The Hemostatic Pathway Novel Anticoagulants David Bolos PGY

The Hemostatic Pathway & Novel Anticoagulants David Bolos, PGY 5 Fellow Talk 9/6/2016

Overview Phases of the Hemostatic Process Control Mechanisms of Clotting Novel Anticoagulants/Oral Anticoagulants Transitioning between Anticoagulants Management of Bleeding Anticoagulants in Development

Phases of the Hemostatic Process Endothelial Injury and Formation of the Platelet Plug Propagation of the Clotting Process by the Coagulation Cascade Termination of Clotting by Anti-thrombotic Control Mechanisms Removal of the Clot by Fibrinolysis

Formation of the Platelet Plug Platelets are activated at the site of vascular injury to form a “plug” Injury to endothelium exposure of blood to subendothelial elements endothelial cell activation promotes further recruitment Functional response of activated platelets (FOUR) Adhesion: deposition to subendothelial matrix Aggregation: platelet-platelet cohesion Secretion: release of platelet granule proteins Procoagulant activity: enhancement of thrombin generation

Platelet Activation There a number of physiologic platelet stimuli Adenosine Diphosphate (ADP) ADP binds to two G-protein coupled purinergic receptors, P 2 Y 1 & P 2 Y 12. P 2 Y 1 Calcium mobilization, platelet shape change, reversible aggregation P 2 Y 12 platelet secretion and more stable aggregation (Clopidogrel blocks activation) Epinephrine Thrombin (potent) Thrombin activation is mediated by a family of G-protein coupled protease-activated receptors (PARs). Platelets have a dual receptor system for thrombin, with 2 distinct receptors PAR 1 & PAR 4. Collagen (potent) Integrin glycoproteins GPIa/IIa and GPVI are the two most important platelet collagen receptors (bleeding diathesis with deficiencies)

Platelet Adhesion Following activation platelets undergo shape changes (elongated pseudopods) making them “sticky” Adhesion Primarily mediated by binding of platelet surface receptor GP Ib/IX/V complex to VWF in subendothelial matrix. Deficiency of any component of GP Ib/IX/V complex or VWF leads to congenital bleeding disorders Bernard-Soulier Disease Von Willebrand Disease Other adhesive interactions contribute GP Ia/IIa to collagen fibrils in the matrix.

Platelet Aggregation Results in both exposure and conformational changes in the GP IIb/IIIa receptor on platelet surface binding of immobilized VWF and Fibrinogen GP IIb/IIIa (integrin) undergoes a conformation change following platelet stimulation higher affinity fibrinogen receptor When GP IIb/IIIa binds to immobilized VWF, the cytosolic portion of the activated complex binds to cytoskeleton mediating platelet spreading & clot retraction Glanzmann Thrombasthenia mutations in the gene for either the alpha IIb or beta-3 subunit GP IIb/IIIa antagonists in treatment of CAD (Abciximab, Eptifibatide)

Platelet Secretion Two types of Granules: alpha & dense Alpha Granules: many proteins including: Fibrinogen (source at sites of injury in addition to source in plasma) VWF: Von Willebrand Factor Thrombospondin & Fibronectin (adhesive proteins, stabilize aggregates) PDGF (mediates tissue repair), Platelet Factor 4 (chemokine moderating effects of heparinlike molecules) P-selectin (cell adhesion molecule) Dense Granules: ADP, ATP, i. Cal, Histamine, Serotonin

Procoagulant Activity Involves both exposure of procoagulant phospholipids (primarily phosphatidylserine), and the subsequent assembly of the enzyme complexes in the clotting cascade on the platelet surface.

Formation of Platelet Plug Overview https: //www. youtube. com/watch? v=0 pnpo. Ey 0 e. YE

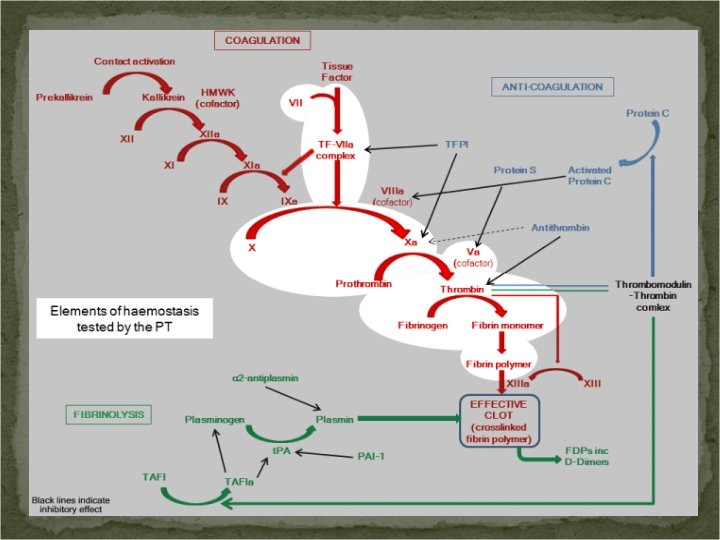
Clotting Cascade & Propagation of the Clot The central feature is the sequential activation of a series of proenzymes (zymogens) to active enzymes Consists of Intrinsic & Extrinsic pathways (see handout 1) The function of the active enzymes is markedly facilitated by the formation of multiple component macromolecular complexes (The X-ases, Prothrombinase) All of the procoagulants are synthesized in Liver, Except VWF (megakaryocytes & endothelial cells) and Factor VIII (produced by endothelial cells) Post-translational modification Occurs Vitamin K dependent Procoagulants (II, VII, IX, X) and anticoagulants (Proteins C & Protein S) Vitamin K dependent carboxylated glutamic acid residues function as Ca-binding sites that are important in the assembly of the membrane bound macromolecular procoagulant complexes

Coagulation Cascade https: //www. youtube. com/watch? v=s 4 Fo. Sf 6 Yi_s Handout 1

Clotting Cascade & Propagation of the Clot Intrinsic Pathway: initiated by exposure of blood to a negatively charged surface (such as celite, kaolin, or silica in the in vitro activated partial thromboplastin clotting time, a. PTT) Extrinsic Pathway: activated by tissue factor exposed at the site of injury or tissue factor-like material (thromboplastin, in the in vitro prothrombin clotting time, PT) Both pathways converge on the activation of Factor X X, as a component of Prothrombinase thrombin Thrombin converts fibrinogen from a soluble plasma protein into an insoluble fibrin clot (testing in the in vitro thrombin time, TT)

Thrombin Generation Primary physiologic event in initiating clotting is exposure of TF at wound site and interaction with Factor VIIa. Small initial amount of Thrombin generated then activates Factor XI (in a feedback manner) amplification of thrombin generation This then further leads to downstream amplification of factors V, VIII, IX, platelets, etc. explosive thrombin generation (see Handout 2)

Multicomponent Complexes Extrinsic X-ase: activated Factor VIIa (protease), TF (cofactor), Factor X (substrate) activates Factor X and Factor IX Intrinsic X-ase: Factor IXa (protease), activated factor VIIIa (cofactor), Factor X (substrate) activates Factor X Prothrombinase: Factor Xa (protease), Factor Va (cofactor), Prothrombin/Factor II (substrate) Thrombin (IIa) Protein C Anticoagulant Complex: Thrombin (enzyme), Thrombomodulin (cofactor), Protein C (substrate) anticoagulation control mechanisms

Critical Role of Polyphosphate (Poly. P) Inorganic polyphosphate (Poly. P) is a linear highly anionic polymer of orthophosphates linked by highenergy phosphoanhydride bonds. In microorganisms, Poly. P is synthesized from ATP and may serve as an energy store allowing bacteria to resynthesize ATP in times of stress. Poly. P is also stored in human platelet dense granules and is efficiently released upon platelet activation

Critical Role of Polyphosphate (Poly. P) Multiple anion charges, Poly. P is a potent procoagulant physiological negatively charged surface that triggers blood coagulation via intrinsic pathway. Source in this setting may be derived from injured tissue Poly. P’s other effects on clotting: accelerates Factor V activation, Dampens the inhibitory effects of Tissue Factor Pathway Inhibitor (TFPI), enhances fibrin polymerization, accelerates Factor XI activation by Thrombin

Deficiencies of Initial Intrinsic Pathway Proteins Physiological relevance of the initial complex of intrinsic pathway is not established Deficiencies in these proteins (Prekallikrein, HMWK, and Factor XII) are not associated with bleeding tendencies Mutations in Factor XII have been linked in a subset of patients with Hereditary Angioedema and normal C 1 inhibitor levels However, injury-related bleeding is seen with deficiency of Factor XI, suggesting it plays important hemostatic role, independent of contact activation and Factor XII (thrombin feedback activates Factor XI, with Poly. P as cofactor).

Continuation of the Cascade Factor Va binds Factor Xa prothrombinase complex Thrombin (IIa) Thrombin converts Fibrinogen Fibrin polymerization Activated Factor XIIIa stabilizes and crosslinks overlapping Fibrin strands Factor XIIIa with fibrinogen controls volume of RBC’s trapped within a thrombus Clot Size

Control Mechanisms & Termination of Clotting Hemostatic response is rapid and if left unchecked would lead to thrombosis, vascular inflammation, and tissue damage Modulated by number of mechanisms Dilution of procoagulants in flowing blood Removal of activated factors through the reticuloendothelial system (esp in Liver), Control by natural antithrombotic pathways anchored on the vascular endothelial cells (maintaining fluidity of blood)

Control Mechanisms & Termination of Clotting Termination Phase of Coagulation Process Involves two circulating enzyme inhibitors Antithrombin and Tissue Factor Pathway Inhibitor (TFPI) & clotting-initiated inhibitory process, The Protein C pathway Prostacyclin, Thromboxane, and Nitric Oxide modulate vascular and platelet reactivity Termination phase is critical problems lead to thrombotic disorders such as Antithrombin, Protein C, and Protein S deficiency

Antithrombin and Heparin Antithrombin is a circulating plasma protease inhibitor, neutralizing many enzymes including thrombin, factors Xa & IXa, XIIa & XIa. Two active functional Sites: the reactive center (Arg 393 -Ser 394), and the Heparin Binding Site located at the amino-terminus The binding of endogenous/exogenous heparins to the Heparin Binding Site on AT produces a conformational change in AT which accelerates the inactivating process 1000 x to 4000 x. See Handout 4

Activated Protein C & Protein S APC inactivates Factors Va & VIIIa, thereby inactivating the Prothrombinase and the intrinsic Xase, respectively Factor Va is first cleaved at Arg 506 and then at Arg 306 & Arg 679 by APC Peptide bone cleavage at Arg 506 is essential for the exposure of cleavage sites at Arg 306 & Arg 679

Activated Protein C & Protein S Factor V Leiden, in which Arginine at position 506 is replaced by Glutamine, is not susceptible to cleavage at position 506 by APC and is therefore inactivated more slowly hypercoagulable state Protein S circulates in two forms, free (active) and bound to C 4 b binding protein of complement system C 4 b is an acute phase reactant, inflammatory states increase its concentration, decrease free Protein S enhancing likelihood of thrombosis in these states

Tissue Factor Pathway Inhibitor Synthesized by the microvascular endothelium TFPI circulates in the plasma, low concentrations compared to AT Inhibits Factor X activation in two ways: Direct inhibition of Factor Xa Complexes with Factor Xa and the complex inhibits TF/FVIIa impairing the triggering mechanism of the Extrinsic pathway

Clot Elimination & Fibrinolysis To restore vessel patency following hemostasis, the clot must be organized and removed by the proteolytic enzyme Plasmin in conjunction with wound healing and tissue remodeling Plasminogen binds fibrin and tissue plasminogen activator (t. PA). This ternary complex leads to conversion of plasminogen Plasmin (active) Plasmin has broad substrate specificity cleaves fibrin, fibrinogen, plasma proteins, clotting factors

Clot Elimination & Fibrinolysis Plasmin cleaves fibrin strands Fibrin degradation products one major product is D-dimer (consists of two D-domains from fibrin adjacent monomers that have been crosslinked by Factor XIIIa) Plasmin also cleaves Factor XIIIa reduces fibrin crosslinking Plasmin-plasminogen activator system is complex, and is regulated with inhibitors as well (plasminogen activator inhibitors, PAI-1/PAI-2)


https: //www. youtube. com/watch? v=Yn. G 3 UTp. Ww. W 4 https: //www. youtube. com/watch? v=u. E 3 Af. RT 4 Vsw

Novel Anticoagulants/Oral Anticoagulants Options for anticoagulation have been expanding In addition to heparins and vitamin K antagonists, anticoagulants that directly target the enzymatic activity of thrombin and Factor Xa are available Appropriate use requires knowledge of their individual characteristics, risks, benefits Here are practical aspects of Novel Anticoagulants Common uses include VTE treatment/ppx, Afib, Unstable Angina, MI, CVA, PCI, and HIT

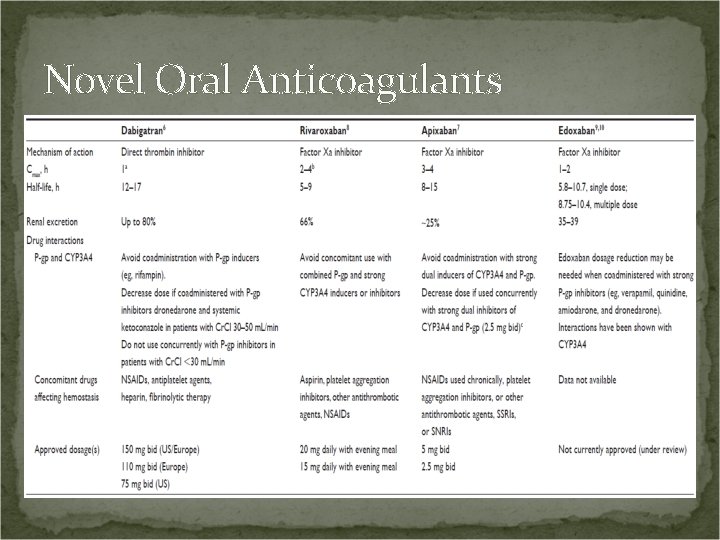
Direct Thrombin Inhibitors (DTIs) Bind to the active site of the thrombin enzyme or to two sites: the active site and “exosite I”, a positively charged region of the thrombin molecule that is physically separated from the active site. See Handout 3 “Exosite I” is also the site of interaction of many physiologic thrombin substrates (fibrinogen, Factor V, Protein C, Thrombomodulin, and thrombin receptors (PAR 1/PAR 4) on platelets) Thrombin is activated in both circulating and clot-bound forms. DTIs are able to block BOTH forms (Heparins are only able to inactivate Thrombin in the fluid phase, via antithrombin)

Direct Thrombin Inhibitors (DTIs) Parenteral DTIs include Bivalirudin, Argatroban, and Desirudin The ONLY oral DTI available for clinical use is Dabigatran (Pradaxa)

Direct Factor Xa Inhibitors Acts immediately upstream to of thrombin, at convergence point of intrinsic/extrinsic pathways Prevents amplified thrombin generation (one molecule of Xa can cleave 1000 molecules of prothrombin) Xa inhibitors bind to the active site of Xa and inhibit factor Xa activity without requirement of cofactors Similar to thrombin, Xa is active in circulating and clot forms, and Xa inhibitors block BOTH forms

Diret Factor Xa Inhibitors No parenteral Factor Xa inhibitors in clinical use Severeal oral direct Factor Xa inhibitors available: Rivaroxaban (Xarelto) Apixaban (Eloquis) Edoxaban (Lixiana) Nomenclature: -xaban (Xa-Ban)

Comparison with Heparin/Warfarin Differ in efficacy depending on clinical setting Dosing Dietary restrictions Ability to tolerate oral intake Monitoring Therapy Drug adherence Drug interactions Time in therapeutic range Cost $$$ Risks & Reversal Agents Monitoring reversal (INR/PTT/PT/TT/Xa) Advantages/disadvantages must be individualized to the patient and clinical setting

Settings in which Heparin/Warfarin Preferred Prosthetic heart valves Pregnancy Renal Impairment Antiphospholipid Syndrome (randomized study comparing Rivaroxaban to Warfarin in progress) Compliance (lack of monitoring, short half lives) Gastrointestinal Disease Dosing Convenience (dabigatran & Apixaban require BID dosing) Cost $$$
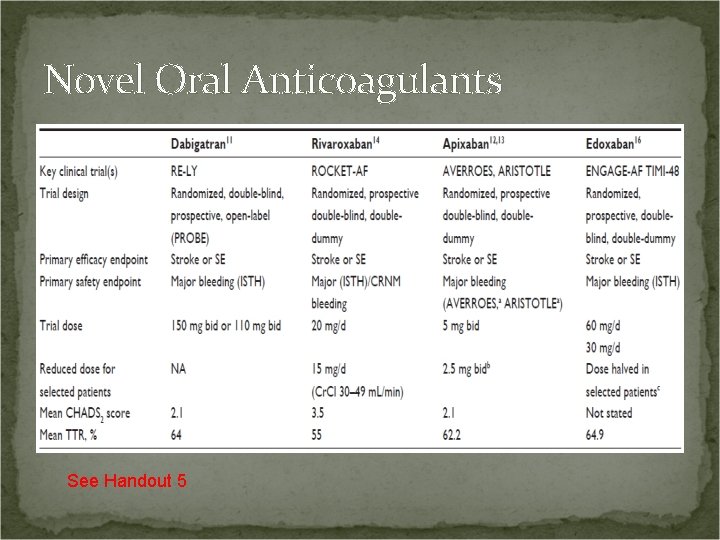
Novel Oral Anticoagulants See Handout 5
Novel Oral Anticoagulants
Novel Oral Anticoagulants

Bivalirudin Synthetic 20 amino acid peptide that binds to the thrombin catalytic site and exosite I, reversibly inhibiting Peptide sequence is analog of hirudin, protein extracted from salivary gland of medicinal leech Indications are PCI and HIT Administered at dose of 0. 75 mg/kg IV bolus, followed by 1. 75 mg/kg/hr during procedure (Cr. Cl<30, 1 mg/kg/hr rate) Metabolized in Kidney/Liver Can be hemodialyzed

Argatroban Synthetic peptide-based direct thrombin inhibitor that interacts with active site of thrombin Short in vivo plasma half life (40 -50 min) Hepatically metabolized (dosing adjustments for hepatic impairment, not for renal) HIT: 2 mcg/kg/min CIVI, monitoring q 2 hrs with a. PTT Dose adjusted to achieve target 1. 5 to 3 x the initial baseline a. PTT, not to exceed 100 seconds Also affects PT, so when transitioning to warfarin must use adjusted INR target Also used for PCI in patients with HIT, bolus of 350 mcg/kg over 3 -5 minuts, followed by 25 mcg/kg/min

Dabigatran (Pradaxa) Orally administered prodrug converted in liver to dabigatran, an active direct thrombin inhibitor (inhibts clot and circulating thrombin) Half life ~ 12 to 17 hours (normal renal function) Capsules should only be dispensed and stored in original bottle (with desiccant) or blister package (product breakdown from moisture) Not to be crushed or have capsule opened (increases)

Dabigatran (Dosing) Fixed dose without monitoring Maximum effects achieved within 2 -3 hours of ingestion Dosing differs based on indication and renal function VTE ppx in surgical patients 110 mg 1 -4 hours after surgery, followed by 220 mg daily for 28 -35 days (hip) or 10 days (knee replacement) Treatment VTE 150 mg BID (Cr. Cl>30) Stroke Prevention in Afib 110 mg BID or 150 mg BID (Cr. Cl>30)

Dabigatran (Dose Modifications) Renal Insufficiency Dose reduction for Cr. Cl 15– 30, generally by 50% Cr. Cl<15, avoid use P-glycoprotein Inhibitors/Inducers (See Handout 5 & 6) Dabigatran is a Substrate for P-glycoprotein, inducers reduce anticoagulation effect, inhibitors increase anticoagulation effect NOTE: not metabolized by Cytochrome P 450 system Obesity Avoid if BMI > 40 kg/m 2 or Weight > 120 kg (same for all DOAC’s) European labeling: Age>75, dose reduce

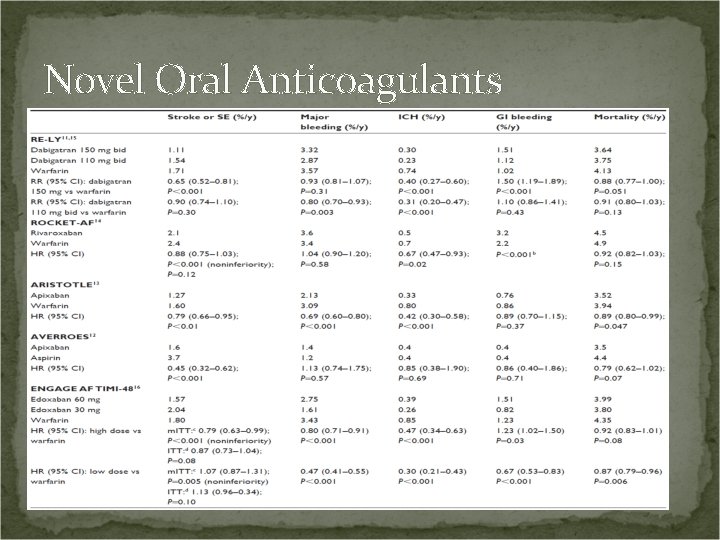
Dabigatran (Risks) As with all anticoagulants, bleeding risk increased Antidote is available Prax. Bind (Idarucizumab) Overall bleeding rates similar to that of warfarin Dabigatran may be associated with slightly lower rate of intracranial hemorrhage and death, and Slightly higher risk of GI bleed at 150 mg BID dose, but not the 100 mg BID dosage Non-bleeding GI events (dyspepsia, dysmotility, GERD) were twice as common compared to Warfarin (RE-LY trial) Black Box Warning regarding the risk of thrombotic events following premature discontinuation

Rivaroxaban (Xarelto) Orally available direct factor Xa inhibitor Half life ~7 -17 hours Given at a fixed dose without monitoring 15 mg and 20 mg tablets are to be taken with food VTE ppx in surgical patients 10 mg daily for 35 days (hip), 12 days (knee) Treatment VTE 15 mg BID x 21 days, then 20 mg daily w/food Stroke prevention in Afib 20 mg daily with evening meal (Cr. Cl>50), or 15 mg daily with evening meal (Cr. Cl 30 -49)

Rivaroxaban (Dose Modifications) Not recommend for Cr. Cl<30 Not to be used with Cr. Cl<15 Avoid use in patients with significant hepatic impairment, Child-Pugh Class B and C with coagulopathy Interacts with drugs that are potent dual inhibitors of CYP -3 A 4 and P-glycoprotein (see handouts 5 & 6) (ketoconazole, itraconazole, voriconazole, posaconazole, clarithromycin, protease inhibitors (ritonavir)) Potent inducers of CYP-3 A 4 reduce effects No specific targeted antidote Black Box warnings similar to Dabigatran

Apixaban (Eliquis) Orally available direct factor Xa inhibitor Half life ~5 -9 hours Less renal excretion than others, better for CKD pts Given at a fixed dose without monitoring VTE ppx in surgical patients 2. 5 mg BID for 35 days (hip), 12 days (knee) Treatment VTE 10 mg BID x 7 days, then 5 mg BID Stroke prevention in Afib 5 mg BID(Cr. Cl>50), or 2. 5 mg BID if Age>80, Weight <60 kg, and/or Cr > 1. 5 Dose reduce for CYP 3 A 4 inhibitors and P-glycoprotein inhibitors Similar black box warnings as stated previously No direct antidote

Edoxaban (Lixiana) Orally available direct factor Xa inhibitor Half life ~6 -11 hours Given at a fixed dose without monitoring, absorption unaffected by food VTE ppx in surgical patients, VTE treatment, Afib stroke prevention 30 to 60 mg daily (depending on risk factors) Dose reduce for P-glycoprotein inhibitors, caution with CYP 3 A 4 inhibitors Similar black box warnings as stated previously No direct antidote

Transitioning Between Anticoagulants Goal maintain stable anticoagulation When transitioning from DOAC to VKA… important to keep in mind that DOACs may alter PT/INR, and full effect of VKA does not occur for first few days, despite prolonged PT/INR Likewise, when transitioning from warfarin to DOAC, the resolution of warfarin effect may take several days

Transitioning Between Anticoagulants Dabigatran to Warfarin: these two agents are overlapped, # days overlap depends on Cr. Cl >50, start VKA 3 days before discontinuing dabigatran Cr. Cl 30 -49, start VKA 2 days before discontinuing dabigatran Cr. Cl 15 -30, start VKA 1 day before discontinuing dabigatran

Transitioning Between Anticoagulants Rivaroxaban to Warfarin & Apixaban to Warfarin Prescribing information suggests stopping Rivaroxaban/Apixaban and providing parenteral agent during warfarin initiation because INR cannot be monitored adequately Edoxaban to Warfarin: Pts taking 60 mg, reduce dose 30 mg and begin VKA concomitantly Pts taking 30 mg, reduce dose 15 mg and begin VKA concomittantly INR measured just prior to daily Edoxaban dose, d/c edoxaban when INR>2 on two consecutive days

Transitioning Between Anticoagulants When switching from Warfarin Direct Oral Anticoagulant, in general it is reasonable to discontinue Warfarin and initiate DOAC when INR<2 Specifically… INR<2 (warfarin argatroban, dabigatran, apixaban) INR<2. 5 (warfarin edoxaban) INR<3 (warfarin rivaroxaban)


Management of DOAC Bleeding Major Bleeding (intracranial, retroperitoneal, compartment syndrome, massive GI) Minor Bleeding (Epistaxis, uncomplicated soft tissue bleeding, minor (sl 0 w) GI bleeding)

Management of DOAC Bleeding Assessment of Anticoagulation Status (agent, dose, time since last dose, renal function) Anticoagulation resolved generally after five half lives since last dose Dabigatran (2. 5 -3 days after last dose) Rivaroxaban (1 -2 days after last dose) Apixaban (1. 5 -3 days after last dose) Edoxaban (1. 3 -2 days after last dose)

Management of DOAC Bleeding Activated Prothrombin Complex Concentrate (a. PCC) FEIBA NF: contains coagulation factors II, VII, IX, X (factor VII mostly activated) Unactivated Prothrombin Complex Concentrate (PCC) Kcentra (II, VII, IX, X inactive forms) Bebulin VH/Profilnine (II, IX, X, no VII) Novo. Seven Coagulation Factor VIIa

Management of DOAC Bleeding Idarucizumab (Prax. Bind), Antidote for Dabigatran Pro-hemostatic Agents Antifibrinolytics: Tranexamic Acid & epsilon-aminocaproic acid DDAVP Oral activated charcoal if last dose within 2 hrs Hemodialysis (Dabigatran) Fresh Frozen Plasma contains all coagulation factors (time to thaw, time to administer, large volume) Cryoprecipitate contains Fibrinogen, Factor VIII, VWF, Factor XIII

Manage Major Bleed: Dabigatran Idarucizumab a. PCC (FEIBA) Tranexamic acid/aminocaproic acid Oral activated charcoal (if within 2 hrs) HD RBC/Platelet transfusion Surgical/Endoscopic intervention if appropriate

Manage Minor Bleed: Dabigatran Local hemostatic measures Consider anticoagulant discontinuation (half life 12 to 17 hours) Antifibrinolytic agents CBC, transfusion if needed

Manage Major Bleed: Xa inhibitors Unactivated PCC (Kcentra) Tranexamic acid/aminocaproic acid Oral activated charcoal (if last dose recent enough) RBC/Platelet transfusion Surgical/Endoscopic intervention if appropriate

Manage Minor Bleed: Xa inhibitors Local hemostatic measures Consider anticoagulant discontinuation Rivaroxaban half-life 5 -9 hours Apixaban half life 8 -15 hours Edoxaban half life 6 -11 hours Antifibrinolytic agents CBC, transfusion if needed


Anticoagulants in Development A variety of anticoagulant strategies targeting other steps in coagulation are in development Tissue Factor Pathway Inhibitor Factor VIII inhibitors Factor IXa inhibitor Factor XIIa inhibitor Polyphosphate inhibitors

- Slides: 66