The Human Body in Health and Illness 4

The Human Body in Health and Illness, 4 th edition Barbara Herlihy Chapter 25: Water, Electrolyte, and Acid-Base Balance 1

Lesson 25 -1 Objectives Describe the two main fluid compartments. Define intake and output. List factors that affect electrolyte balance. Describe the most common ions found in the intracellular and extracellular compartments. • List three mechanisms that regulate p. H in the body. • Discuss acid-base imbalances, acidosis and alkalosis. • • Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 2
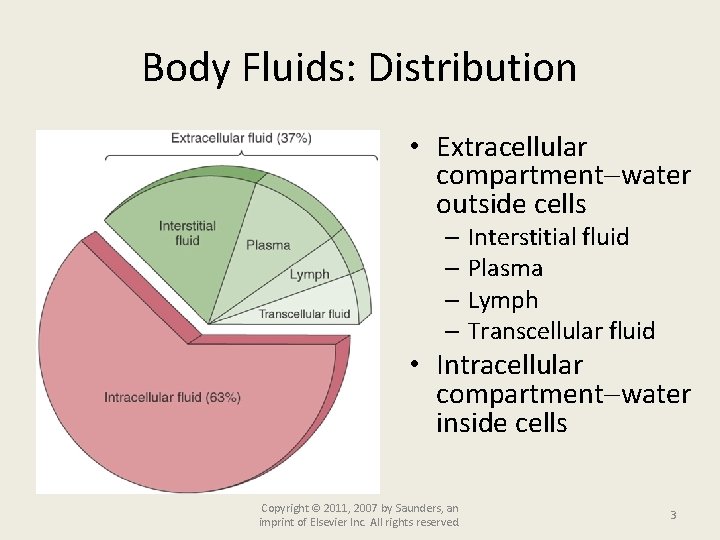
Body Fluids: Distribution • Extracellular compartment water outside cells – Interstitial fluid – Plasma – Lymph – Transcellular fluid • Intracellular compartment water inside cells Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 3

Composition of Body Fluid Extracellular • Na+ • Cl • HCO 3 - Intracellular • K+ • Phosphate • Mg 2+ • Plasma contains more protein than other extracellular compartments. Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 4
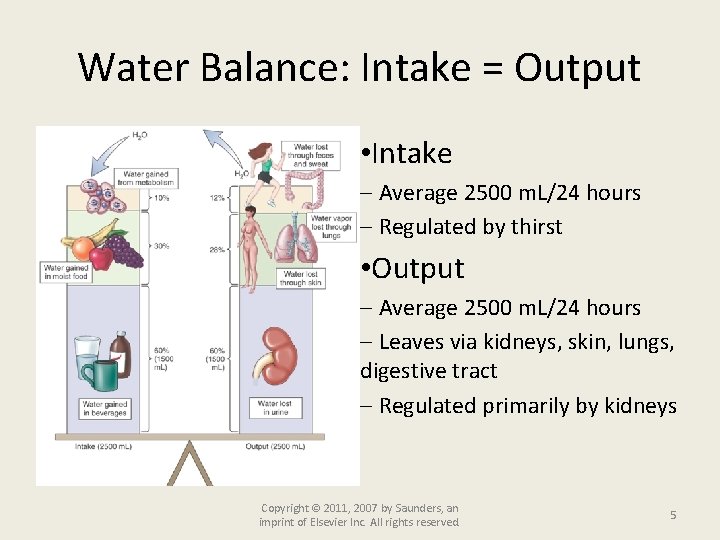
Water Balance: Intake = Output • Intake Average 2500 m. L/24 hours Regulated by thirst • Output Average 2500 m. L/24 hours Leaves via kidneys, skin, lungs, digestive tract Regulated primarily by kidneys Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 5

Water Imbalances • Dehydration – Deficiency of body water in interstitium – Poor skin turgor and “tenting” – Untreated hypovolemia, shock • Edema – Excess body water, usually in interstitium – Clinical assessment daily weights – Sudden weight gain suggests water retention. 2. 2 lb = 1 liter of water Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 6

Fluid Spacing: Where the Water Is • First spacing: Normal distribution • Second spacing: Interstitial edema – Responds to treatment with diuretics, excreted • Third spacing: Excess water in poorly accessible spaces – Examples: Ascites, water in digestive tract (paralytic ileus) Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 7

Electrolyte Balance: Review of Terms • Examples of ions: Na+, Cl-, K+, HCO 3 Cation examples: Na+, K+, Ca 2+, Mg 2+ Anion examples: Cl-, HCO 3 - • Examples of electrolytes: Na. Cl, KCl, Na. HCO 3 • Example of ionization: Na. Cl Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. Na+ + Cl- 8
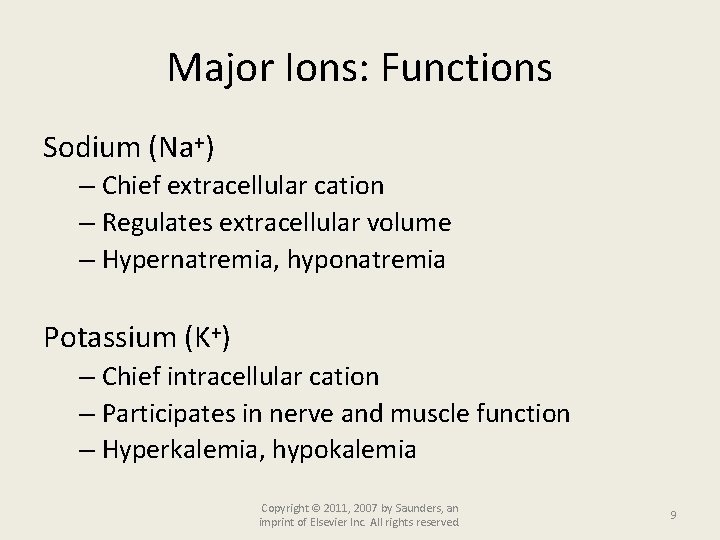
Major Ions: Functions Sodium (Na+) – Chief extracellular cation – Regulates extracellular volume – Hypernatremia, hyponatremia Potassium (K+) – Chief intracellular cation – Participates in nerve and muscle function – Hyperkalemia, hypokalemia Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 9

Major Ions: Functions (cont’d. ) • Calcium (Ca 2+) – Strengthens bone and teeth – Participates in muscle contraction – Hypocalcemic tetany, hypercalcemia • Magnesium (Mg 2+) – Participates in nerve and muscle function Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 10

Major Ions: Functions (cont’d. ) • Chloride (Cl−) – Chief extracellular anion – Involved in extracellular volume control • Bicarbonate (HCO 3−) – Part of bicarbonate buffer system – Participates in acid-base balance – Alterations in plasma Cl- affect HCO 3− levels. Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 11

Acid-Base: Review of Terms • Acid: Dissociates into H+ (the acid) and an anion • Base: Removes H+ from solution • p. H: Unit of measurement indicating [H+] in solution Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 12
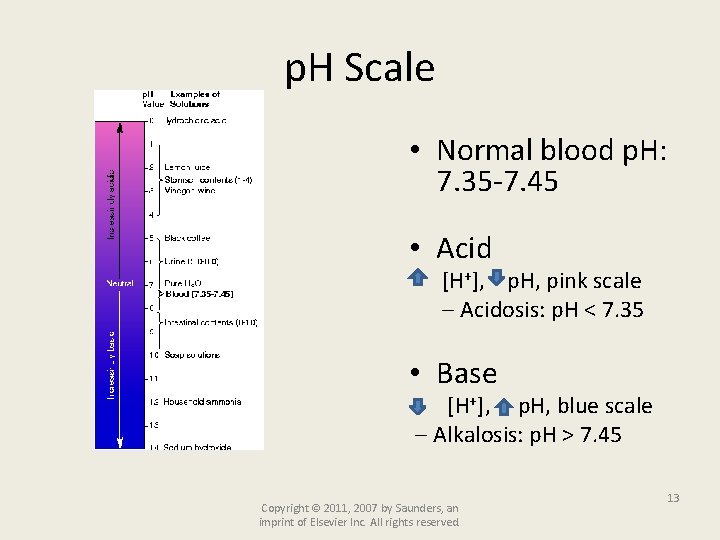
p. H Scale • Normal blood p. H: 7. 35 -7. 45 • Acid [H+], p. H, pink scale Acidosis: p. H < 7. 35 • Base [H+], p. H, blue scale Alkalosis: p. H > 7. 45 Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 13

Regulation of p. H • Buffers: First line of defense in regulating p. H • Lungs: Second line of defense • Kidneys: Third line of defense Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 14

Acid-Base Balance: Buffers • Buffers maintain normal p. H. • Always work in pairs – One part of a buffer (base) removes H+. – The other part (acid) donates H+. • Buffer systems – Bicarbonate-carbonic acid buffer pair – Other buffer pairs Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 15

Respiratory Control of Acids and Bases CO 2 + H 2 O ⇄ H 2 CO 3 ⇄ H+ + HCO 3 - • Slower respiration can generate H+ • Faster respiration can diminish H+ Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 16

Renal Control of Acid-Base • Reabsorb or excrete H+, as needed. • Reabsorb or excrete bicarbonate, as needed. – Kidneys reabsorb filtered bicarbonate. – Kidneys also make bicarbonate. Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 17

Causes of Imbalances Acid-Base Imbalances Acidosis Alkalosis Respiratory Metabolic Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 18
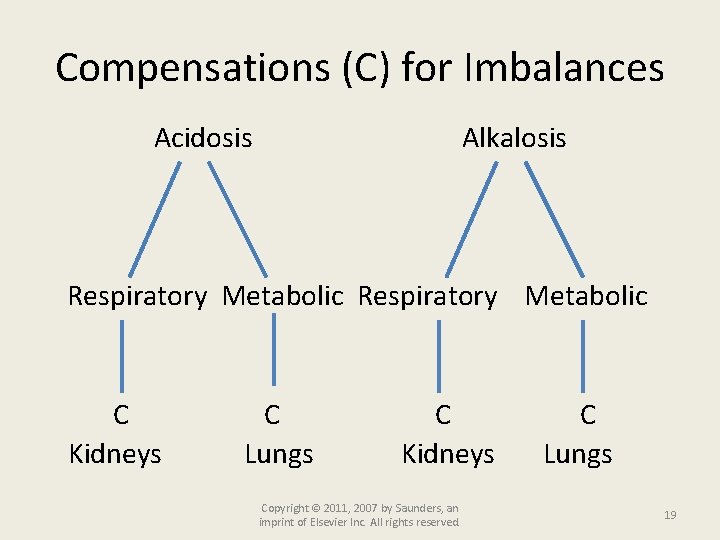
Compensations (C) for Imbalances Acidosis Alkalosis Respiratory Metabolic C Kidneys C Lungs C Kidneys Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. C Lungs 19
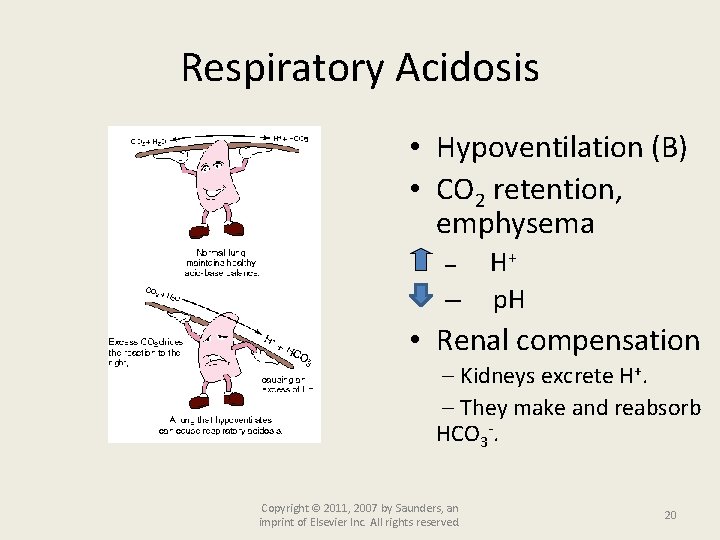
Respiratory Acidosis • Hypoventilation (B) • CO 2 retention, emphysema – – H+ p. H • Renal compensation Kidneys excrete H+. They make and reabsorb HCO 3 -. Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 20

Metabolic Acidosis • Caused by nonrespiratory conditions • Example: Diabetic ketoacidosis (DKA) H+ H + H + H+ • CO 2 + H 2 O ⇄ H 2 CO 3 ⇄ H+ + HCO 3 Respiratory compensation (Kussmaul respirations) Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 21
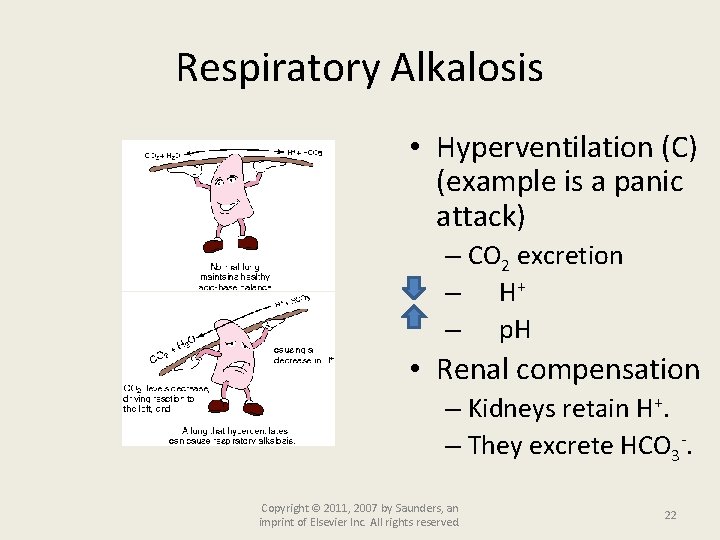
Respiratory Alkalosis • Hyperventilation (C) (example is a panic attack) – CO 2 excretion – H+ – p. H • Renal compensation – Kidneys retain H+. – They excrete HCO 3 -. Copyright © 2011, 2007 by Saunders, an imprint of Elsevier Inc. All rights reserved. 22
Metabolic Alkalosis • Caused by nonrespiratory conditions – Example: Persistent vomiting (loss of H+) • Loss of H+ drives the reaction to the right to produce more H+ CO 2 + H 2 O H 2 CO 3 ⇄ H+ + HCO 3 - • Compensation: Hypoventilation – Increased [COCopyright production of H+ © 2011, 2007 by Saunders, an 2] triggers imprint of Elsevier Inc. All rights reserved. 23
- Slides: 23