The hydrostatic equation and its applications Let us
























- Slides: 26

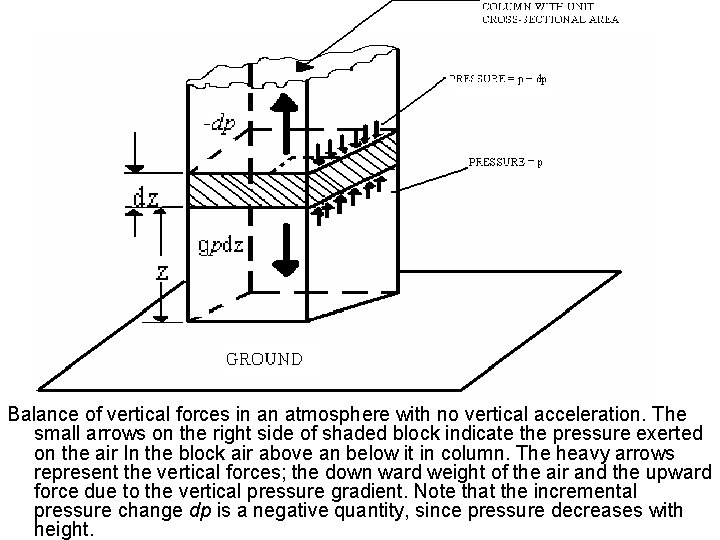
The hydrostatic equation and its applications • Let us consider a vertical column of air with unit cross-sectional area. • The mass of air between heights z and z = dz in the column is p dz, where is density of the air at height z. the force acting on this column due to the weight of the air is gp dz, where g is the acceleration due to gravity at height z. • Now let us consider the net vertical force on the block due to the pressure of the surrounding air. Will assume that in going from height z to height z + dz the pressure changes by amount dp.

Balance of vertical forces in an atmosphere with no vertical acceleration. The small arrows on the right side of shaded block indicate the pressure exerted on the air In the block air above an below it in column. The heavy arrows represent the vertical forces; the down ward weight of the air and the upward force due to the vertical pressure gradient. Note that the incremental pressure change dp is a negative quantity, since pressure decreases with height.

• Since we know that pressure decreases with height, dp must be a negative quantity, and the upward pressure on the lower face of the shaded block must be slightly greater than the downward pressure on the upper face of the block. • Thus the net vertical force on the block due to the vertical gradient of pressure is upward and given by the positive quantity – dp as indicated in the figure. The balance of forces in the vertical requires that • Or • Equation 2 is termed the hydrostatic equation. It should be noted that the negative sign in equation 2 ensures that the pressure decreases with increasing height. Since ρ = 1/α, equation 2 can be rearranged to give

• If pressure at height z is p(z), we have, from equation 2, • Or since p ( ) – 0, • That is, the pressure at level z is equal to the weight of the air in the vertical column of unit cross-sectional area lying above that level. If the mass of earth’s atmosphere were uniformly distributed over the globe, the pressure at sea level would be 1013 mb, or 1. 013 × 10 PA, which is referred to a normal atmosphere pressure and abbreviated as 1 atm.

Virtual temperature • Moist air has a lower apparent molecular weight than dry air. Therefore, the gas constant for 1 kg of moist air is larger than that for 1 kg of dry air. • However, rather than use a gas constant for 1 kg of moist air, the exact value of which would depend o the amount of water vapor in the air , it is more convenient to retain the gas constant for dry air and use a fictitious temperature ( called the virtual temperature) in the ideal gas equation. • We can derive an expression for the virtual temperature in the following way. • Consider a volume V of moist air at temperature T and the total pressure P which contains mass m of dry air and mass m of water vapor. The density of P of the moist air is given by

• Where ρ is the density which the same mass of dry air would have if it alone occupied the volume V and ρ the density which the same mass of water vapor would have if it alone occupied the volume V. we may call these partial densities. At first glance it might appear from that the density of moist air is grater than that of dry air. This is not the case since the partial density ρ is less than that of dry air. Applying the ideal gas equation in the form the water vapor and dry air in turn, we have • And • Where e and p are the partial pressures exerted by the water vapor and the dry air, respectively.

• Also, from Dalton’s law of partial pressures, • Combining the last three equations, • Or • Where ε is defined and the last equation may be written as • Where

• T is called the virtual temperature. if this fictitious temperature, rather than the actual temperature, is used for moist air, the total pressure p and density ρ of the, moist air are related by the ideal gas equation with the gas constant the same as that for a unit mass of dry air (R). • It follows that the virtual temperature is the temperature that dry air must have in order to have the same density as moist air at the same pressure. • Moist air is less dense than dry air: therefore, the virtual temperature is always greater than actual temperature. However, even for very warm, most air the virtual temperature exceeds the actual temperature by only few degrees.

Geopotential • The geopotential Φ at any point in the atmosphere is defined as the work that must be done against the earth’s gravitational field in order to raise a mass of 1 kg from sea level to that point. • In other words, Φ is the gravitational potential for unit mass. The units of geopotential are J kg or m s. the force (in Newton’s) acting on 1 kg at height z above sea level is numerically equal to g. the work ( in joules) in raising 1 kg from z to z + dz is g dz; therefore, • The geopotential Φ(z) at height z is thus given by • Where the geopotential Φ (0) at sea level (z = 0) has, by convention, been taken as zero.

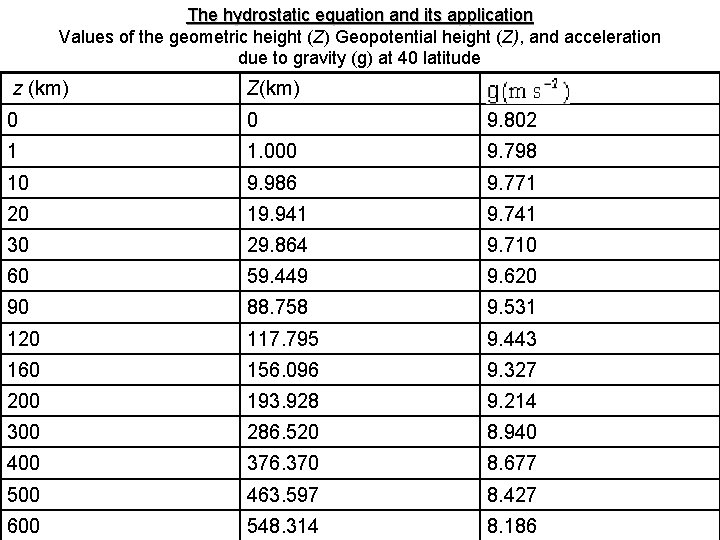
• Where the geopotential Φ (0) at sea level (z = 0) has, by convention, been taken as zero. It should be emphasized that the geopotential at a particular point in the atmosphere depends only on the height of that point and not on the path through which the unit mass is taken in reaching that point. The work done in taking a mass of 1 kg from point A with geopotential Φ to point B with geopotential Φ is Φ - Φ. • We can also define a quantity called the geopotential height Z as • where go is the globally averaged acceleration due to gravity at the earth’s surface (taken as 9. 8 m s ). Geopotential height is used as the vertical coordinate in most atmosphere applications in which energy plays an I portent role ( for example, the largescale motions discussed in chapter 8). It can be seen from table 2. 1 that the values of z and Z are almost the same in the lower atmosphere where g g.

The hydrostatic equation and its application Values of the geometric height (Z) Geopotential height (Z), and acceleration due to gravity (g) at 40 latitude z (km) Z(km) 0 0 9. 802 1 1. 000 9. 798 10 9. 986 9. 771 20 19. 941 9. 741 30 29. 864 9. 710 60 59. 449 9. 620 90 88. 758 9. 531 120 117. 795 9. 443 160 156. 096 9. 327 200 193. 928 9. 214 300 286. 520 8. 940 400 376. 370 8. 677 500 463. 597 8. 427 600 548. 314 8. 186

• In meteorological practice it is not convenient to deal with density p, which cannot be measured directly. By making use of (12) or (16) to eliminate p in (18), we obtain • Rearranging the last expression and using (20), • If we now integrate between levels p 1 and p geopotential Φ 1 and Φ , respectively, , with • Dividing both sides of the last equation by go and reversing the limits of integration yields

Scale height and hypsometric equation • For an isothermal (temperature constant with height) and dry atmosphere, ( 24) become • Or • Where • H is called the scale height. If Z 2 – Z 1 is set successfully equal to 0, H, 2 H, 3 H, …. , p 1 / p 2 is equal to 1, exp ( -1), exp ( -2), exp( -3), ……. That is, the pressure decrease by a factor e (= 2. 7180 for each increase H in geopotential height. It should be noted that (2. 26) is equivalent to (1. 8) which was derived empirically in section 1. 3. 1.

• The temperature of the atmosphere generally varies with height. In this case (2. 24) may be integrated if we define a mean virtual temperature T with respect to in p as shown in fig. 2. 2. That is, • Then, from (24) and (28) • Whether scale height H is now defined as • Equation ( 2. 29) is called the hypsometric equation

2. 24 Reduction of pressure to sea level • In mountainous regions the difference in surface pressure from one station to another largely due to differences in elevation. In order to isolate that part of the pressure field which is due to the passage of weather systems, it is necessary to reduce the pressures to a common reference level. For this purpose, sea level is normally used. • Let the subscripts g and 0 refer to conditions at the grounds and at sea level (Z = 0), respectively. For the layer between the earth’s surface and sea level the hypsometric equation (2. 29) assumes the form

which can be solved to obtain the sea level pressure • Where (2. 30) has been used to obtain the last expression which shows how the sea level pressure depends on the mean virtual temperature between ground and sea level. If Z is small, the scale height can be evaluated from the ground temperature. Also, if Z / << 1, the exponential in (2. 31) can be approximated by 1 + Z /. Since 8 km for the observed range of ground temperatures on earth, this approximation is satisfactory provided that Z is less than a few hundred meters. With this approximation, (2. 31) becomes

• Since P 1000 mb and 8 km, the pressure correction (in millibars) is roughly equal to Z ( in meters) divided by 8. in other words, near sea level the pressure falls about 1 mb for every 8 m of vertical ascent. • Whrn Z is on the order of 1 km or greater, there is difficulty in estimating what the mean virtual temperature of the layer would be in the absence of topography. In practice, a number of empirical corrections are applied to the surface temperature in the estimation of the scale height. These procedures are not entirely satisfactory in eliminating the effects of topography. Therefore, sea level pressure analysis in mountainous regions still leaves much to be desired.


Specific heats • Supposes a small quantity of heat dp is given to a unit mass of material and, as a consequence, its temperature increase from T to T + d. T without a phase change occurring. The ratio dp / d. T is called the specific heat of the material. However, the specific heat defined in this way can have any number of values, depending on how the materials change as it receives the heat. If the volume of the material is kept constant, a specific heat a constant volume c is defined which is given by

• But if the specific volume is constant (2. 42) becomes dp = du: therefore, ’ • But for an ideal gas joule’s law applies and therefore u depends upon temperature alone and we may write • Therefore, from (2. 41) and (2. 43), the first law of thermodynamics for an ideal gas can be written in the form • We may also define a specific heat at constant pressure c as

• Where the material is allowed to expand as the heat is added and its temperature rises but its pressure is kept constant. In this case, a certain amount of the heat added will have to be expended to do work as the material expands against the constant pressure of the environment. Therefore, larger quantity of heat must be added to the material to raise its temperature by a give amount than if the volume of the materials were kept constant. This can be seen mathematically in the following way. The equation can be written as • making use of the ideal gas equation, this becomes

• At constant pressure, the last term vanishes; therefore, from. • The specific heats at constant volume and at constant pressure for dry air are 71. 7 and 1004 J deg kg , respectively, and the difference between them is 287 j deg kg , which is numerically equal to the gas constant for dry air. For an ideal monatomic gas c : R = 5: 3: 2 ad for and ideal diatomic gas c : R = 7: 5: 2. • By combining both we obtain an alternate form of the first law of thermodynamics:

The adiabatic lapse rate • We will now derive an expression for the rate of change of temperature with height of parcel of dry air which moves about in the earth’s atmosphere while always satisfying the conditions listed above. Since the air parcel undergoes only adiabatic transformations • (dp = 0), and the atmosphere is in hydrostatic equilibrium, for a unit mass of air in the parcel we have, from • Dividing through by dz and making use of it we obtain

• where Ґ is called the dry adiabatic lapse rate. Since an air parcel expands as it rises in the atmosphere, its temperature will decrease with height so that Ґ as defined, is a positive quantity. Substituting g = 9. 81 m s and c = 1004 j kg deg into gives Ґ = 0. 0098 deg m or 9. 8 deg km. • It should be emphasized again that Ґ is the rate of change of temperature following a parcel of dry air which is being raised or lowered adiabatically. The actual lapse rate of temperature( which we will indicate by Ґ) in the atmosphere, as measure by a radiosonde averages 6 -7 deg km in the troposphere but it takes on a wide range of values at individual locations.

Potential temperature • The potential temperature θ of an air parcel is defined as the temperature which the parcel of air would have if it were expanded or compressed adiabatically from its existing pressure and temperature to a standard pressure po (generally taken as 1000 mb). • We can derive an expression for the potential temperature of air on terms of its pressure p, temperature T, and the standard pressure Po as follows. • For an adiabatic transformation (dp = 0), becomes

• Combining this and rearranging terms yields • Integrating upward from Po (where T = θ) to p, we obtain