The Hybrid Capture 2 hc 2 System HPV

The Hybrid Capture 2 (hc 2) System HPV DNA Test by Digene by Brenda Palacios

Objectives • • • State the 2 main low-risk HPV DNA types State the 2 main high-risk HPV DNA types State what type of test method is used for the detection of HPV • State the minimum sample volume required for testing • State what the molecular sandwich consist of

Human Papillomavirus (HPV) • Primary etiological agent in cervical cancer • 2 nd most common type of cancer in women world wide • 3 rd leading cause of cancer-related deaths in women worldwide • Most common viral sexually transmitted infection, that goes undiagnosed due to no symptoms developed

HPV Characteristics • Non-enveloped double-stranded DNA virus • Epitheliotrophic- has great affinity for epithelial cells • Obligatory intracellular parasites that deliver their genome and accessory proteins into host cells for viral replication
HPV Infection • E 6 oncogene binds to p 53 protein in host cell – p 53 protein is a negative regulator • E 6 protein mutates p 53 protein removing its protective function • Mutation disables p 53 gene switch, permitting cell to multiply uncontrolled

HPV Types • Types 6 &11–most common low-risk HPV – Associated with genital warts – Rarely found in cervical cancer • Types 16 & 18 - most common high-risk HPV – Associated with cancers of the cervix, vagina, vulva, anus, and penis
HPV Detection • Traditional screening by Papanicolau (pap smear) test • Used universally for initial detection of intraepithelial abnormalities www. unsw. edu. au/. . . /sep/cells_image_inside. jp g • In case of atypical squamous cells of undetermined significance HPV DNA detection is recommended

Molecular Testing • Hybrid Capture 2 (hc 2) System HPV DNA Test – Approved by FDA • Nucleic acid hybridization assay • No target DNA amplification www. clpmag. com/graphics/mags/0409/sl 03. jpg • Single amplification using microplate chemiluminescence for qualitative detection of 18 types of HPV

Molecular Testing cont. • Differentiates between low and high risk HPV – Low-risk HPV: 6, 11, 42, 43, 44 – High-risk HPV: 16, 18, 31, 35, 39, 45, 51, 52, 56, 58, 59, 68 • Does not determine specific HPV genotype

Controls and Reagents • Controls – High-risk control: cloned HPV 16 DNA – Low-risk control: cloned HPV 6 DNA • Negative control – Carrier DNA • Calibrators (run in triplicate) – Low-risk Calibrator: Cloned HPV 11 DNA – High-risk Calibrator: cloned HPV 16 DNA – Ensure that the reagents and calibrator materials are functioning properly, for determination of assay cut-off value

Controls and Reagents cont. • Probes – Low-risk Probe: HPV 6, 11, 42, 43, 44 RNA cocktail – High-risk Probe: HPV 16, 18, 31, 33, 35, 39, 45, 51, 52, 58, 59, 68, RNA cocktail • Capture Microplate – Coated with goat polyclonal anti-RNA: DNA hybrid antibodies • Detection Reagent 1 – Alkaline phosphatase-conjugated murine monoclonal antibodies to RNA: DNA hybrids • Detection Reagent 2 – Chemiluminescent substrate

Specimen Requirements • Cervical specimens collected using a broom type collection device placed in Preserv. Cyt Solution • Cervical biopsies btw 2 -5 mm in Digene Specimen Transport Medium • Specimens collected with the Digene Cervical Sampler, placed in Sure. Path Preservative Fluid

Denaturation Process • Samples are mixed with Sample Conversion buffer • Then mixed with a 2: 1 ratio of Specimen Transport Medium (STM) and Denaturation Reagent (DNR) – STM-preservative that retards bacterial growth and retains DNA integrity – DNR-dilute sodium hydroxide solution, which lysis cells and denatures HPV DNA

Detection • The Hybrid Capture 2 is a fluid-based molecular hybridization assay • Does not use HPV DNA target amplification • Uses hybridized signal amplification • Denatured HPV DNA is hybridized with low-risk or high-risk RNA probe • Resulting hybrids are captured on microplate wells by immobilized antibody

Detection • Detection is sandwich-style with a second anti-RNA: DNA hybrid conjugated to alkalinephosphatase • Bound alkaline-phosphatase is revealed by addition of a chemiluminescent dioxetanebased substrate • Substrate is cleaved by bound alkaline phosphate and emitted light is measured in a microplate luminometer
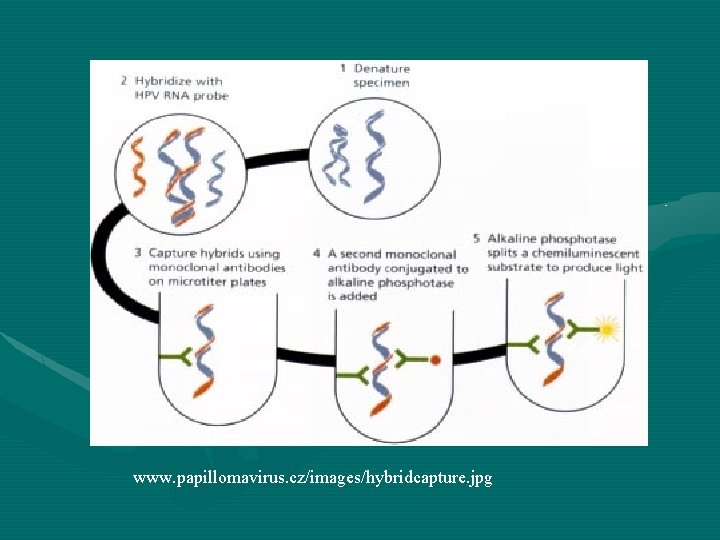
www. papillomavirus. cz/images/hybridcapture. jpg

Test Interpretation • Emitted light is measured in Relative Light Units (RLUs) – Specimens with RLU/CO ratio ≥ 1. 7 with lowrisk HPV probe are considered positive for lowrisk HPV – Specimens with RLU/CO ratio ≥ 1. 7 with highrisk HPV probe are considered positive for highrisk HPV – Specimens with RLU/CO ratio btw 0. 80 -1. 7 are considered indeterminate for either low-risk or high-risk HPV and must be repeated

Limitations • Significant number of false-positives (10%19%) due to cross reactivity with low-risk HPV DNA • HPV DNA not amplified – Negative predicted value may be compromised in cases in which HPV DNA copy number is low • No internal control used for sample sufficiency – Not possible to determine if results are due to insufficient DNA or true negative • Large number of inconclusive results

Sources of Error • Large concentrations of whole blood, douche, anti-fungal cream, and contraceptive jelly – May cause false-negative • Contamination of Capture Microplate and Detection Reagent 2 with exogenous alkaline phosphatase – May cause false-positive • Presence of nucleases found on human skin and materials – Causes nucleic acid degradation • In accurate volume delivery of samples and

Conclusion • Due to various types of HPVs the most reliable method of detection is through molecular testing • The Hybrid Capture 2 System helps differentiate btw low-risk and high-risk HPV infections • Is used in conjunction with pap smears to diagnose, treat, and prevent cervical cancer
- Slides: 20