety and Efficacy n Cost-effectiveness* n In general, formulary design will be similar to that of commercial plans today, with the added benefit of CMS oversight for adherence to published guidelines. n *The federal government can’t negotiate or mandate pharmacy payment rates or manufacturer rebate levels

Provision of Notice Regarding Formulary Changes n Prior to removing/changing drug from formulary the plan must: Provide 60 days notice to prescribers, network pharmacies, pharmacists and other health plans n CMS will review and approve modifications n For enrollees, must provide either: n n Direct written notice at least 60 days prior to date the change becomes effective, or n At the time a refill is requested, provide a 60 day supply of drug and written notice

Exception Requests Enrollees or their authorized representative may request an exception when: n A non-formulary drug is prescribed and is medically necessary n The cost-sharing status of a drug an enrollee is using changes n A drug covered under a more expensive cost-sharing tier is prescribed because the drug covered under the less expensive cost-sharing tier is medically inappropriate n The enrollee is using a drug that has been removed from the formulary n Ensures access to medically necessary Medicare D covered prescription drugs

Cost and Utilization Controls n Prior Authorization n Step Therapy n Quantity Limits n Frequency Limits n Generic Substitution n Drug Utilization Review-Prospective and Retrospective n Tiered formulary design

Appeal Processes n 1 st Step: Plan Re-determination n 7 days to respond n 72 hours, if expedited n 2 nd Step: IRE Reconsideration n Independent Review Entity (IRE), CMS contractor, which reviews plan redeterminations n 7 days to respond n 72 hours, if expedited n 3 rd Step: Administrative Law Judge n Must satisfy minimum amount requirement n 4 th Step – Medicare Appeals Council n 5 th Step – Federal District Court
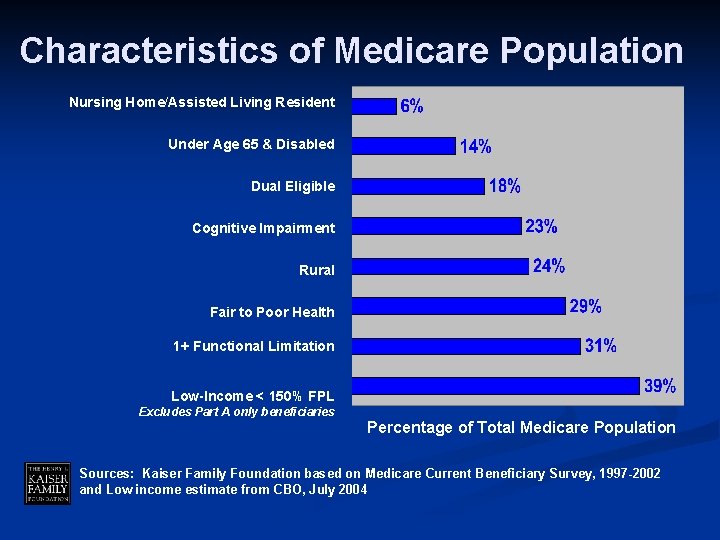
Characteristics of Medicare Population Nursing Home/Assisted Living Resident Under Age 65 & Disabled Dual Eligible Cognitive Impairment Rural Fair to Poor Health 1+ Functional Limitation Low-Income < 150% FPL Excludes Part A only beneficiaries Percentage of Total Medicare Population Sources: Kaiser Family Foundation based on Medicare Current Beneficiary Survey, 1997 -2002 and Low income estimate from CBO, July 2004

Issues for the Duals What happens, when they … n n Ignore notices regarding Rx changes Don’t know how to use their assigned plan Learn the drug Medicaid paid for isn’t covered by their new Medicare plan Have higher out of pocket costs for copays, non - covered drugs

Formularies – Transition Process • Plans have flexibility, but CMS guidance expects: – 1 -time transition supply for new enrollees Ambulatory 30 days Nursing Home 90 to 180 days – 1 -time temporary emergency supply for others For changes in level of care (nursing home, acute hospital, etc. ) or during appeals Drug plans that want to serve Medicare beneficiaries enrolling in the new prescription drug benefit next year must meet strict standards to assure that older and disabled Americans will be able to make the transition to the new coverage smoothly. Mark B. Mc. Clellan, March 16, 2005, CMS Press Release

Indiana Medicaid: Specific Approach for Dual Eligibles Indiana Medicaid will continue to cover Medicare D excluded drugs to the extent that they are covered in the Medicaid program today. Current dual population estimated at 100, 000 lives. Examples: n Over the counter drugs on the Indiana Medicaid formulary n Agents for treating symptoms of cough/colds and prescription vitamins n Barbiturates and benzodiazepines

Everyone Agrees: It’s Difficult to Comprehend all the Details “You choose a prescription drug plan and pay a monthly $35 premium. Okay, now it gets a little complex…” - Reader’s Digest, April 2004
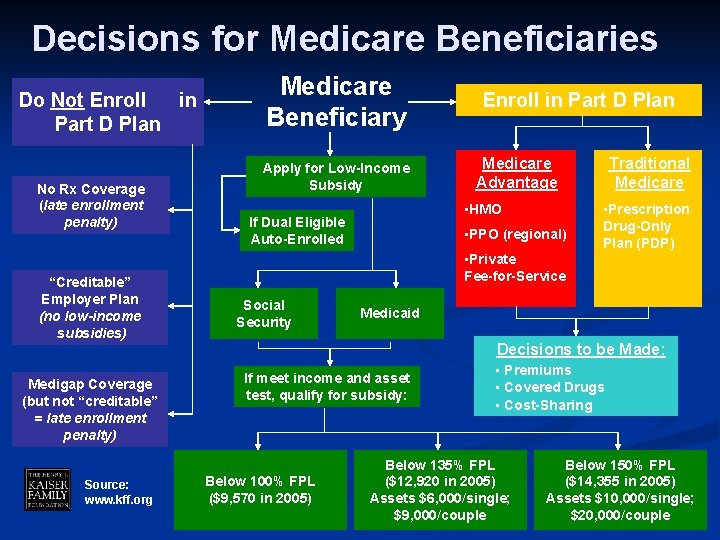
Decisions for Medicare Beneficiaries Do Not Enroll in Part D Plan No Rx Coverage (late enrollment penalty) “Creditable” Employer Plan (no low-income subsidies) Medicare Beneficiary Apply for Low-Income Subsidy Enroll in Part D Plan Medicare Advantage • HMO If Dual Eligible Auto-Enrolled • PPO (regional) Traditional Medicare • Prescription Drug-Only Plan (PDP) • Private Fee-for-Service Social Security Medicaid Decisions to be Made: Medigap Coverage (but not “creditable” = late enrollment penalty) Source: www. kff. org If meet income and asset test, qualify for subsidy: Below 100% FPL ($9, 570 in 2005) • Premiums • Covered Drugs • Cost-Sharing Below 135% FPL ($12, 920 in 2005) Assets $6, 000/single; $9, 000/couple Below 150% FPL ($14, 355 in 2005) Assets $10, 000/single; $20, 000/couple

“ Unfortunately, you have what we call ‘no insurance. ’ ” 26

Issues for Practicing Physicians n Assisting beneficiaries with understanding the new coverage available n Motivating patients to take action and apply for the benefit that comes closest to meeting their needs n Navigating multiple drug formularies n Coordinating prior authorizations & appeals n Comprehending the ongoing changes that will likely occur

Medicare Prescription Drug Benefit Positive Effects n Enhancement of existing Medicare benefit package n Access to subsidized prescription drug coverage n Improved availability and compliance with treatment regimens n Improved health and reduction of adverse health effects

Medicare Prescription Drug Benefit Timeline n n n n n January 21, 2005 - Final Rule Published June 6 - Bid submission July - Finalization pharmacy contracts September 14 - PDPs announced October 1 - Marketing begins October 13 - Prescription Drug Plan Finder Tool rollout November 15 - Enrollment begins January 1, 2006 - Benefit begins May 15, 2006 – Last day to enroll before late enrollment penalty

Sources of Information n CMS Website n www. cms. hhs. gov/medicarereform/pdbma n www. hhs. gov/medlearn/drugcoverage. asp n www. cms. hhs. gov/medicarereform/factsheets. asp n www. cms. hhs. gov/medicarereform/drugcoveragefaqs. asp n www. cms. hhs. gov/partnerships/news/mma/default. asp n www. cms. hhs. gov/mailinglist n www. cms. hhs. gov/providers n http: //www. cms. hhs. gov/medlearn/drugcoverage. asp#train n 1 -800 -Medicare n Social Security Administration n www. ssa. gov [Look under Medicare Outreach] n Kaiser Family Foundation n www. kff. org/rxdrugs/index. cfm n United States Pharmacopoeia (USP) n www. usp. org/Healthcare. Info/mmg/
- Slides: 30