The MADIT II Trial Multicenter Autonomic Defibrillator Implantation

The MADIT II Trial Multicenter Autonomic Defibrillator Implantation Trial II Presented at the American College of Cardiology 51 st Annual Scientific Session Atlanta, GA Arthur J. Moss, M. D. for the MADIT II investigators www. Clinical trial results. org

Multicenter Autonomic Defibrillator Implantation Trial II (MADIT II): Background • MADIT & Multicenter Unsustained Tachycardia Trial (MUSTT): Demonstrated a benefit from ICD implantation in patients with CAD, reduced ventricular function, unsustained ventricular tachycardia, and inducible ventricular tachycardia on EP testing. • In MADIT II: No EP study required, inclusion criteria included a prior MI at least 30 days previous to implantation, and EF of 30% or less. • MADITT randomized 1232 patients from 71 US centers and 5 European centers to receive either an ICD, or to continue on conventional medical therapy • 3 to 4 million patients fit the entry criteria • 400, 000 new cases annually www. Clinical trial results. org

MADIT II: Study Design Patients with prior MI within 30 days and LVEF < 30% randomized in a 3: 2 ratio 71 US centers and 5 European centers Implantable defibrillator Conventional medical therapy (n=742) (n=490) All Cause Mortality - Average follow-up of 20 months Stopped early by Data Safety Monitoring Board www. Clinical trial results. org
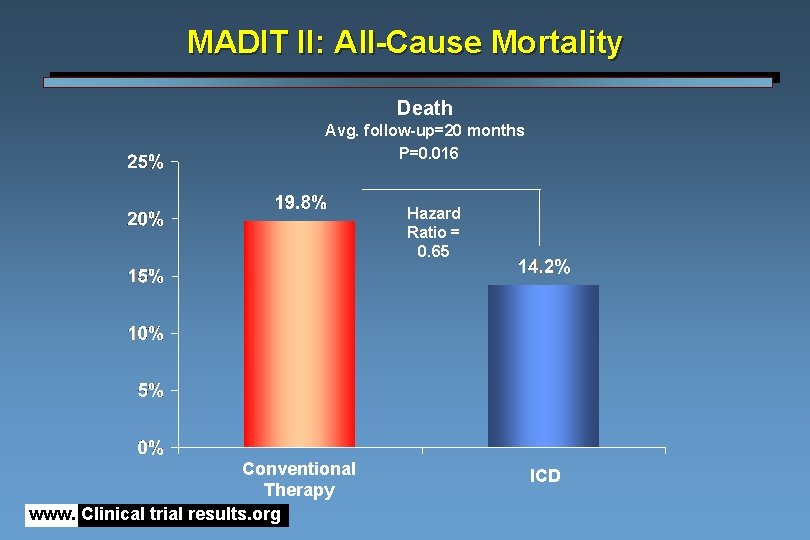
MADIT II: All-Cause Mortality Death Avg. follow-up=20 months P=0. 016 Hazard Ratio = 0. 65 Conventional Therapy www. Clinical trial results. org ICD
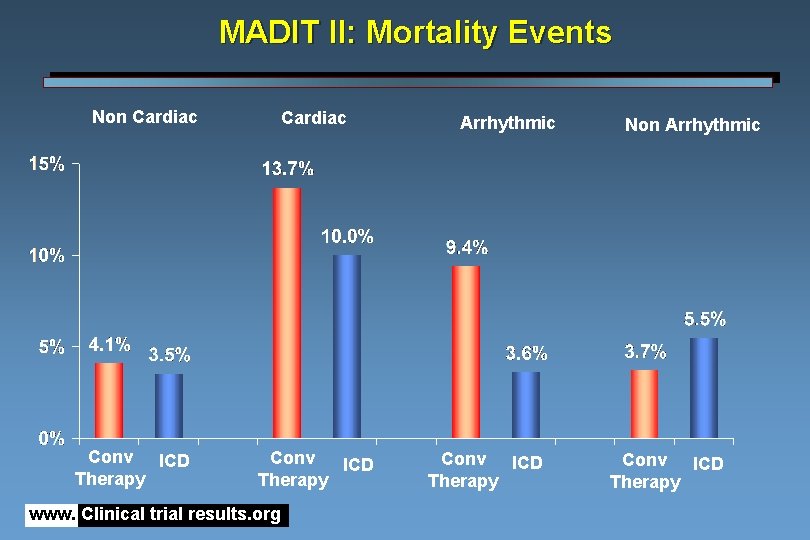
MADIT II: Mortality Events Non Cardiac Conv ICD Therapy www. Clinical trial results. org Arrhythmic Conv ICD Therapy Non Arrhythmic Conv ICD Therapy
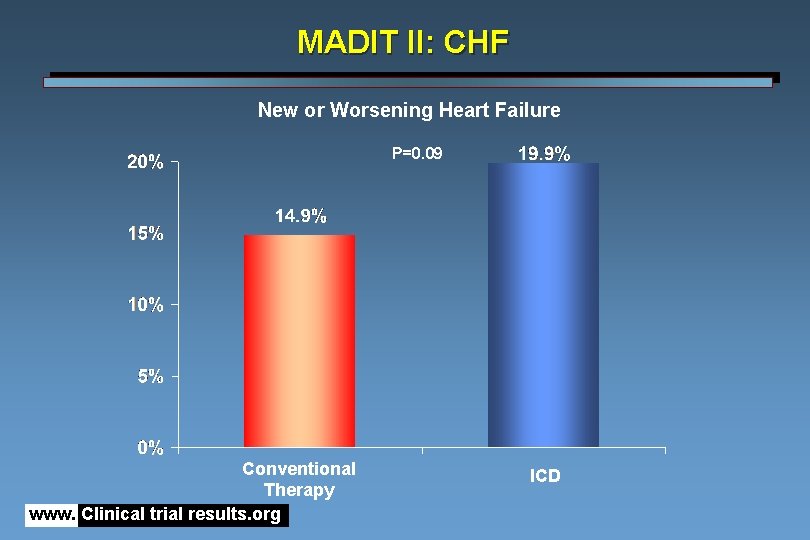
MADIT II: CHF New or Worsening Heart Failure P=0. 09 Conventional Therapy www. Clinical trial results. org ICD
- Slides: 6