The in vitro chromosome aberration assay is very

The in vitro chromosome aberration assay is very sensitive to chemical mutagens Note lack of cytotoxicity
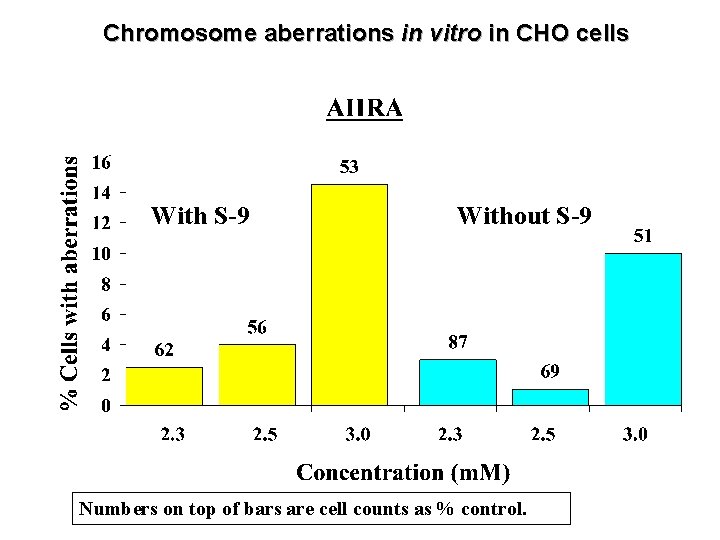
Chromosome aberrations in vitro in CHO cells With S-9 Without S-9 Numbers on top of bars are cell counts as % control.

Negative genotoxicity assays of Angiotensin II receptor antagonist and analogs ¨ Microbial mutagenicity in Salmonella and E. Coli ¨ Alkaline elution (DNA strand breaks) in rat hepatocytes ¨ Mammalian cell mutagenicity; hprt in V 79 cells ¨ In vivo: l Chromosome abs in M & F mice (1500 mg/kg) l Alkaline elution in F rat liver (1978 mg/kg) ¨ Additional negative tests on analogs: l DNA adducts in calf thymus DNA l ‘phage DNA fragmentation l unscheduled DNA synthesis
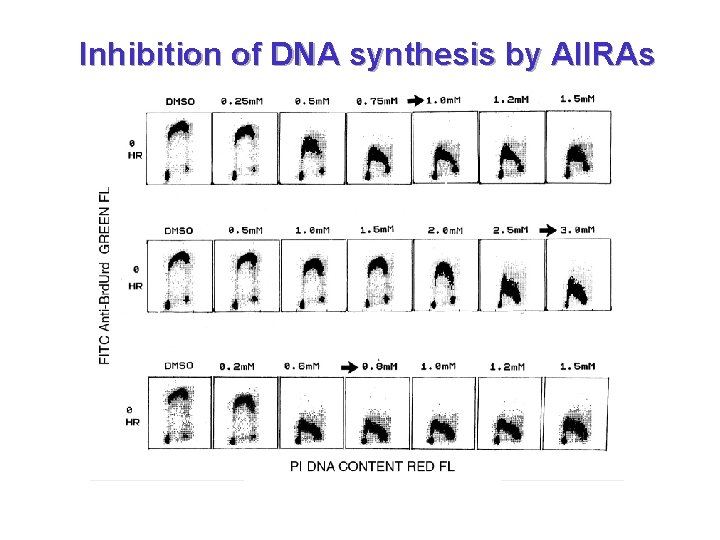
Inhibition of DNA synthesis by AIIRAs

Significance of aberrations associated with cytotoxicity ¨ Potential threshold l l l no structural alert no mutation no DNA binding ¨ no aberrations in vivo ¨ likely secondary to toxicity, not relevant at human exposure levels l l l In vitro positive at 1400 µg/ml In vivo: mouse exposure at 500 mg/kg/d x 8 ®C Max 74 µg/ml, AUC 90 µg/ml. hr Human 40 mg oral: ®C Max 0. 2 µg/ml, AUC 0. 4 µg/ml. hr

Mechanisms of chromosome aberrations ¨ Aberrations result from DNA strand breaks ¨ Strand breaks induced by e. g. , l l ionizing radiation processing of lesions/adducts/abnormal bases ¨ but also by l l l inhibition of DNA synthesis inhibition of topoisomerases Cytotoxicity ¨ Mis-incorporation, altered bases l nucleoside analogs; ribonucleotide reductase inhibitors; folate antagonists ®DNA synthesis inhibition; chain termination; mis-pairs, gaps;

Mechanisms of genotoxicity that may have threshold ¨ Disruption of cell division ¨ Disruption of chromosome segregation ¨ Inhibition of DNA synthesis ¨ Inhibition of topoisomerases ¨ Nucleotide pool imbalance ¨ Overloading of oxidative defence mechanisms ¨ Metabolic overload (phase II enzymes etc) ¨ Ion chelation; disturbance of metal homeostasis ¨ Extremes of p. H/osmolality ¨ Cytotoxicity (adapted from Henderson, Albertini & Aardema, Mutat Res 464, 123 -128, 2000)

Possible mechanisms for chromosome aberration induction by nucleoside analogs ¨ DNA synthesis inhibition ¨ incorporation l chain termination l attempted repair of mispaired bases ¨ pool imbalance l DNA synthesis inhibition l polymerase errors- attempted repair
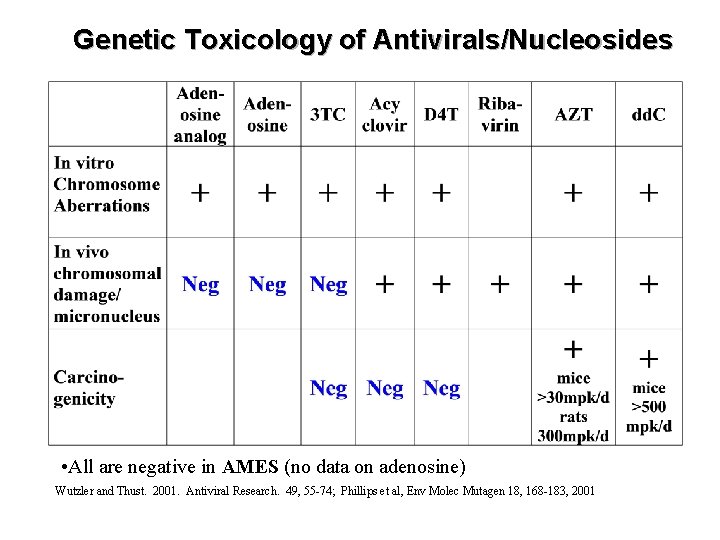
Genetic Toxicology of Antivirals/Nucleosides • All are negative in AMES (no data on adenosine) Wutzler and Thust. 2001. Antiviral Research. 49, 55 -74; Phillips et al, Env Molec Mutagen 18, 168 -183, 2001

Risk evaluation for nucleoside analog ¨ Specificity for viral vs mammalian enzyme ¨ Is it incorporated into DNA? RNA? ¨ If incorporated is it pro-mutagenic? (AZT) ¨ If not incorporated: High dose effect. ¨ Possible mechanisms l pool imbalance l DNA synthesis inhibition (try to restore) (demonstrate) ¨ Genotoxicity may have threshold ® in vivo cytogenetic assay negative ® good safety margin (exposure) ¨ May not be genotoxic risk to humans.
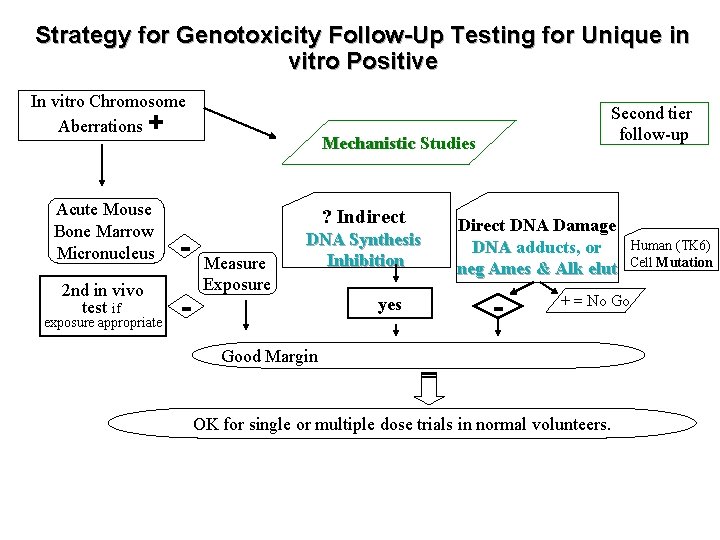
Strategy for Genotoxicity Follow-Up Testing for Unique in vitro Positive In vitro Chromosome Aberrations + Acute Mouse Bone Marrow Micronucleus - 2 nd in vivo test if - exposure appropriate Second tier follow-up Mechanistic Studies ? Indirect Measure Exposure DNA Synthesis Inhibition yes Direct DNA Damage DNA adducts, or neg Ames & Alk elut - Human (TK 6) Cell Mutation + = No Go Good Margin OK for single or multiple dose trials in normal volunteers.

Risk evaluation when mechanism is indirect ¨ Chromosome aberrations may be induced only above a threshold dose. ¨ Therefore if: l in vivo cytogenetic assay negative l second in vivo assay negative l good safety margin (exposure) ¨ May not be genotoxic risk to humans.
- Slides: 12