The LUTONIX DCB Program Safety Track Record and

The LUTONIX DCB Program Safety Track Record and Other Considerations Kenneth Rosenfield, MD Massachusetts General Hospital Boston, MA

disclosures
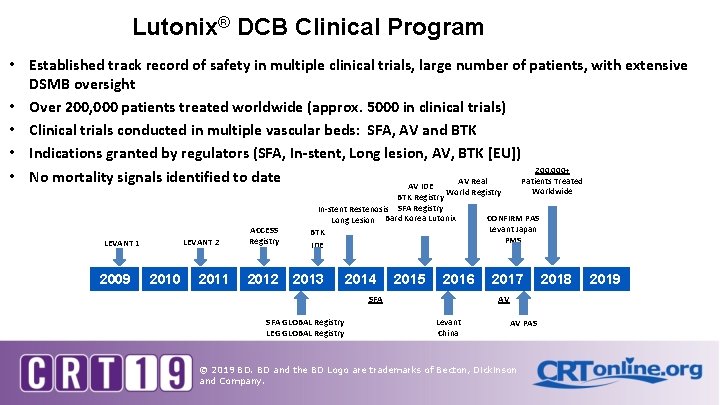
Lutonix® DCB Clinical Program • Established track record of safety in multiple clinical trials, large number of patients, with extensive DSMB oversight • Over 200, 000 patients treated worldwide (approx. 5000 in clinical trials) • Clinical trials conducted in multiple vascular beds: SFA, AV and BTK • Indications granted by regulators (SFA, In-stent, Long lesion, AV, BTK [EU]) 200, 000+ • No mortality signals identified to date Patients Treated AV Real AV IDE LEVANT 2 LEVANT 1 2009 2010 2011 ACCESS Registry 2012 Worldwide World Registry BTK Registry In-stent Restenosis SFA Registry CONFIRM PAS Long Lesion Bard Korea Lutonix Levant Japan BTK PMS IDE 2013 2014 2015 2016 SFA GLOBAL Registry LEG GLOBAL Registry 2017 AV Levant China AV PAS © 2019 BD. BD and the BD Logo are trademarks of Becton, Dickinson and Company. 2018 2019

Lutonix® DCB Design Optimized for Safety and Efficacy Levant 2 pharmacokinetics (subset analysis): -serum paclitaxel < 3 ng/m. L @ 1 h -mean elimination half-life = 6. 88 h -below quantifiable in plasma at 24 h GLP porcine studies: Downstream No ischemia from muscles downstream emboli Clearance organs No systemic toxicity Supports safety of 6 mmx 1 meter lesion! (37 mg)
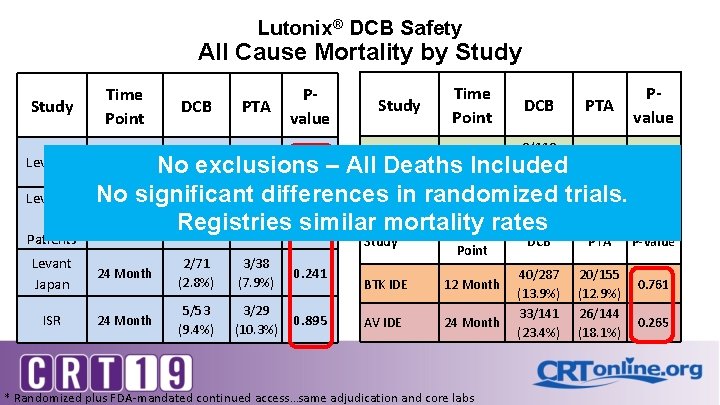
Lutonix® DCB Safety All Cause Mortality by Study Levant 1 Levant 2 All Patients Time Point DCB PTA Pvalue 4/49 (8. 2%) 5/52 (9. 6%) 0. 798 Study Time Point Long Lesion 24 month DCB PTA 8/118 (6. 8%) No exclusions – All Deaths Included NA Global 36/691 No significant differences in randomized trials. 24 Month NA (5. 2%) Registry 147/1029* 17/160 60 months 0. 198 Registries similar mortality (14. 3%) (10. 6%) Time rates 24 Month Study Levant Japan 24 Month 2/71 (2. 8%) 3/38 (7. 9%) 0. 241 ISR 24 Month 5/53 (9. 4%) 3/29 (10. 3%) 0. 895 Point BTK IDE 12 Month AV IDE 24 Month * Randomized plus FDA-mandated continued access…same adjudication and core labs DCB PTA 40/287 (13. 9%) 33/141 (23. 4%) 20/155 (12. 9%) 26/144 (18. 1%) Pvalue NA NA P-value 0. 761 0. 265
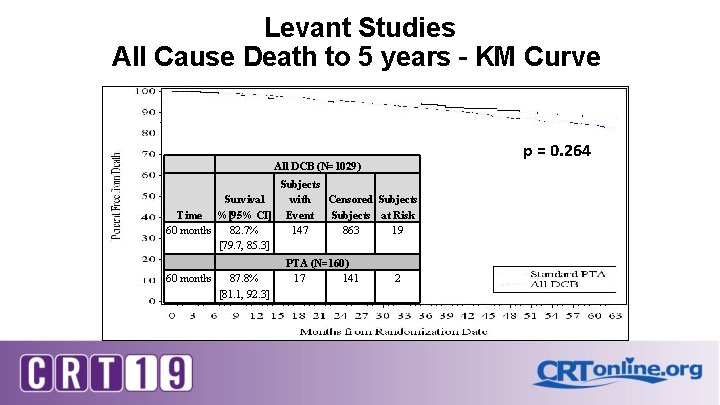
Levant Studies All Cause Death to 5 years – KM Curve p = 0. 264 All DCB (N=1029) Survival Time %[95% CI] 60 months 82. 7% [79. 7, 85. 3] 60 months 87. 8% [81. 1, 92. 3] Subjects with Censored Subjects Event Subjects at Risk 147 863 19 PTA (N=160) 17 141 2
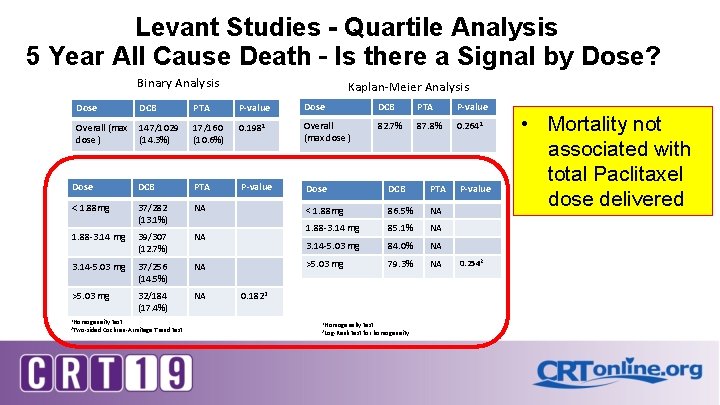
Levant Studies - Quartile Analysis 5 Year All Cause Death – Is there a Signal by Dose? Binary Analysis Kaplan-Meier Analysis Dose DCB PTA P-value Overall (max dose ) 147/1029 (14. 3%) 17/160 (10. 6%) 0. 1981 Overall (max dose ) 82. 7% 87. 8% 0. 2641 Dose DCB PTA P-value Dose DCB PTA < 1. 88 mg 37/282 (13. 1%) NA < 1. 88 mg 86. 5% NA 1. 88 -3. 14 mg 39/307 (12. 7%) NA 1. 88 -3. 14 mg 85. 1% NA 3. 14 -5. 03 mg 84. 0% NA 3. 14 -5. 03 mg 37/256 (14. 5%) NA >5. 03 mg 79. 3% NA >5. 03 mg 32/184 (17. 4%) NA 1 Homogeneity 2 Two-sided test Cochran-Armitage Trend test 0. 1822 1 Homogeneity 2 Log-Rank test for homogeneity P-value 0. 2542 • Mortality not associated with total Paclitaxel dose delivered
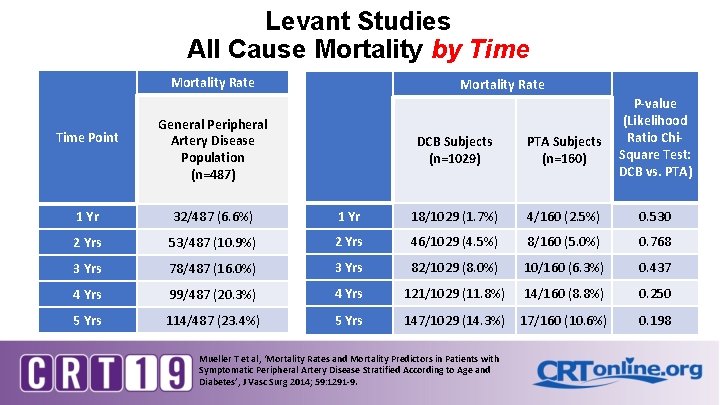
Levant Studies All Cause Mortality by Time Mortality Rate Time Point Mortality Rate General Peripheral Artery Disease Population (n=487) DCB Subjects (n=1029) PTA Subjects (n=160) P-value (Likelihood Ratio Chi. Square Test: DCB vs. PTA) 1 Yr 32/487 (6. 6%) 1 Yr 18/1029 (1. 7%) 4/160 (2. 5%) 0. 530 2 Yrs 53/487 (10. 9%) 2 Yrs 46/1029 (4. 5%) 8/160 (5. 0%) 0. 768 3 Yrs 78/487 (16. 0%) 3 Yrs 82/1029 (8. 0%) 10/160 (6. 3%) 0. 437 4 Yrs 99/487 (20. 3%) 4 Yrs 121/1029 (11. 8%) 14/160 (8. 8%) 0. 250 5 Yrs 114/487 (23. 4%) 5 Yrs 147/1029 (14. 3%) 17/160 (10. 6%) 0. 198 Mueller T et al, ‘Mortality Rates and Mortality Predictors in Patients with Symptomatic Peripheral Artery Disease Stratified According to Age and Diabetes’, J Vasc Surg 2014; 59: 1291 -9.
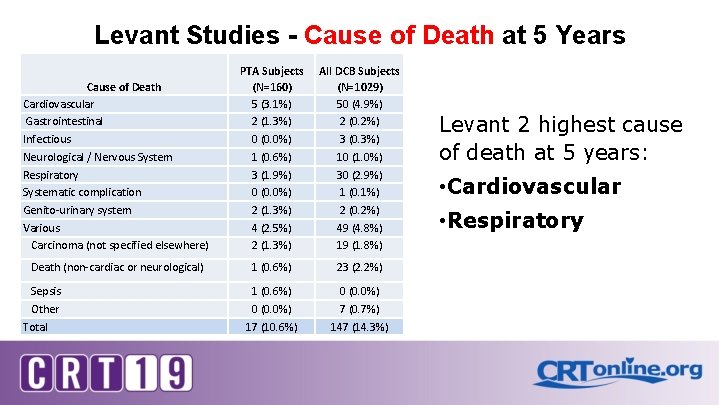
Levant Studies - Cause of Death at 5 Years Cause of Death Cardiovascular Gastrointestinal Infectious Neurological / Nervous System Respiratory Systematic complication Genito-urinary system Various Carcinoma (not specified elsewhere) PTA Subjects (N=160) 5 (3. 1%) 2 (1. 3%) 0 (0. 0%) 1 (0. 6%) 3 (1. 9%) 0 (0. 0%) 2 (1. 3%) 4 (2. 5%) 2 (1. 3%) All DCB Subjects (N=1029) 50 (4. 9%) 2 (0. 2%) 3 (0. 3%) 10 (1. 0%) 30 (2. 9%) 1 (0. 1%) 2 (0. 2%) 49 (4. 8%) 19 (1. 8%) Death (non-cardiac or neurological) 1 (0. 6%) 23 (2. 2%) 1 (0. 6%) 0 (0. 0%) 17 (10. 6%) 0 (0. 0%) 7 (0. 7%) 147 (14. 3%) Sepsis Other Total Levant 2 highest cause of death at 5 years: • Cardiovascular • Respiratory

Lutonix® DCB Safety Summary • Biology and kinetics: 2μg/mm 2 dose (lowest); Plasma PTX is rapidly cleared (plasma half life = 6. 88 hours, below detectable limits at 24 hours) • Clinical Studies – SFA/pop o Levant Studies o No significant difference in all-cause death rates between DCB (n=1029) / PTA (n=160) o No difference all death between DCB and PTA by dose/time o Levant 1, Levant Japan and ISR randomized studies: o No significant difference for all deaths at 24 months between DCB and PTA o Global Registry and Long Lesion (sicker patients, longer balloon length/patient) o Low death rates at 24 months as compared to literature • Clinical Studies - BTK & AV (Randomized studies in other vascular beds) o No significant difference for all deaths between DCB & PTA
- Slides: 10