The Lotus Device Next Generation Improvements and Design







- Slides: 11

The Lotus Device: Next Generation Improvements and Design Features Ted Feldman, M. D. , MSCAI FACC FESC Evanston Hospital CRT Cardiovascular Research Technologies Washington D. C. March 4 -6 th, 2018

Ted Feldman MD, MSCAI FACC FESC Disclosure Information The following relationships exist: Grant support: Abbott, BSC, Cardiokinetics, Corvia, Edwards, WL Gore Consultant: Abbott, BSC, Edwards, WL Gore Stock Options: Mitralign Off label use of products and investigational devices will be discussed in this presentation

Background • Transcatheter aortic valve implantation (TAVI): Well-established alternative to surgery for patients with severe aortic stenosis • Current TAVI limitations include: Suboptimal deployment & paravalvular leak • Lotus Valve System: Fully repositionable & retrievable TAVI device; Adaptive Seal to minimise PVL Lotus Nitinol Bovine Valve Frame Pericardium System Radiopaque Marker Adaptive Seal • Controlled mechanical expansion; rapid pacing not needed during deployment • Early valve function; haemodynamic stability during implantation • Complete assessment before release; reposition/retrieve if not acceptable

Lotus Valve Deployment Phases 1. Elongated Configuration (for Delivery) 2. Intermediate Configuration 25 mm 20 mm 3. Final Locked Configuration 27 mm 23 mm 40 mm 35 mm 23 mm Valve 19 mm 27 mm Valve 23 mm Valve 27 mm Valve

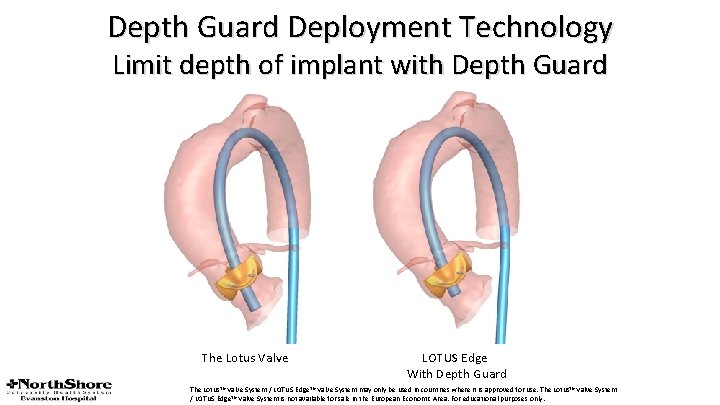
Depth Guard Technology (A) The current-generation Lotus valve (Boston Scientific, Marlborough, Massachusetts) results in deep left ventricular outflow tract (LVOT) interaction due to the foreshortening from both ends that occurs during locking of the valve. (B) The next-generation Lotus valve with Depth Guard technology involves foreshortening of the stent frame from the top down during unsheathing. This results in a less deep protrusion of the distal edge of the stent frame into the LVOT. Consequently, LVOT protrusion with Depth Guard is predictable and less pronounced resulting in less interaction with the LVOT.

Depth Guard Deployment Technology Limit depth of implant with Depth Guard The Lotus Valve LOTUS Edge With Depth Guard The Lotus™ Valve System / LOTUS Edge™ Valve System may only be used in countries where it is approved for use. The Lotus™ Valve System / LOTUS Edge™ Valve System is not available for sale in the European Economic Area. For educational purposes only.

LOTUS Edge™ Valve System Design Attributes • Maintains benefits of first generation Lotus • Improves delivery More ergonomic handle • • • Lower profile system • Greater catheter flexibility Optimizes deployment • Depth Guard limits depth of implant • One-view locking

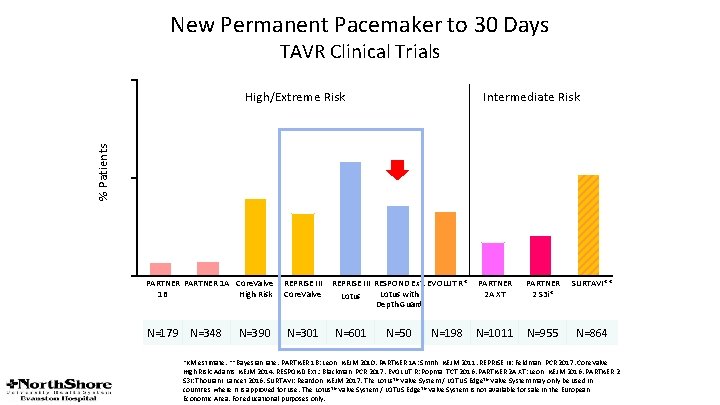
New Permanent Pacemaker to 30 Days TAVR Clinical Trials 50 High/Extreme Risk Intermediate Risk % Patients 29. 1 25 19. 8 3. 4 0 25. 9 18. 0 16. 4 15. 8 8. 5 3. 8 PARTNER 1 A Core. Valve 1 B High Risk N=179 N=348 N=390 REPRISE III Core. Valve N=301 REPRISE III RESPOND Ext. EVOLUT R* Lotus with Lotus Depth Guard N=601 N=50 N=198 10. 2 PARTNER 2 A XT PARTNER 2 S 3 i* SURTAVI** N=1011 N=955 N=864 *KM estimate. **Bayesian rate. PARTNER 1 B: Leon, NEJM 2010. PARTNER 1 A: Smith, NEJM 2011. REPRISE III: Feldman, PCR 2017. Core. Valve High Risk: Adams, NEJM 2014. RESPOND Ext. : Blackman, PCR 2017. EVOLUT R: Popma, TCT 2016. PARTNER 2 A XT: Leon, NEJM 2016. PARTNER 2 S 3 i: Thourani, Lancet 2016. SURTAVI: Reardon, NEJM 2017. The Lotus™ Valve System / LOTUS Edge™ Valve System may only be used in countries where it is approved for use. The Lotus™ Valve System / LOTUS Edge™ Valve System is not available for sale in the European Economic Area. For educational purposes only.

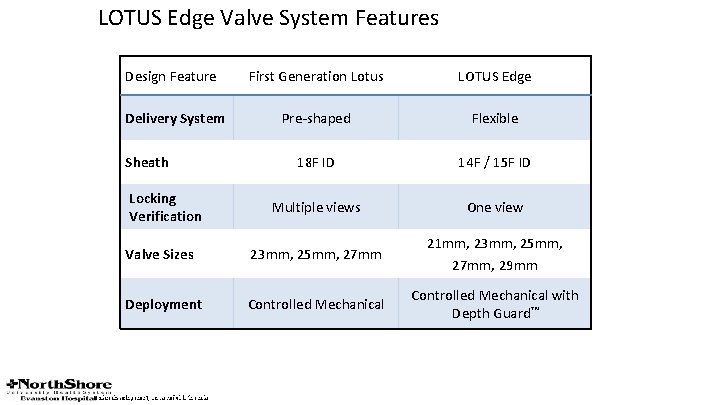
LOTUS Edge Valve System Features Design Feature First Generation Lotus LOTUS Edge Delivery System Pre-shaped Flexible 18 F ID 14 F / 15 F ID Multiple views One view Valve Sizes 23 mm, 25 mm, 27 mm 21 mm, 23 mm, 25 mm, 27 mm, 29 mm Deployment Controlled Mechanical with Depth Guard™ Sheath Locking Verification CAUTION: Lotus is an investigational device and restricted under federal law to investigational use only. Not available for sale. Under development, not available for sale

REPRISE IV: LOTUS Edge Clinical Study LOTUS Edge REPRISE IV Intermediate Risk Patients Bicuspid Subset

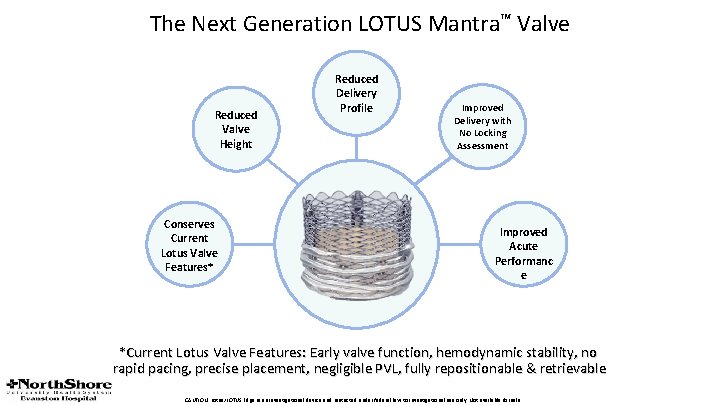
The Next Generation LOTUS Mantra™ Valve Reduced Valve Height Conserves Current Lotus Valve Features* Reduced Delivery Profile Improved Delivery with No Locking Assessment Improved Acute Performanc e *Current Lotus Valve Features: Early valve function, hemodynamic stability, no rapid pacing, precise placement, negligible PVL, fully repositionable & retrievable CAUTION: Lotus/LOTUS Edge is an investigational device and restricted under federal law to investigational use only. Not available for sale.