THE HIGHWAY TO m Ab drug candidates 1

THE HIGHWAY TO m. Ab drug candidates 1 CONFIDENTIAL

MANAGEMENT TEAM AND BOARD OF DIRECTORS COMEX 2 Laurent POUYET, Ph. D Patrick VLIEGHE, Ph. D Head of R&D Programs Head of R&D Operations Head Administration & Finance Ex Px. Tx Ex Tr. GT Ex Vect-Horus, SATT Sud- Thierry JEAN, Ph. D 1 Pr. François ROMAGNE, Ph. D 1 Stéphanie BLANCHIN, Ph. D President & CEO Chief Scientific Officer Ex Founder, President and Ex CSO & Co-founder CEO Cerep for 25 years Innate Pharma 1 Actionnaires fondateurs Est CONFIDENTIAL
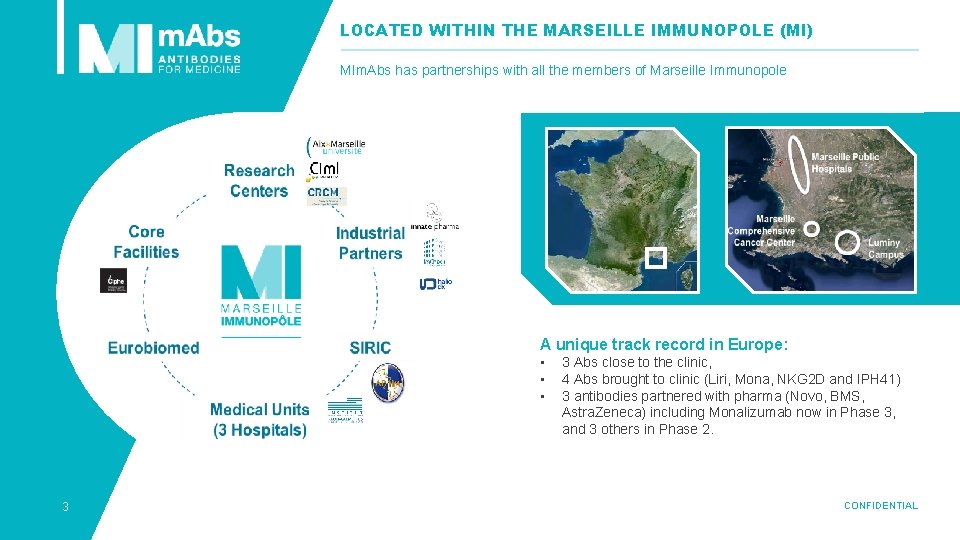
LOCATED WITHIN THE MARSEILLE IMMUNOPOLE (MI) MIm. Abs has partnerships with all the members of Marseille Immunopole A unique track record in Europe: • • • 3 3 Abs close to the clinic, 4 Abs brought to clinic (Liri, Mona, NKG 2 D and IPH 41) 3 antibodies partnered with pharma (Novo, BMS, Astra. Zeneca) including Monalizumab now in Phase 3, and 3 others in Phase 2. CONFIDENTIAL
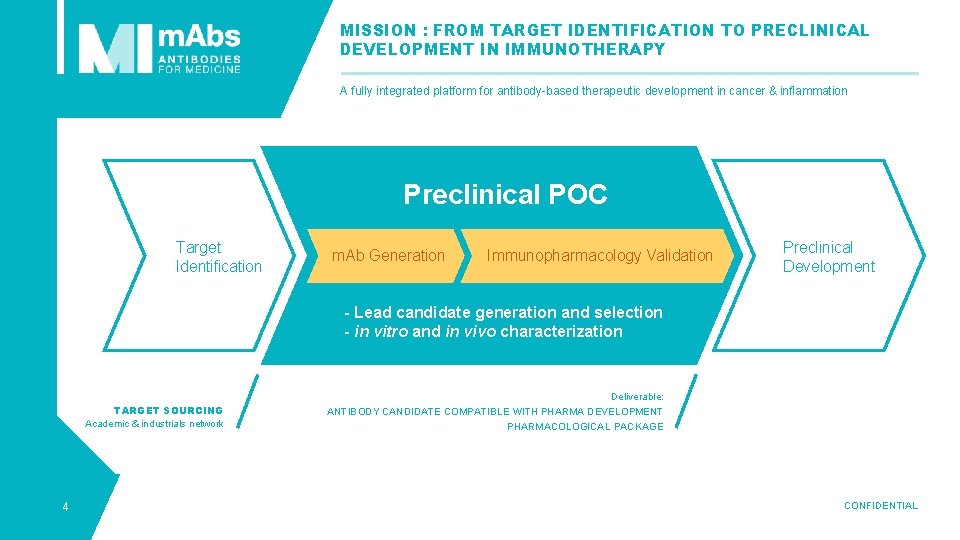
MISSION : FROM TARGET IDENTIFICATION TO PRECLINICAL DEVELOPMENT IN IMMUNOTHERAPY A fully integrated platform for antibody-based therapeutic development in cancer & inflammation Preclinical POC Target Identification m. Ab Generation Immunopharmacology Validation Preclinical Development - Lead candidate generation and selection - in vitro and in vivo characterization TARGET SOURCING Academic & industrials network 4 Deliverable: ANTIBODY CANDIDATE COMPATIBLE WITH PHARMA DEVELOPMENT PHARMACOLOGICAL PACKAGE CONFIDENTIAL
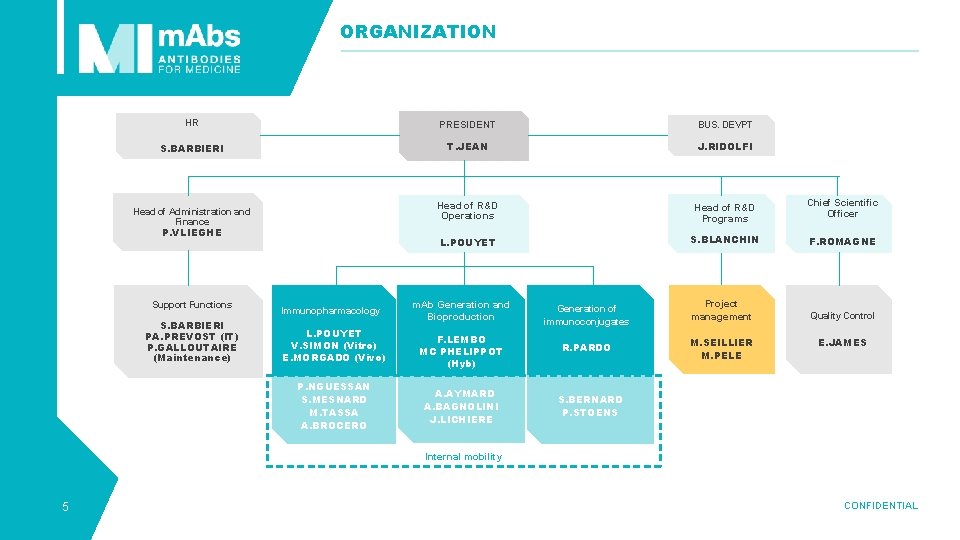
ORGANIZATION HR PRESIDENT BUS. DEVPT S. BARBIERI T. JEAN J. RIDOLFI Head of R&D Operations Head of R&D Programs L. POUYET S. BLANCHIN F. ROMAGNE Quality Control Head of Administration and Finance P. VLIEGHE Support Functions S. BARBIERI PA. PREVOST (IT) P. GALLOUTAIRE (Maintenance) Immunopharmacology L. POUYET V. SIMON (Vitro) E. MORGADO (Vivo) P. NGUESSAN S. MESNARD M. TASSA A. BROCERO m. Ab Generation and Bioproduction Generation of immunoconjugates Project management F. LEMBO MC PHELIPPOT (Hyb) R. PARDO M. SEILLIER M. PELE A. AYMARD A. BAGNOLINI J. LICHIERE Chief Scientific Officer E. JAMES S. BERNARD P. STOENS Internal mobility 5 CONFIDENTIAL

MAB GENERATION AND VALIDATION PROCESS Full m. Ab/target validation : ~2 years, 3 -4 FTE Project analysis • Target analysis • Immunogen and screen design • Desired m. Ab format(s) • Design of pharmacology models (decision to make KI) or surrogates Generation of rec-m. Abs • Immunization of WT or genetically engineered mice • High throughput cloning from Hybridoma • Fc modification (human and mice isotypes), • Production in CHO or HEK, • Secondary screening on recombinant m. Abs Antibody engineering and production (mg to g) • Fc engineering, humanization, ADC, bispecific (KIH, AEX) • Transient or stable production in bioreactor • Biochemistry, industry level characterization and QC Immunopharmacology • In vitro & in vivo • ADCC, CDC, cytotox • Syngenic, xenogenic test of m. Abs, PK studies • Creation of genetically engineered mice model (KI or KO) 6 CONFIDENTIAL

SCIENTIFIC TRACK RECORD : PUBLIC SNAPSHOT • More than 100 immunizations programs (tools and drug candidates) • ~15 target validation programs Biotechs & Pharmas • 3 Antibody drug conjugate programs: • Coupling of toxins on several targets • in vitro & in vivo demonstration • 3 bispecific programs • 1 new Ab in clinical pipeline of Astra-Zeneca (CD 39 in-licensed by AZ from Innate pharma) • 3 new Ab transferred to regulatory development of Innate Pharma and Sanofi : CD 73, Siglec, new bispecific format engaging NK cells • 2 ADC (MICA, NKp 46) close to be transferred to enter clinical development of partners • 1 new tool for bispecifics : a human CD 3 e KI mice model • 13 publications in peer reviewed journal (Cell, Cell report, Immunity…) Others non-disclosed Academia • Significant contribution to the creation of start-ups • Emergence. Therapeutics, Kalsiom • Main clients • Emergence. Therapeutics • Kalsiom • Innate Pharma 7 • Domain Therapeutics • Imcheck CONFIDENTIAL

BUSINESS MODEL MIm. Abs is seeking collaborations with biotechs / pharmas / academia Preferred Model: FTE-based partnership • Definition of early target validation workplan (immunogen design, specifications of m. Abs (naked, ADC, bispecifics), in vitro and in vivo POC design (KI models)), • Generation and characterization of a collection of antibodies • Antibody engineering in different formats (naked humanized, ADC, bispecific) • In vivo proof of concept in mice model Alternative Option: fee-for-service collaboration • Antibody production, reformatting • In vitro tests (ADCC, CDC, Immunopharmacology tests, custom tests…) • In vivo tests, PK/PD, efficacy, immune profiling. In both scenarios MIm. Abs does not retain any IP rights • All IP generated during the collaboration belongs to Client • MIm. Abs has no proprietary drug-discovery program 8 CONFIDENTIAL

MIm. Abs www. mi-mabs. org 117 Avenue de Luminy CS 10951 F 13288 Marseille Cedex 9 Tel: +33 (0)9 70 81 48 21 Projet financé avec le concours de l’Union européenne avec le Fonds Européen de Développement Régional 9 CONFIDENTIAL
- Slides: 9