The internal or thermal energyof moving molecules is













- Slides: 27

The “internal” or “thermal energy”of moving molecules is already something we have identified with TEMPERATURE LIQUIFIES/BOILS SOLIDIFIES/MELTS MERCURY 356. 58 o. C -38. 87 o. C WATER 100. 00 o. C 0. 000 o. C NITROGEN OXYGEN HYDROGEN HELIUM -195. 8 o. C -183. 0 o. C -252. 5 o. C -268. 6 o. C -210. 1 o. C -218. 4 o. C -259. 1 o. C -272. 1 o. C

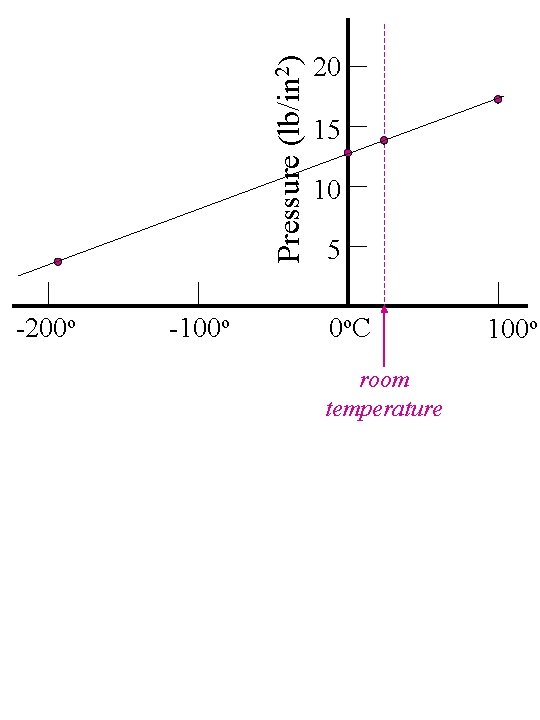
Pressure (lb/in 2) -200 o -100 o 20 15 10 5 0 o. C room temperature 100 o

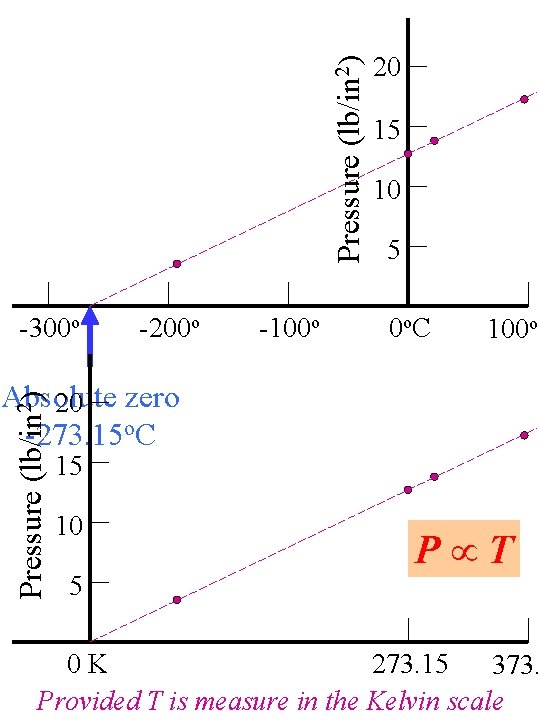
Pressure (lb/in 2) -300 o -200 o -100 o 20 15 10 5 0 o. C 100 o Pressure (lb/in 2) Absolute 20 zero -273. 15 o. C 15 10 5 P T 0 K 273. 15 373. Provided T is measure in the Kelvin scale

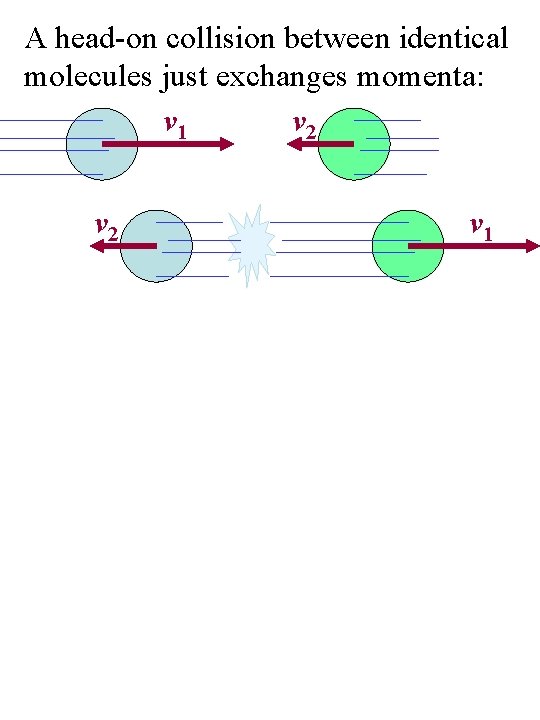
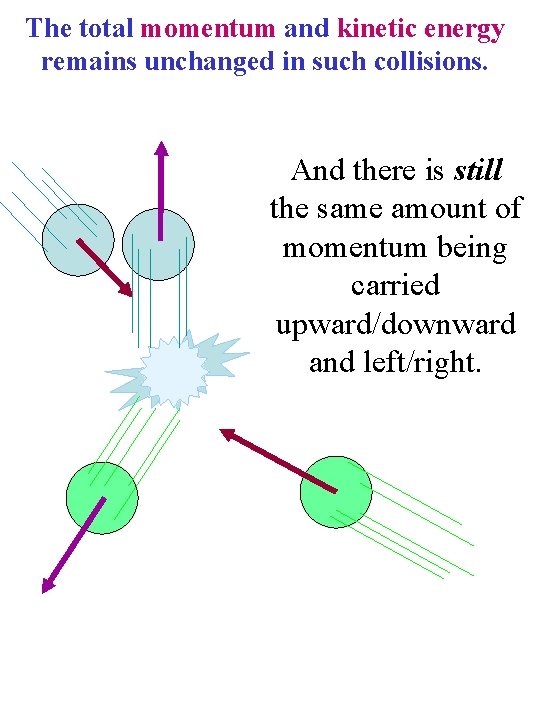
A head-on collision between identical molecules just exchanges momenta: v 1 v 2 v 1

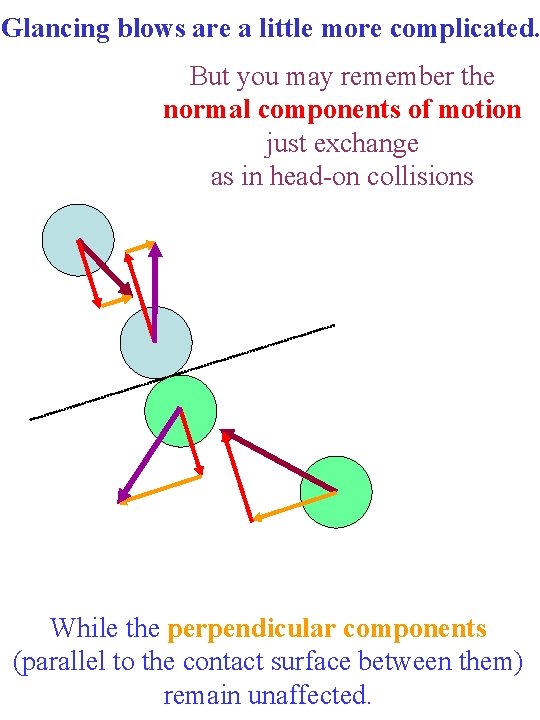
Glancing blows are a little more complicated. But you may remember the normal components of motion just exchange as in head-on collisions While the perpendicular components (parallel to the contact surface between them) remain unaffected.

The total momentum and kinetic energy remains unchanged in such collisions. And there is still the same amount of momentum being carried upward/downward and left/right.

The more and more air we force into a tire (basketball or football) the more taut its rubber/fabric casing becomes and the firmer and more solid its surface grows.

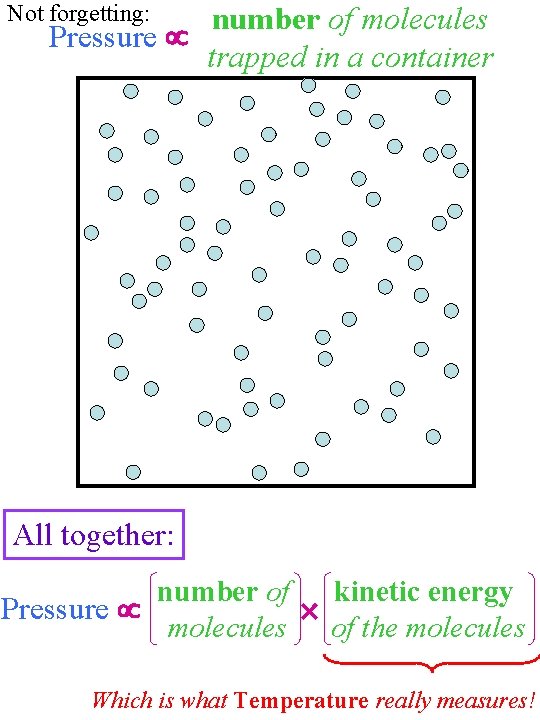
number of molecules Pressure trapped in a container We also saw by a simple demonstration that this pressure is UP as well as DOWN and increased as the speed of the particles creating it increased.

The force of any individual impact in a collision is delivered by the impulse mv -mv Impulse delivered to wall 2 mv

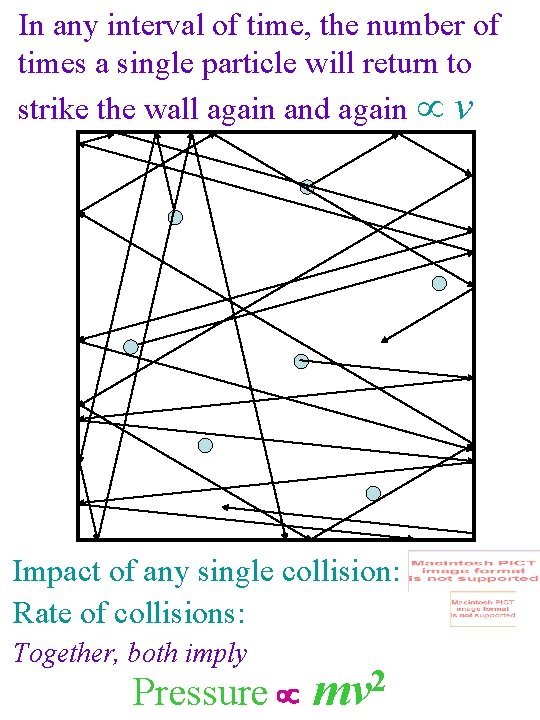
In any interval of time, the number of times a single particle will return to strike the wall again and again v Impact of any single collision: Rate of collisions: Together, both imply 2 Pressure mv

Not forgetting: number of molecules Pressure trapped in a container All together: number of kinetic energy Pressure molecules of the molecules Which is what Temperature really measures!


Raising T increases molecular kinetic energy outward pressure of gas volume occupied by gas

Some of the expanding air escapes out the bottom; it takes less of the warmer air to fill the same space as the cooler air. Essentially the SAME VOLUME now occupied by less gas …which weighs less!

Static equilibrium of fluids P 1 P 2 • All fluids have settled. • Pressures have equalized and are exerted in all directions. P 1 = P 2 • No fluid is moving. Only when there’s a pressure difference P 1 P 2 can there be any acceleration with flow from regions of high pressure to regions of lower pressure.

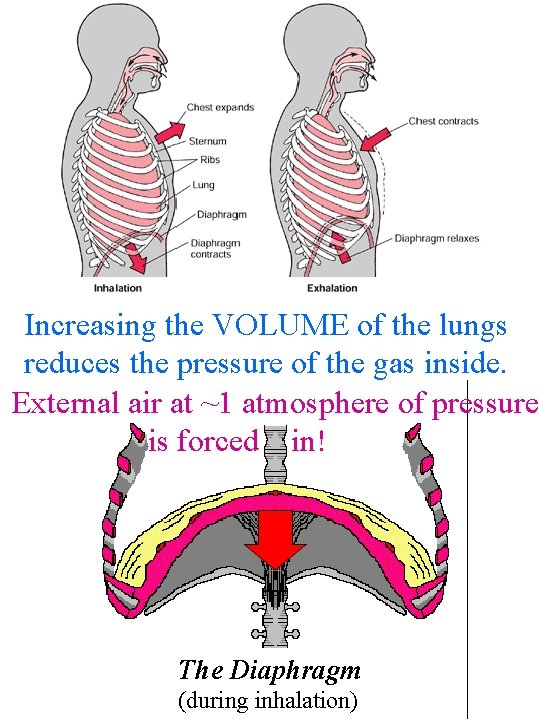
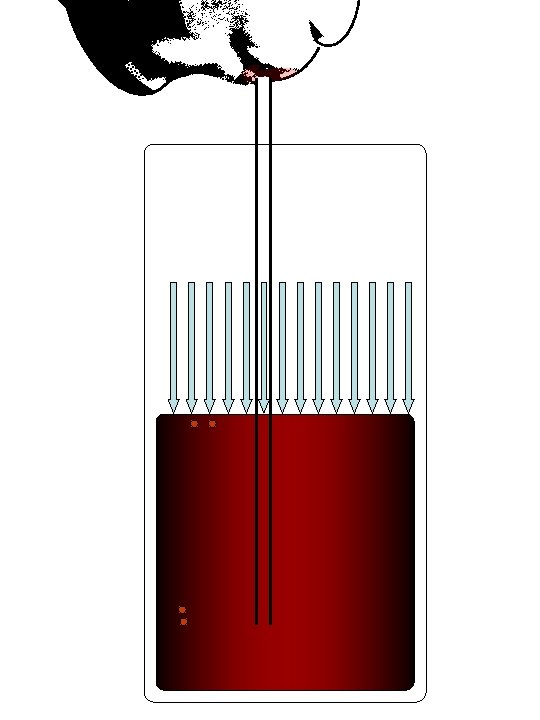
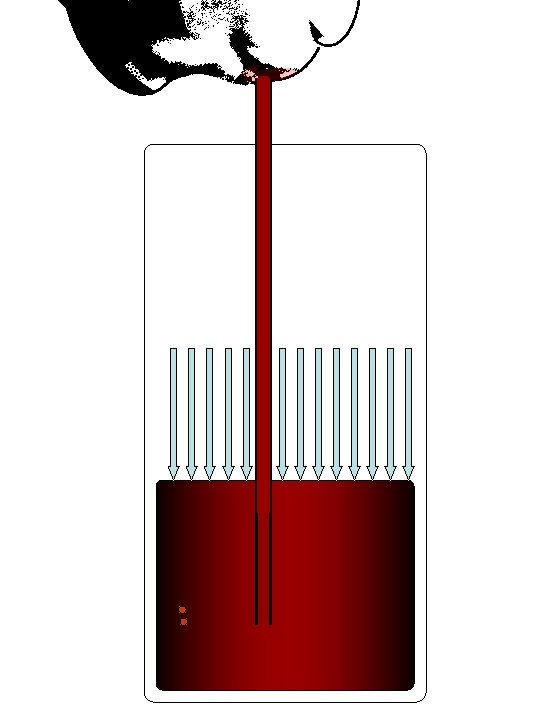
Increasing the VOLUME of the lungs reduces the pressure of the gas inside. External air at ~1 atmosphere of pressure is forced in! The Diaphragm (during inhalation)




Simple Ball Float Valve prevents drains from backing up Ordinarily water drains easily, but a backed up sewer during a storm seals the floating ball so no water flows into your home. Any water above the drain cannot push down the ball float unless its standing water with pressure greater than that pushing the ball float up.


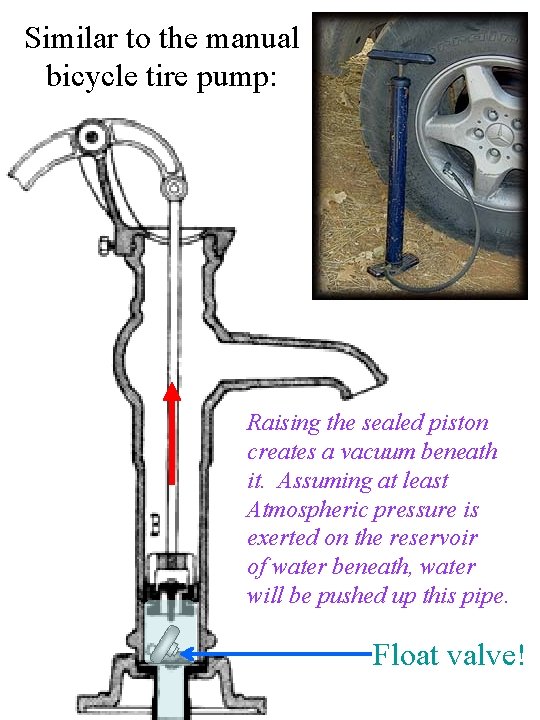
Similar to the manual bicycle tire pump: Raising the sealed piston creates a vacuum beneath it. Assuming at least Atmospheric pressure is exerted on the reservoir of water beneath, water will be pushed up this pipe. Float valve!

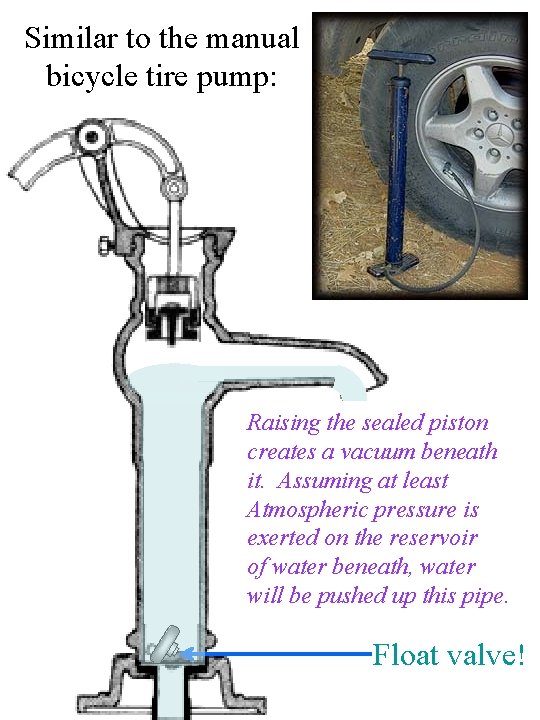
Similar to the manual bicycle tire pump: Raising the sealed piston creates a vacuum beneath it. Assuming at least Atmospheric pressure is exerted on the reservoir of water beneath, water will be pushed up this pipe. Float valve!


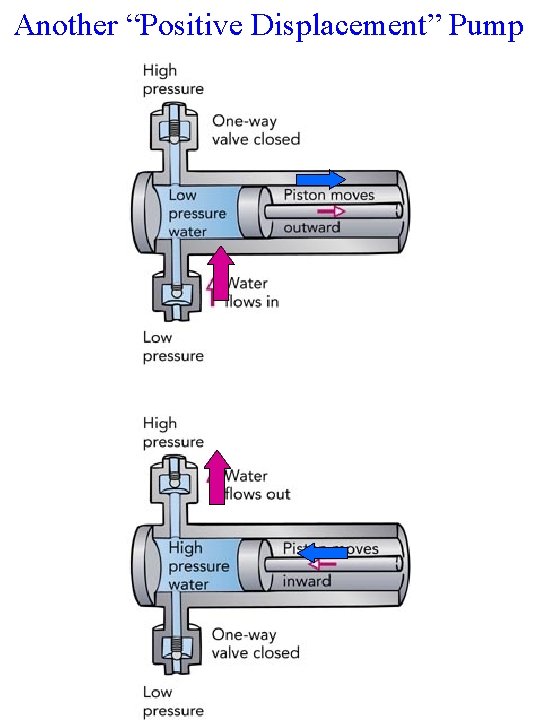
Another “Positive Displacement” Pump

Much simpler to understand is the impeller pump OUT IN which can continuously drive water through. Momentum Transfer Pumps

Industrial impeller