The Kinetic Molecular Theory Movement in solids liquids













- Slides: 16

The Kinetic Molecular Theory Movement in solids, liquids, and gases

The theory of moving molecules! • Describes the differences between gas, liquid, and solid states.

Assumptions within the KMT • 1. Gases are made of tiny particles that are far apart from each other. • 2. Gas particles are in continuous, rapid, random motion

More Assumptions • 3. There are no attractive forces between molecules under normal conditions. • 4. Collisions between particles are elastic (no energy is lost due to friction).

Last Assumption • 5. Particles at the same temperature have the same average kinetic energy.

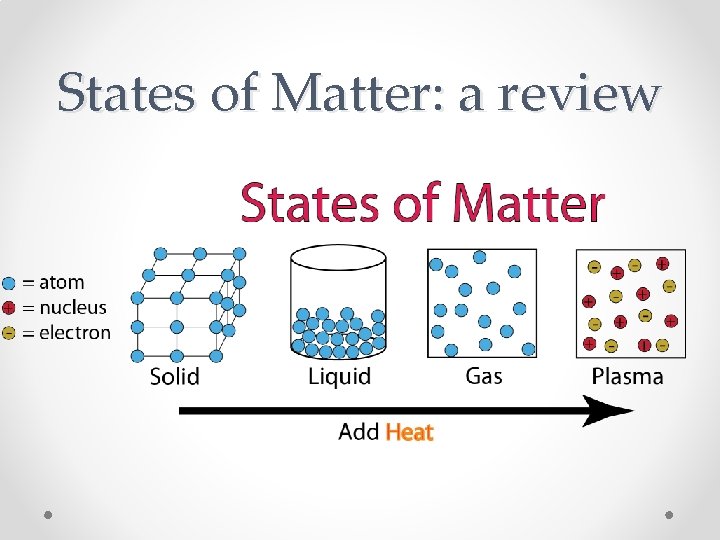
States of Matter: a review

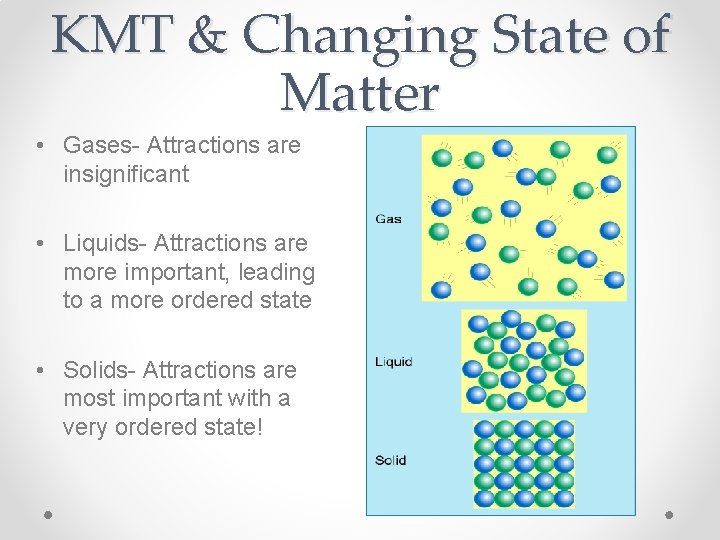
KMT & Changing State of Matter • Gases- Attractions are insignificant • Liquids- Attractions are more important, leading to a more ordered state • Solids- Attractions are most important with a very ordered state!

KMT & Changing State • Involves adding or removing energy (changing temperature) or changing pressure.

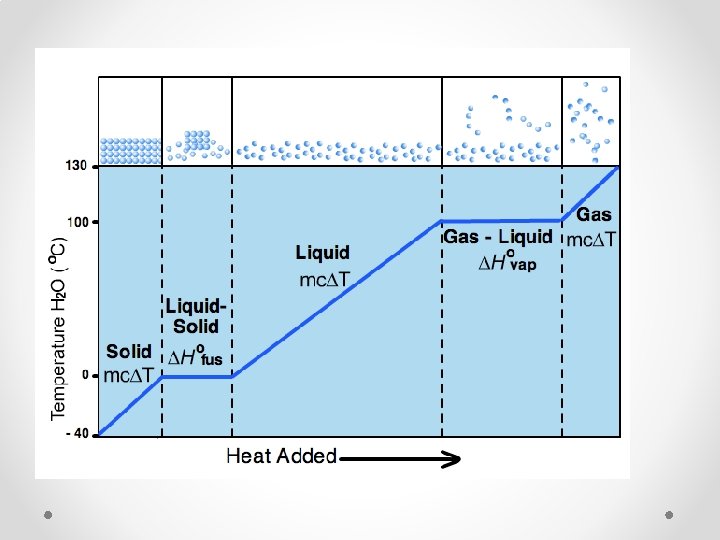
KMT & Changing States of Matter • What happens, on a molecular level, when you add heat to a solid? o Molecules vibrate more rapidly o Molecules “escape” to the liquid states and slide past each other. • What happens when you boil/evaporate a liquid? o Molecules absorb more energy, move faster o Can break weak bonds that keep them liquid and “escape: into the gas state.

KMT and Changing State of Matter • The opposite occurs when you cool a gas down until it becomes a liquid and then cool the liquid until it solidifies.

What happens when we change pressure? • Pressure – the force per unit area • Volume- amount of space an object takes up • Temperature- average kinetic energy of the particles • These 3 concepts are related to each other!

Pressure, Volume, & Temperature • What would happen to the pressure from the molecules in the balloon if I decreased the volume?

Pressure & Volume • Decreasing the Volume would Increase the Pressure V P • There is an inverse relationship between pressure and volume!

Volume & Temperature • What would happen to the balloon if I increased the temperature?

Volume & Temperature • Adding heat would increase the speed of the molecules, which increases the pressure inside the balloon, which increases the volume!
