The industrial revolution and air pollution Pittsburgh in

The industrial revolution and air pollution Pittsburgh in the 1940 s “Make great efforts to build China into a strong and prosperous industrialized country under the leadership of the Party and chairman Mao!”

LONDON FOG Aerosols a. k. a. particulate matter (PM) from domestic+industrial combustion “Killer fog” of December 1952 resulted in 10, 000 excess deaths in London in 4 days Altitude inversion < 1 km particles Temperature Coal combustion sulfate organic carbon black carbon
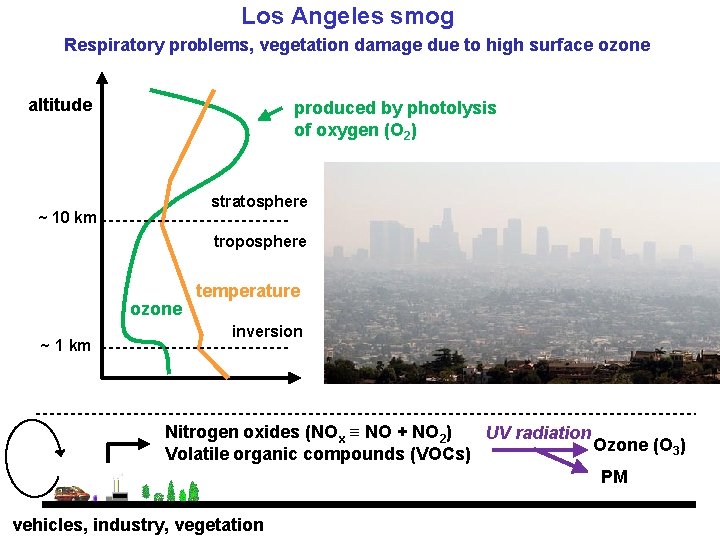
Los Angeles smog Respiratory problems, vegetation damage due to high surface ozone altitude produced by photolysis of oxygen (O 2) stratosphere ~ 10 km troposphere ozone ~ 1 km temperature inversion Nitrogen oxides (NOx ≡ NO + NO 2) UV radiation Ozone (O 3) Volatile organic compounds (VOCs) PM vehicles, industry, vegetation
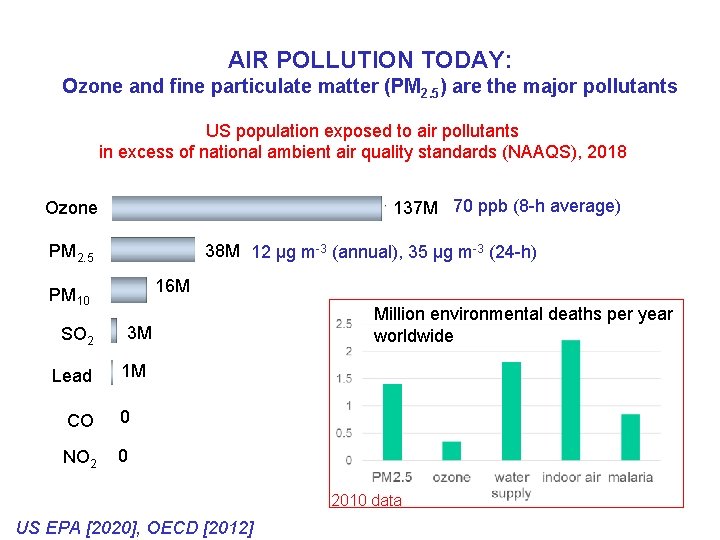
AIR POLLUTION TODAY: Ozone and fine particulate matter (PM 2. 5) are the major pollutants US population exposed to air pollutants in excess of national ambient air quality standards (NAAQS), 2018 137 M 70 ppb (8 -h average) Ozone 38 M 31 12 µg m-3 (annual), 35 µg m-3 (24 -h) PM 2. 5 16 M PM 10 SO 2 Lead 3 M Million environmental deaths per year worldwide 1 M CO 0 NO 2 0 2010 data US EPA [2020], OECD [2012]
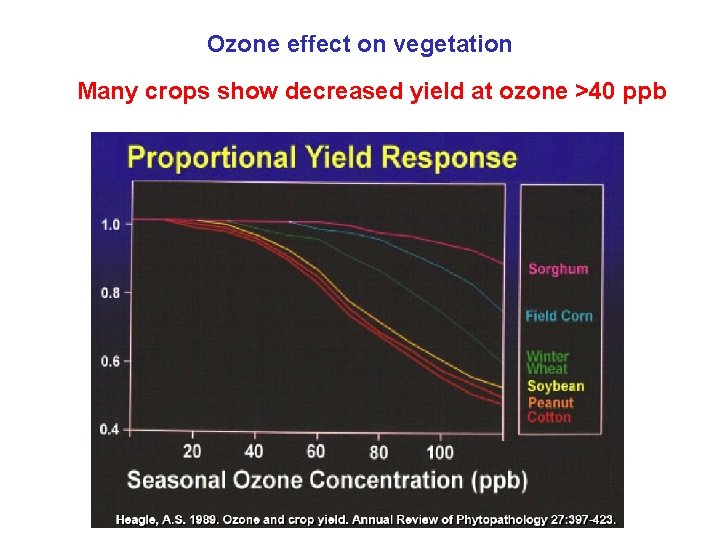
Ozone effect on vegetation Many crops show decreased yield at ozone >40 ppb

Ozone air quality standards in the US and in the world Canadian AQS (8 -h avg. ) Europe AQS (seasonal) U. S. AQS (8 -h avg. ) 2014 2008 0 Preindustrial ozone background 20 40 60 Present-day ozone background at northern mid-latitudes 80 China AQS (8 -h avg. ) U. S. AQS (1 -h avg. ) 1997 100 120 ppb
![Days per year exceeding 70 ppb ozone standard, 2010 -2014 TOAR [2017] Days per year exceeding 70 ppb ozone standard, 2010 -2014 TOAR [2017]](http://slidetodoc.com/presentation_image_h2/91a66b2471c935dee4892e40e34efee9/image-7.jpg)
Days per year exceeding 70 ppb ozone standard, 2010 -2014 TOAR [2017]
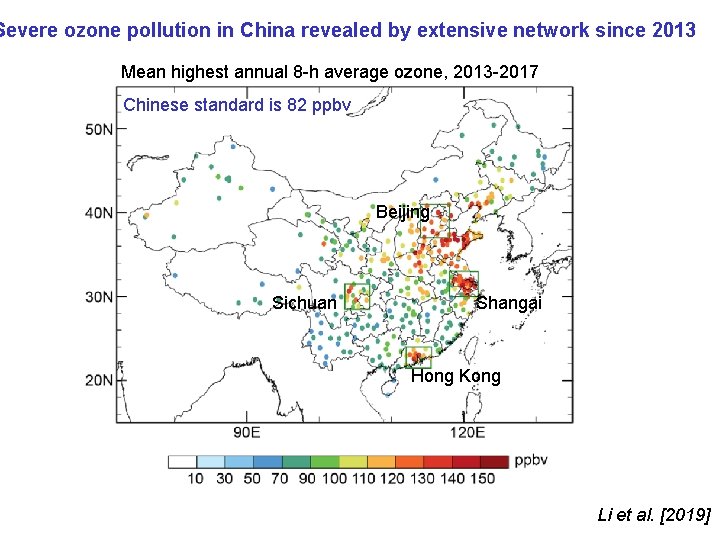
Severe ozone pollution in China revealed by extensive network since 2013 Mean highest annual 8 -h average ozone, 2013 -2017 Chinese standard is 82 ppbv Beijing Sichuan Shangai Hong Kong Li et al. [2019]

Fast ozone production from oxidation of short-lived VOCs catalyzed by HOx and NOx radicals Initiation: production of HOx radicals Propagation: oxidation of VOCs (hydrocarbon RH) in presence of NOx organic peroxy aldehyde Termination: loss of HOx radicals
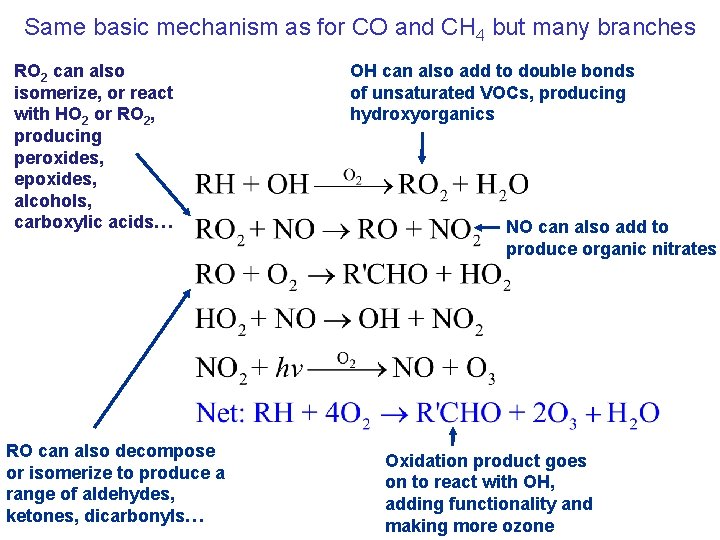
Same basic mechanism as for CO and CH 4 but many branches RO 2 can also isomerize, or react with HO 2 or RO 2, producing peroxides, epoxides, alcohols, carboxylic acids… RO can also decompose or isomerize to produce a range of aldehydes, ketones, dicarbonyls… OH can also add to double bonds of unsaturated VOCs, producing hydroxyorganics NO can also add to produce organic nitrates Oxidation product goes on to react with OH, adding functionality and making more ozone
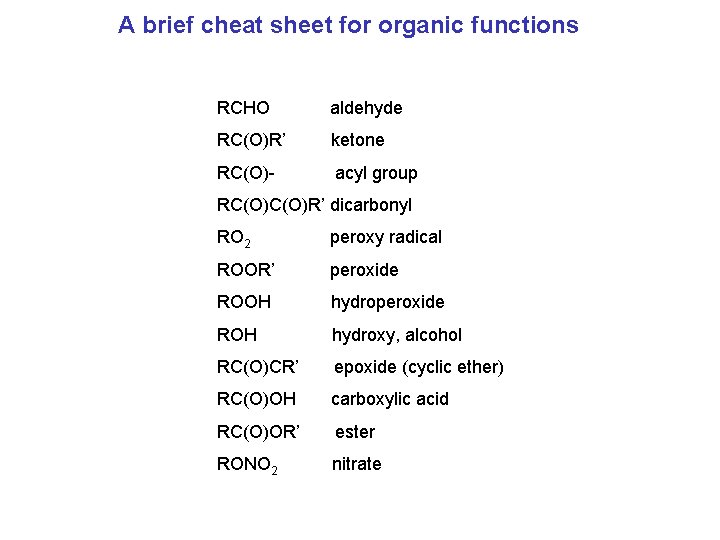
A brief cheat sheet for organic functions RCHO aldehyde RC(O)R’ ketone RC(O)- acyl group RC(O)R’ dicarbonyl RO 2 peroxy radical ROOR’ peroxide ROOH hydroperoxide ROH hydroxy, alcohol RC(O)CR’ epoxide (cyclic ether) RC(O)OH carboxylic acid RC(O)OR’ ester RONO 2 nitrate
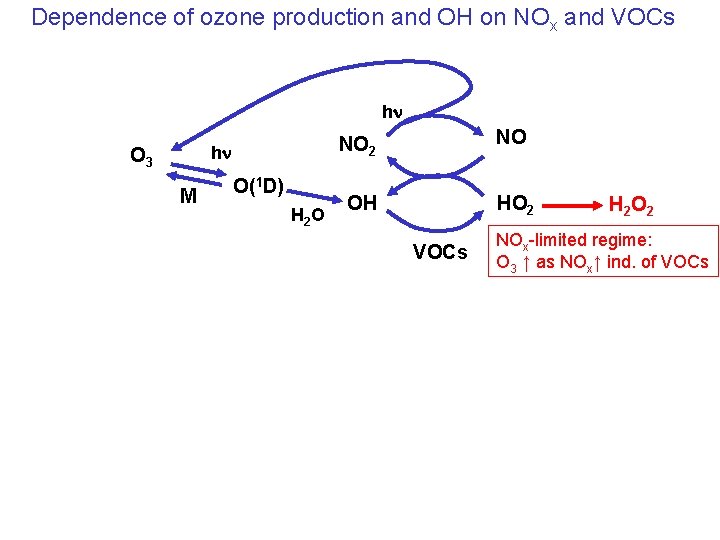
Dependence of ozone production and OH on NOx and VOCs hn hn O 3 M O(1 D) H 2 O NO 2 NO OH HO 2 VOCs H 2 O 2 NOx-limited regime: O 3 ↑ as NOx↑ ind. of VOCs

Dependence of ozone production and OH on NOx and VOCs hn hn O 3 M O(1 D) H 2 O NO 2 NO OH HO 2 NOx-saturated regime: O 3 ↑ as NOx↓ VOCs↑ HNO 3 VOCs
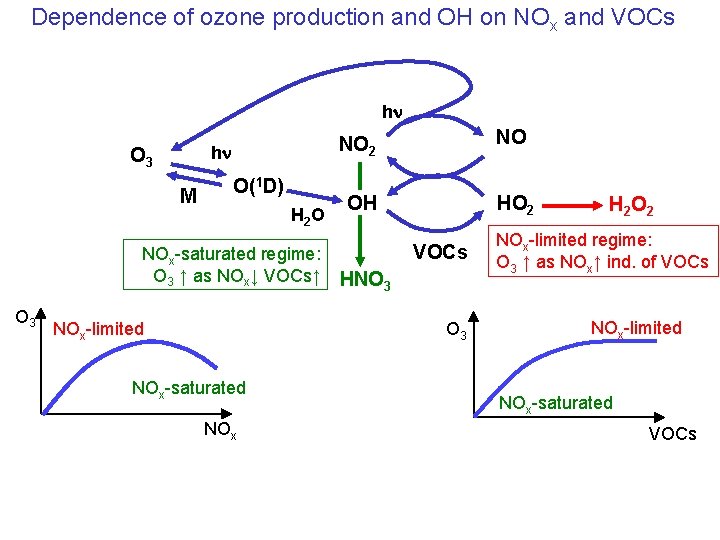
Dependence of ozone production and OH on NOx and VOCs hn hn O 3 M O(1 D) H 2 O NOx-saturated regime: O 3 ↑ as NOx↓ VOCs↑ O 3 NOx-limited NO 2 NO OH HO 2 VOCs HNO 3 NOx-saturated NOx H 2 O 2 NOx-limited regime: O 3 ↑ as NOx↑ ind. of VOCs NOx-limited NOx-saturated VOCs
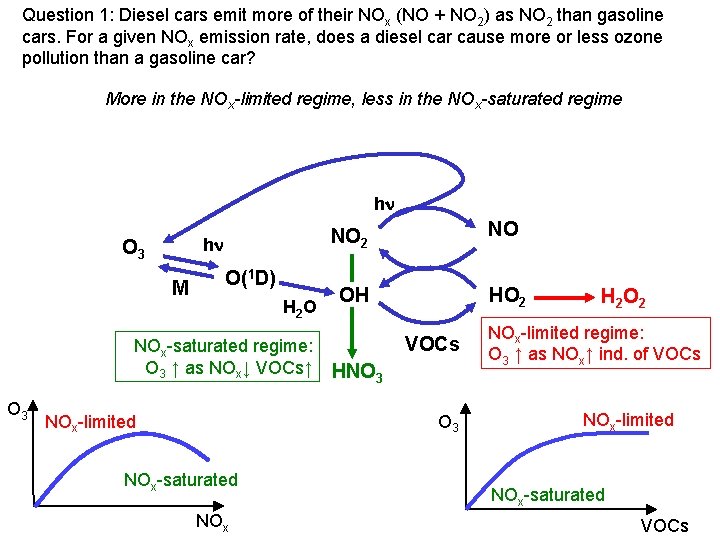
Question 1: Diesel cars emit more of their NOx (NO + NO 2) as NO 2 than gasoline cars. For a given NOx emission rate, does a diesel car cause more or less ozone pollution than a gasoline car? More in the NOx-limited regime, less in the NOx-saturated regime hn hn O 3 M O(1 D) H 2 O NOx-saturated regime: O 3 ↑ as NOx↓ VOCs↑ O 3 NOx-limited NO 2 NO OH HO 2 VOCs HNO 3 NOx-saturated NOx H 2 O 2 NOx-limited regime: O 3 ↑ as NOx↑ ind. of VOCs NOx-limited NOx-saturated VOCs
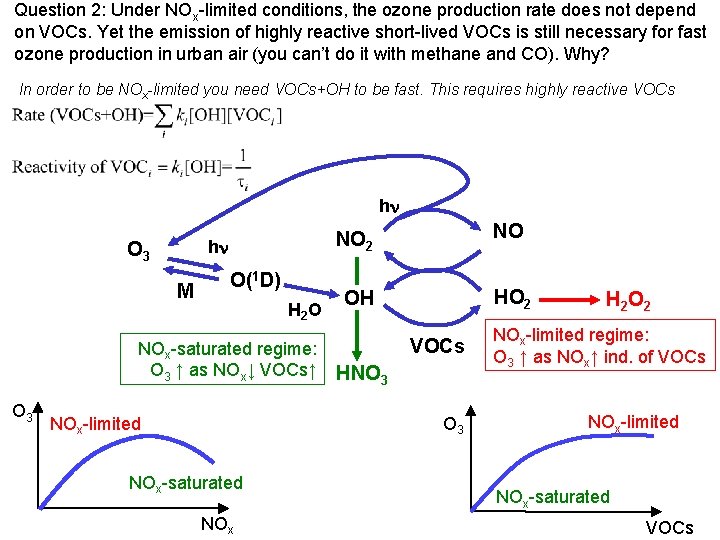
Question 2: Under NOx-limited conditions, the ozone production rate does not depend on VOCs. Yet the emission of highly reactive short-lived VOCs is still necessary for fast ozone production in urban air (you can’t do it with methane and CO). Why? In order to be NOx-limited you need VOCs+OH to be fast. This requires highly reactive VOCs hn hn O 3 M O(1 D) H 2 O NOx-saturated regime: O 3 ↑ as NOx↓ VOCs↑ O 3 NOx-limited NO 2 NO OH HO 2 VOCs HNO 3 NOx-saturated NOx H 2 O 2 NOx-limited regime: O 3 ↑ as NOx↑ ind. of VOCs NOx-limited NOx-saturated VOCs
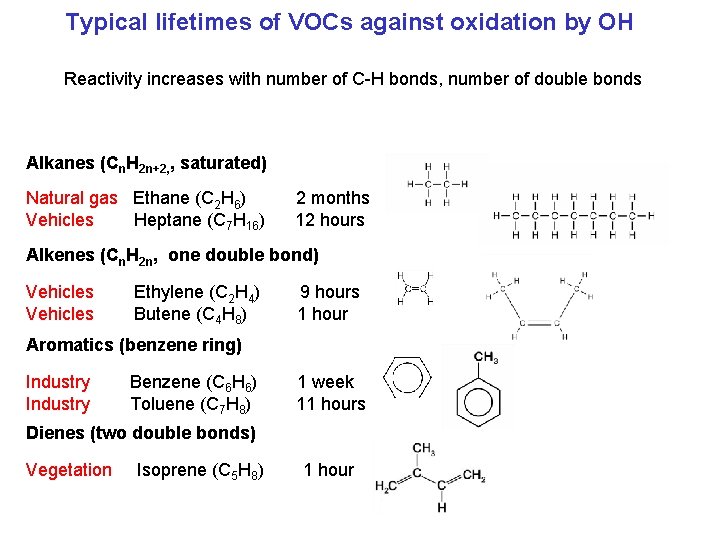
Typical lifetimes of VOCs against oxidation by OH Reactivity increases with number of C-H bonds, number of double bonds Alkanes (Cn. H 2 n+2, , saturated) Natural gas Ethane (C 2 H 6) Vehicles Heptane (C 7 H 16) 2 months 12 hours Alkenes (Cn. H 2 n, one double bond) Vehicles Ethylene (C 2 H 4) Butene (C 4 H 8) 9 hours 1 hour Aromatics (benzene ring) Industry Benzene (C 6 H 6) Toluene (C 7 H 8) 1 week 11 hours Dienes (two double bonds) Vegetation Isoprene (C 5 H 8) 1 hour
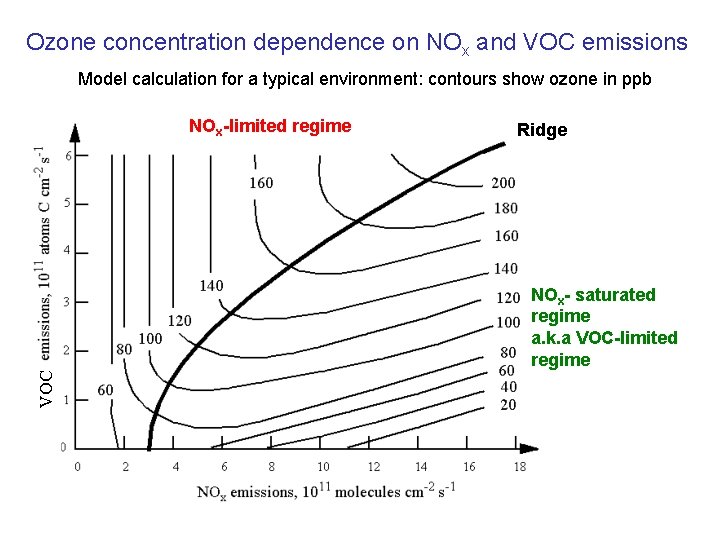
Ozone concentration dependence on NOx and VOC emissions Model calculation for a typical environment: contours show ozone in ppb NOx-limited regime Ridge VOC NOx- saturated regime a. k. a VOC-limited regime
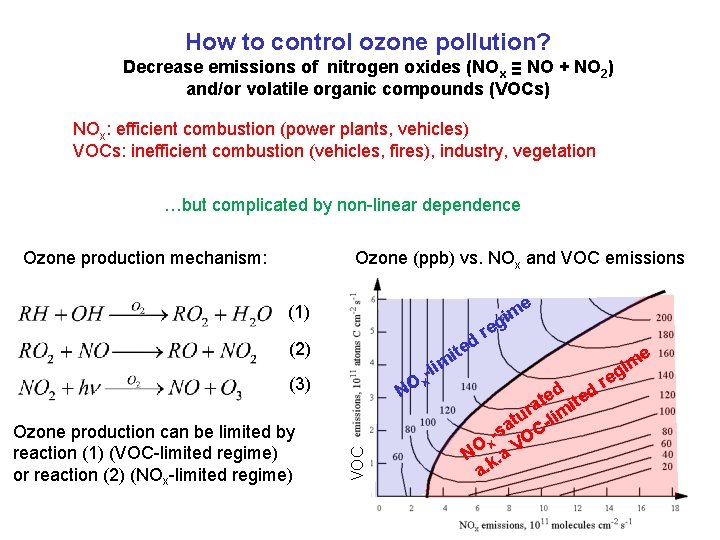
How to control ozone pollution? Decrease emissions of nitrogen oxides (NOx ≡ NO + NO 2) and/or volatile organic compounds (VOCs) NOx: efficient combustion (power plants, vehicles) VOCs: inefficient combustion (vehicles, fires), industry, vegetation …but complicated by non-linear dependence Ozone production mechanism: Ozone (ppb) vs. NOx and VOC emissions e (1) ite (2) (3) VOC Ozone production can be limited by reaction (1) (VOC-limited regime) or reaction (2) (NOx-limited regime) NO m -li x d m gi re e e d ed r e at mit r tu -li a -s OC x NO k. a V a. m gi
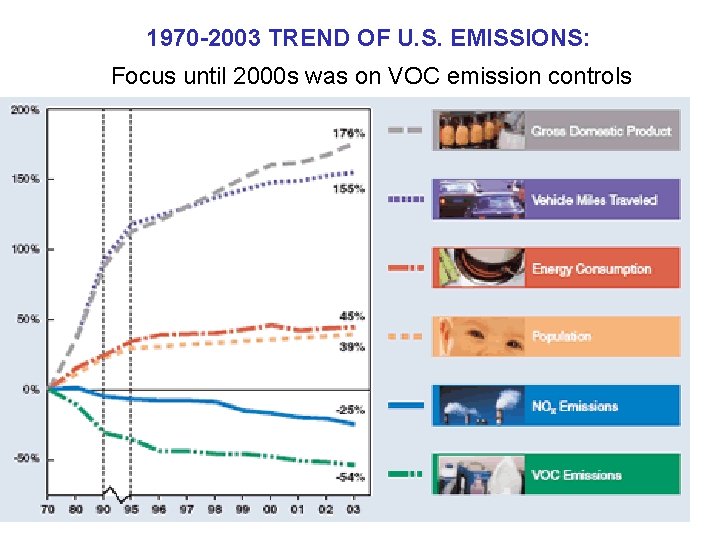
1970 -2003 TREND OF U. S. EMISSIONS: Focus until 2000 s was on VOC emission controls
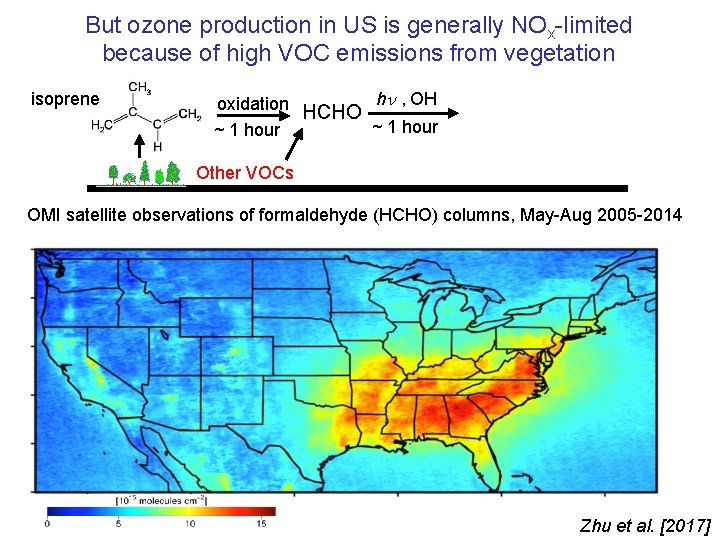
But ozone production in US is generally NOx-limited because of high VOC emissions from vegetation isoprene oxidation HCHO h , OH ~ 1 hour Other VOCs OMI satellite observations of formaldehyde (HCHO) columns, May-Aug 2005 -2014 Zhu et al. [2017]
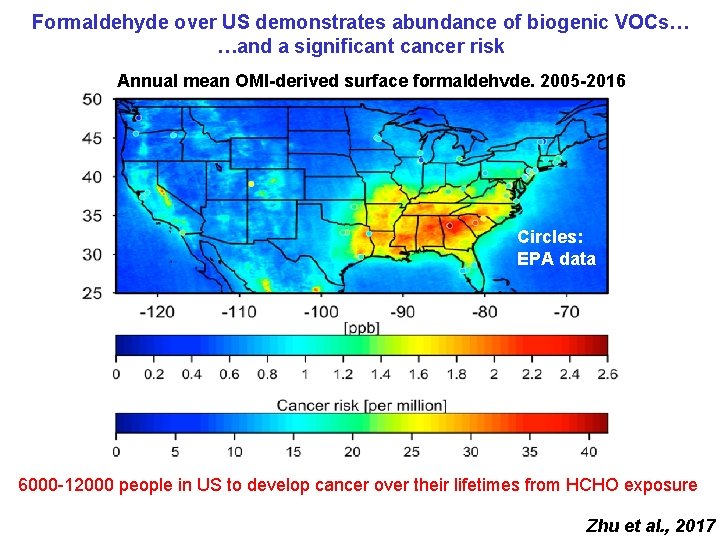
Formaldehyde over US demonstrates abundance of biogenic VOCs… …and a significant cancer risk Annual mean OMI-derived surface formaldehyde, 2005 -2016 Circles: EPA data 6000 -12000 people in US to develop cancer over their lifetimes from HCHO exposure Zhu et al. , 2017
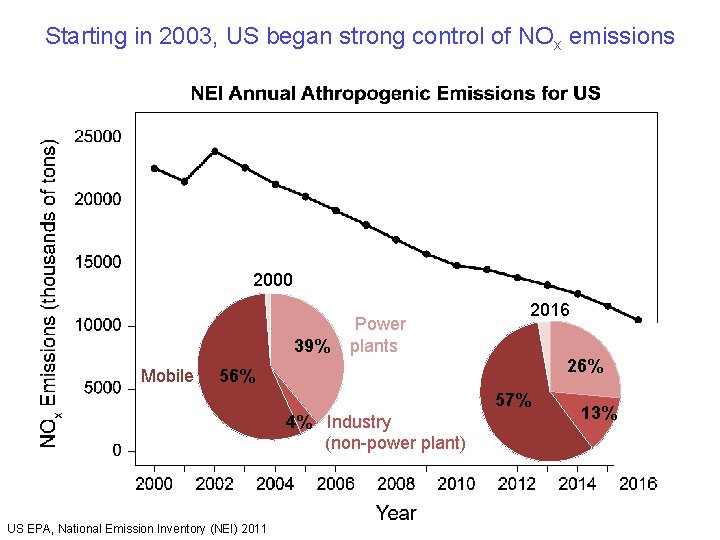
Starting in 2003, US began strong control of NOx emissions 2000 39% Mobile Power plants 2016 26% 57% 4% Industry (non-power plant) US EPA, National Emission Inventory (NEI) 2011 13%
![…and ozone has responded! Silvern et al. [2019] …and ozone has responded! Silvern et al. [2019]](http://slidetodoc.com/presentation_image_h2/91a66b2471c935dee4892e40e34efee9/image-24.jpg)
…and ozone has responded! Silvern et al. [2019]
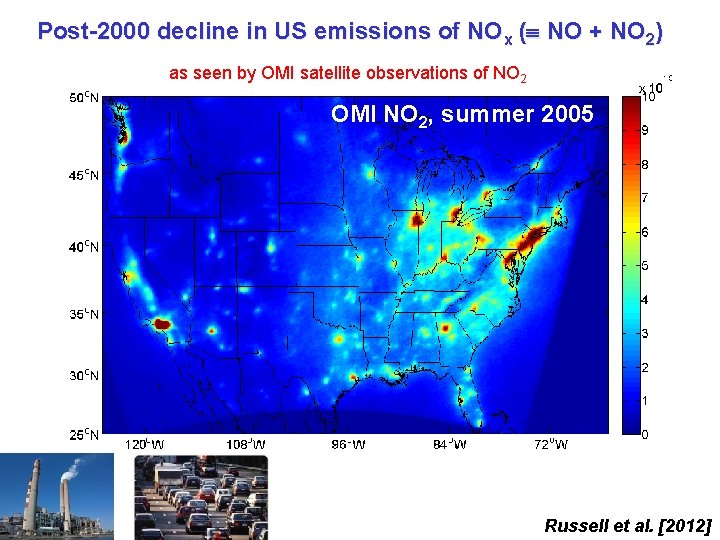
Post-2000 decline in US emissions of NOx ( NO + NO 2) as seen by OMI satellite observations of NO 2 OMI NO 2, summer 2005 Russell et al. [2012]
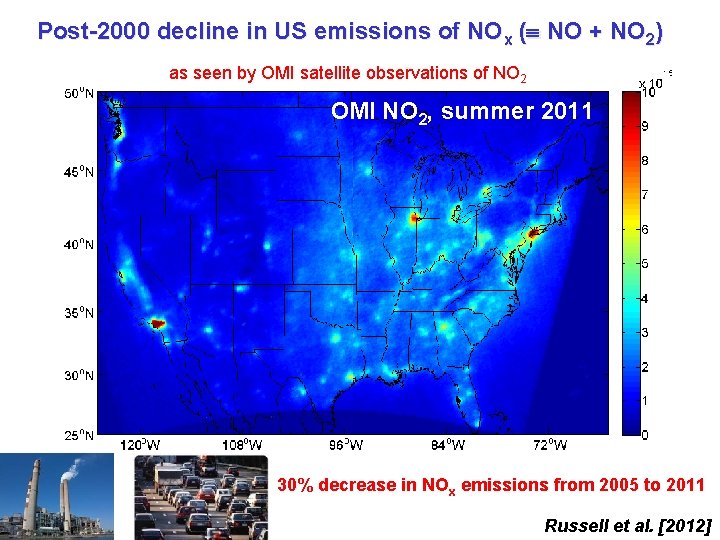
Post-2000 decline in US emissions of NOx ( NO + NO 2) as seen by OMI satellite observations of NO 2 OMI NO 22, summer 2011 2005 30% decrease in NOx emissions from 2005 to 2011 Russell et al. [2012]
![OMI annual NO 2 trends over CONUS, 2005 -2017 Silvern et al. [2019] OMI annual NO 2 trends over CONUS, 2005 -2017 Silvern et al. [2019]](http://slidetodoc.com/presentation_image_h2/91a66b2471c935dee4892e40e34efee9/image-27.jpg)
OMI annual NO 2 trends over CONUS, 2005 -2017 Silvern et al. [2019]
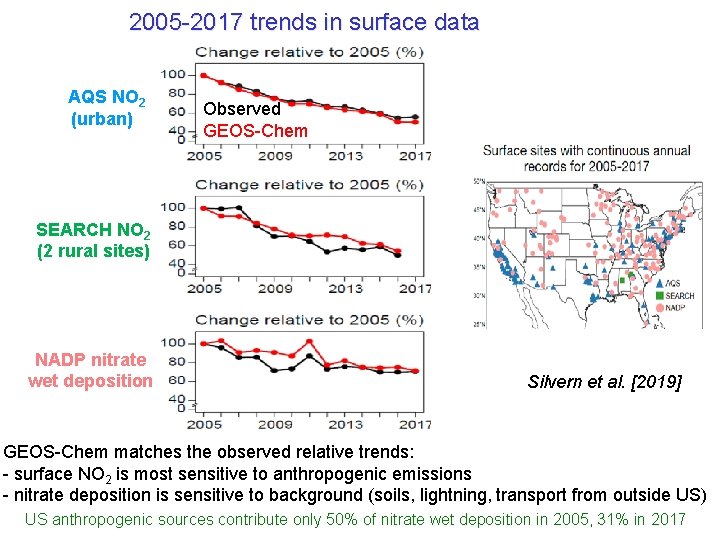
2005 -2017 trends in surface data AQS NO 2 (urban) Observed GEOS-Chem SEARCH NO 2 (2 rural sites) NADP nitrate wet deposition Silvern et al. [2019] GEOS-Chem matches the observed relative trends: - surface NO 2 is most sensitive to anthropogenic emissions - nitrate deposition is sensitive to background (soils, lightning, transport from outside US) US anthropogenic sources contribute only 50% of nitrate wet deposition in 2005, 31% in 2017
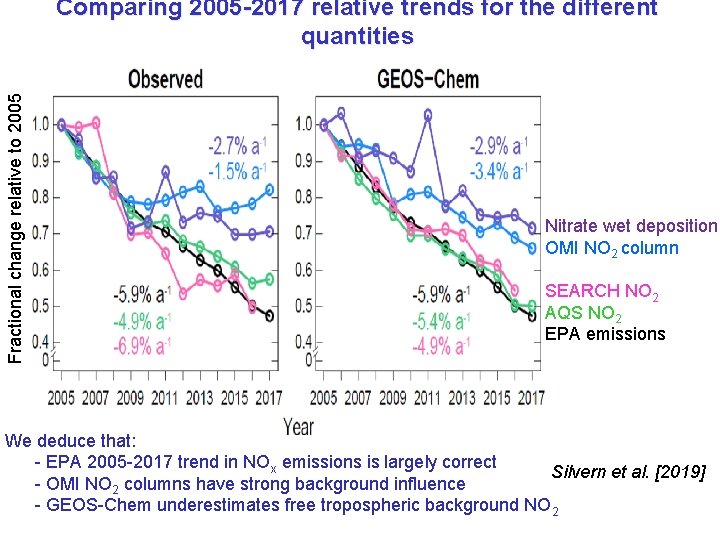
Fractional change relative to 2005 Comparing 2005 -2017 relative trends for the different quantities Nitrate wet deposition OMI NO 2 column SEARCH NO 2 AQS NO 2 EPA emissions We deduce that: - EPA 2005 -2017 trend in NOx emissions is largely correct Silvern et al. [2019] - OMI NO 2 columns have strong background influence - GEOS-Chem underestimates free tropospheric background NO 2
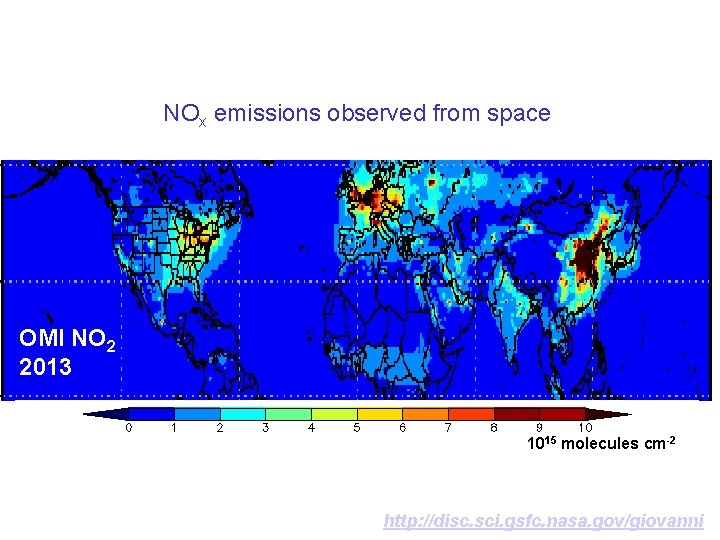
NOx emissions observed from space OMI NO 2 2013 1015 molecules cm-2 http: //disc. sci. gsfc. nasa. gov/giovanni
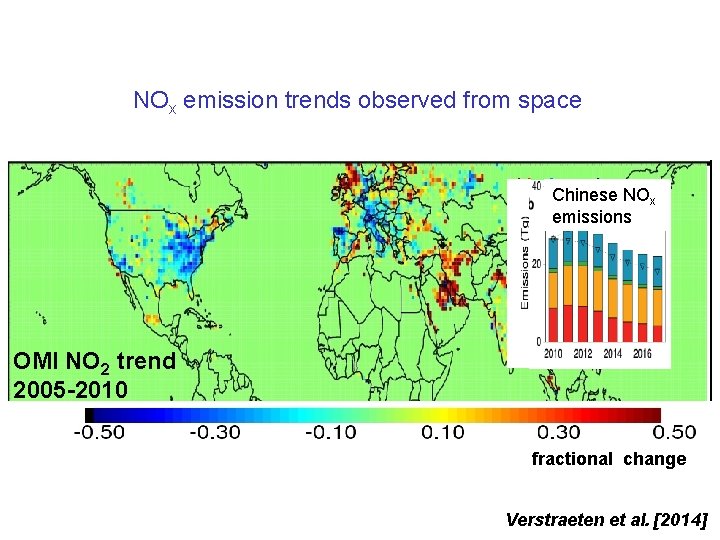
NOx emission trends observed from space Chinese NOx emissions OMI NO 2 OMI 2013 NO 2 trend 2005 -2010 1015 molecules cm-2 s-1 fractional change Verstraeten et al. [2014]
![Trend in #days/year with ozone > 70 ppb, summer 2000 -2014 TOAR [2017] Trend in #days/year with ozone > 70 ppb, summer 2000 -2014 TOAR [2017]](http://slidetodoc.com/presentation_image_h2/91a66b2471c935dee4892e40e34efee9/image-32.jpg)
Trend in #days/year with ozone > 70 ppb, summer 2000 -2014 TOAR [2017]
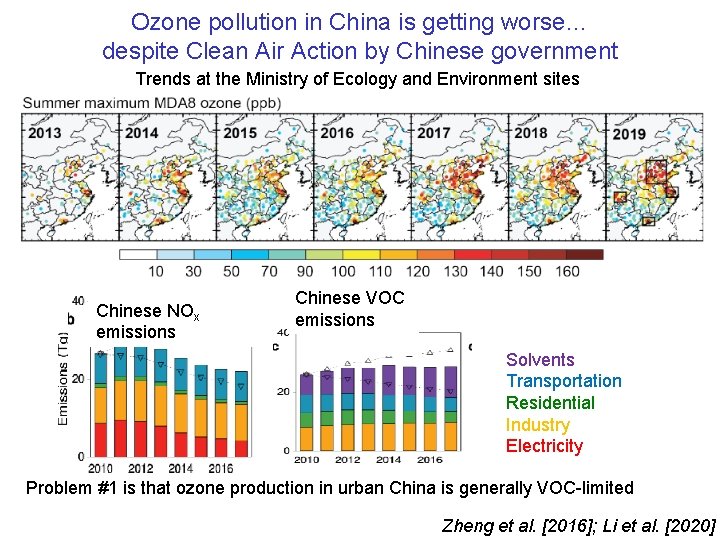
Ozone pollution in China is getting worse… despite Clean Air Action by Chinese government Trends at the Ministry of Ecology and Environment sites Chinese NOx emissions Chinese VOC emissions Solvents Transportation Residential Industry Electricity Problem #1 is that ozone production in urban China is generally VOC-limited Zheng et al. [2016]; Li et al. [2020]
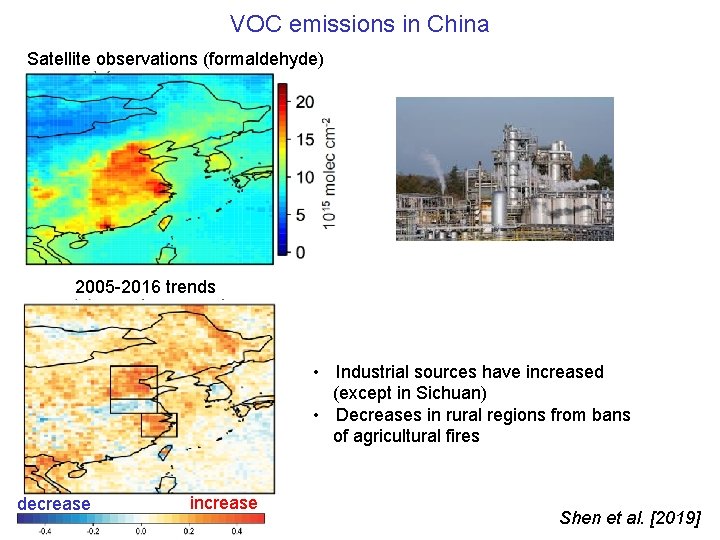
VOC emissions in China Satellite observations (formaldehyde) 2005 -2016 trends • Industrial sources have increased (except in Sichuan) • Decreases in rural regions from bans of agricultural fires decrease increase Shen et al. [2019]
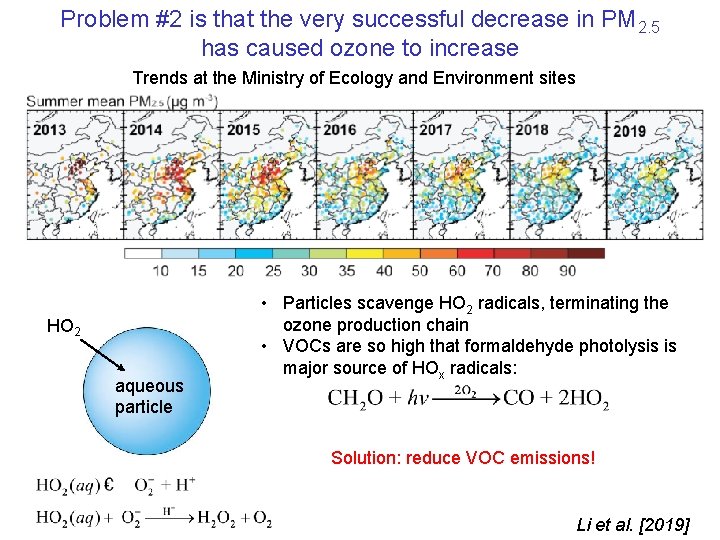
Problem #2 is that the very successful decrease in PM 2. 5 has caused ozone to increase Trends at the Ministry of Ecology and Environment sites HO 2 aqueous particle • Particles scavenge HO 2 radicals, terminating the ozone production chain • VOCs are so high that formaldehyde photolysis is major source of HOx radicals: Solution: reduce VOC emissions! Li et al. [2019]
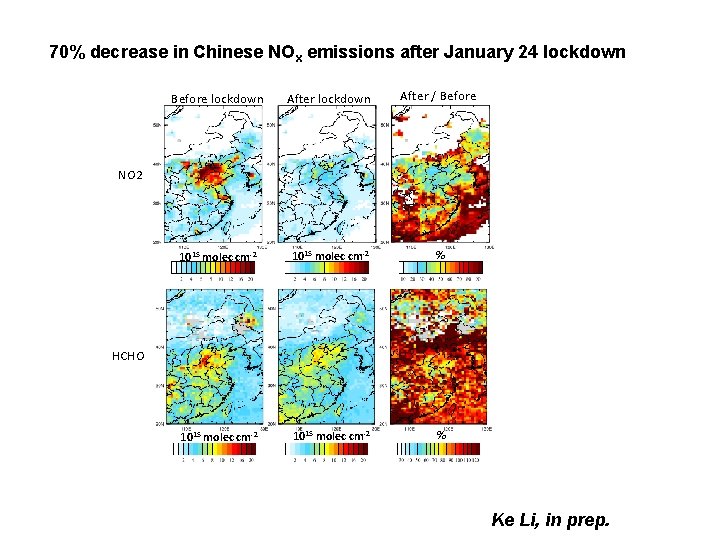
70% decrease in Chinese NOx emissions after January 24 lockdown Before lockdown After / Before 1015 molec cm-2 % NO 2 HCHO Ke Li, in prep.
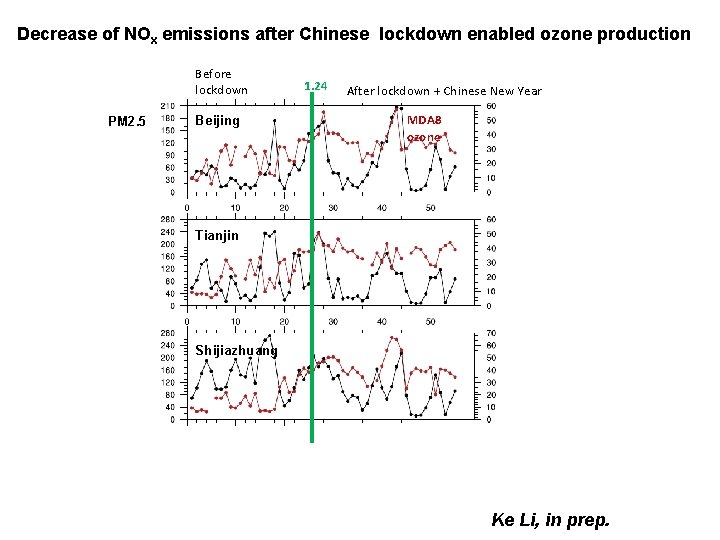
Decrease of NOx emissions after Chinese lockdown enabled ozone production Before lockdown PM 2. 5 Beijing 1. 24 After lockdown + Chinese New Year MDA 8 ozone Tianjin Shijiazhuang Ke Li, in prep.
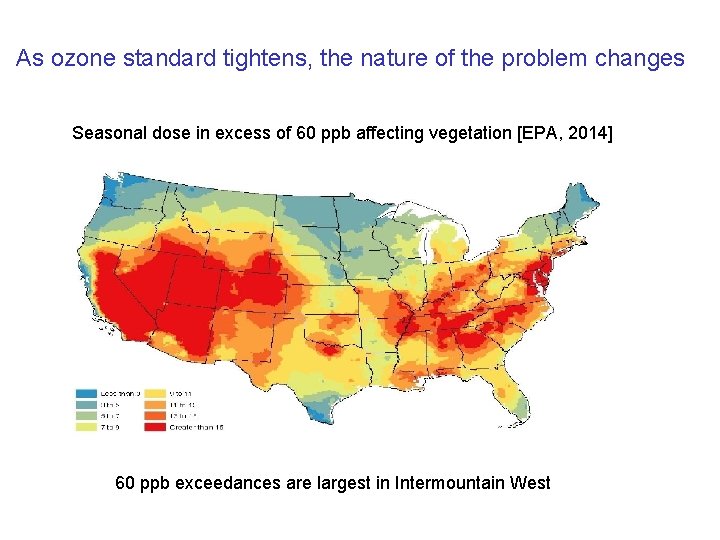
As ozone standard tightens, the nature of the problem changes Seasonal dose in excess of 60 ppb affecting vegetation [EPA, 2014] 60 ppb exceedances are largest in Intermountain West
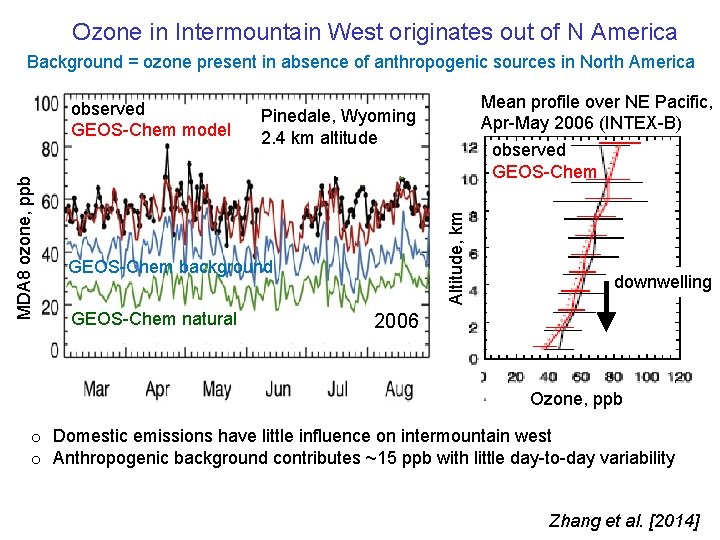
Ozone in Intermountain West originates out of N America Background = ozone present in absence of anthropogenic sources in North America Pinedale, Wyoming 2. 4 km altitude GEOS-Chem background GEOS-Chem natural Mean profile over NE Pacific, Ozone over Pacific Apr-May 2006 NE (INTEX-B) (INTEX-B, Apr-May 2006) observed GEOS-Chem Altitude, km MDA 8 ozone, ppb observed GEOS-Chem model downwelling 2006 Ozone, ppb o Domestic emissions have little influence on intermountain west o Anthropogenic background contributes ~15 ppb with little day-to-day variability Zhang et al. [2014]
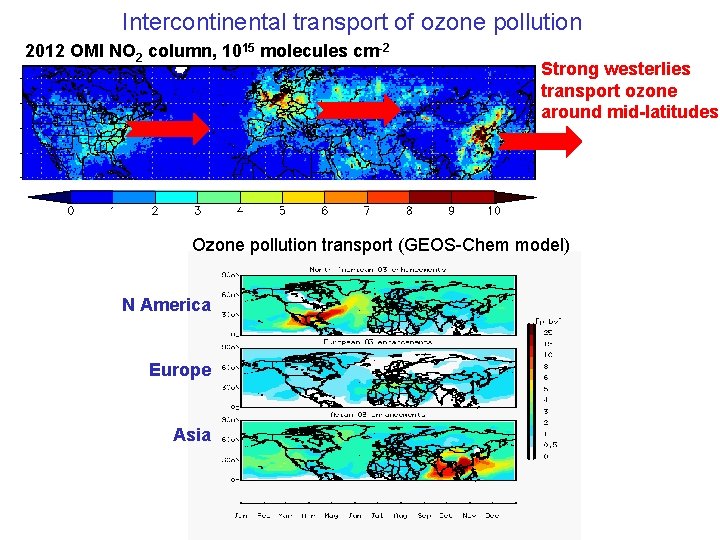
Intercontinental transport of ozone pollution 2012 OMI NO 2 column, 1015 molecules cm-2 Strong westerlies transport ozone around mid-latitudes Ozone pollution transport (GEOS-Chem model) N America Europe Asia
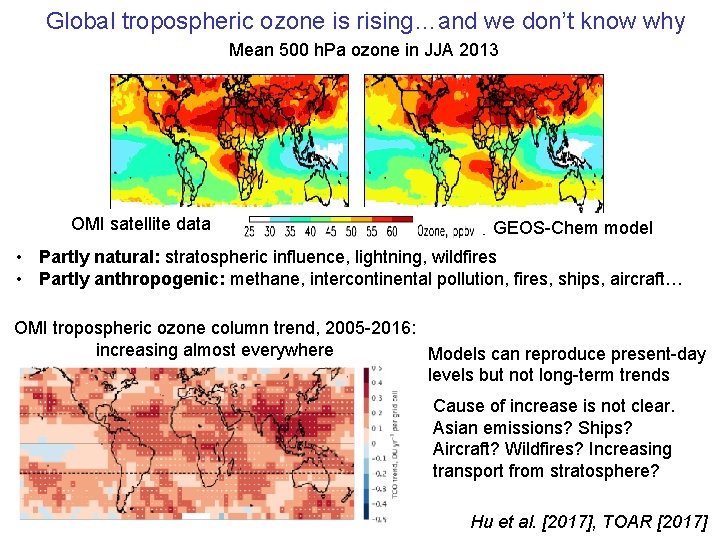
Global tropospheric ozone is rising…and we don’t know why Mean 500 h. Pa ozone in JJA 2013 OMI satellite data GEOS-Chem model • Partly natural: stratospheric influence, lightning, wildfires • Partly anthropogenic: methane, intercontinental pollution, fires, ships, aircraft… OMI tropospheric ozone column trend, 2005 -2016: increasing almost everywhere Models can reproduce present-day levels but not long-term trends Cause of increase is not clear. Asian emissions? Ships? Aircraft? Wildfires? Increasing transport from stratosphere? Hu et al. [2017], TOAR [2017]
- Slides: 41