The Heisenberg Uncertainty Principle Inderjit Singh Heisenberg realized

The Heisenberg Uncertainty Principle Inderjit Singh

Heisenberg realized that. . . l In the world of very small particles, one cannot measure any property of a particle without interacting with it in some way l This introduces an unavoidable uncertainty into the result l One can never measure all the properties exactly
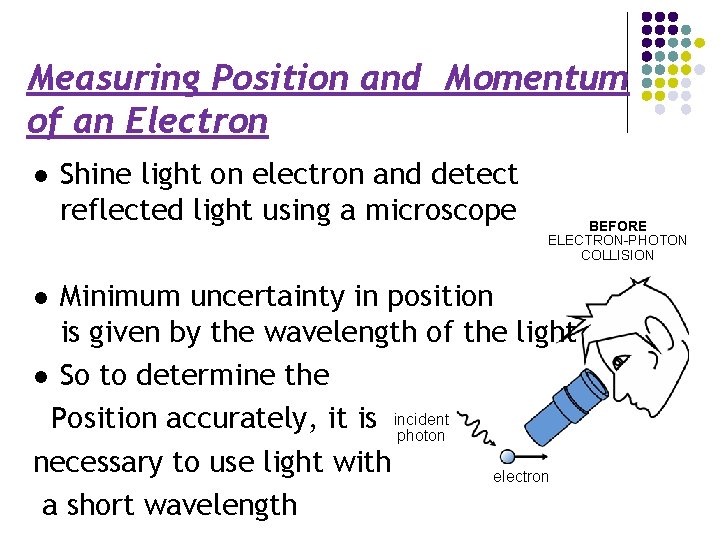
Measuring Position and Momentum of an Electron l Shine light on electron and detect reflected light using a microscope BEFORE ELECTRON-PHOTON COLLISION Minimum uncertainty in position is given by the wavelength of the light l So to determine the Position accurately, it is incident photon necessary to use light with electron a short wavelength l

Measuring Position and Momentum of an Electron By Planck’s law E = hc/λ, a photon with a short wavelength has a large energy AFTER ELECTRON-PHOTON l Thus, it would impart a large COLLISION ‘kick’ to the electron l But to determine its momentum accurately, electron must only be given a small kick scattered photon l This means using light of long wavelength ! recoiling l electron

Implications l It is impossible to know both the position and momentum exactly, i. e. , Δx=0 and Δp=0 l These uncertainties are inherent in the physical world and have nothing to do with the skill of the observer l Because h is so small, these uncertainties are not observable in normal everyday situations

Example of Baseball l A pitcher throws a 0. 1 -kg baseball at 40 m/s l So momentum is 0. 1 x 40 = 4 kg m/s l Suppose the momentum is measured to an accuracy of 1 percent , i. e. , Δp = 0. 01 p = 4 x 10 -2 kg m/s

Example of Baseball (cont’d) l The uncertainty in position is then l No wonder one does not observe the effects of the uncertainty principle in everyday life!

Example of Electron l Same situation, but baseball replaced by an electron which has mass 9. 11 x 10 -31 kg traveling at 40 m/s l So momentum = 3. 6 x 10 -29 kg m/s and its uncertainty = 3. 6 x 10 -31 kg m/s l The uncertainty in position is then

Classical World l The observer is objective and passive l Physical events happen independently of whethere is an observer or not l This is known as objective reality

Role of an Observer in Quantum Mechanics l The observer is not objective and passive l The act of observation changes the physical system irrevocably l This is known as subjective reality

The Heisenberg Uncertainty Principle l l Whenever a measurement is made there is always some uncertainty Quantum mechanics limits the accuracy of certain measurements because of wave – particle duality and the resulting interaction between the target and the detecting instrument

4. The Heisenberg’s uncertainty principle • In the example of a free particle, we see that if its momentum is completely specified, then its position is completely unspecified • When the momentum p is completely specified we write: (because: and when the position x is completely unspecified we write: • In general, we always have: This constant is known as: (called h-bar) h is the Planck’s constant

So we can write: That is the Heisenberg’s uncertainty principle “ it is impossible to know simultaneously and with exactness both the position and the momentum of the fundamental particles” N. B. : • We also have for the particle moving in three dimensions • With the definition of the constant

Energy Uncertainty The energy uncertainty of a Gaussian wave packet is combined with the angular frequency relation l Energy-Time Uncertainty Principle: .

Derivation of Uncertainty Principle.

Derivation - Continued w

Derivation - Continued The condition for the formation of the node is that amplitude should be zero or

Derivation - Continued If x 1 and x 2 be the position of two consecutive nodes, then So that and

Derivation - Continued So uncertainity in measurement of position of the particle (x 1 -x 2)

Physical Origin of the Uncertainty Principle Heisenberg (Bohr) Microscope l l The measurement itself introduces the uncertainty When we “look” at an object we see it via the photons that are detected by the microscope l l l These are the photons that are scattered within an angle 2θ and collected by a lens of diameter D Momentum of electron is changed Consider single photon, this will introduce the minimum uncertainty
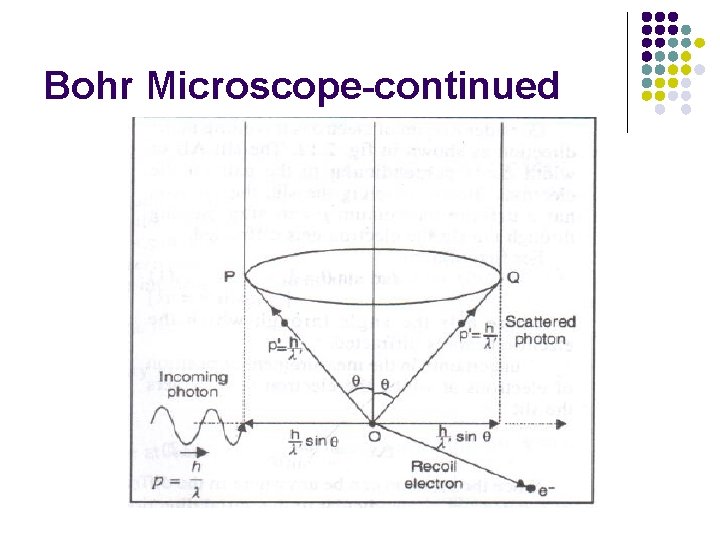
Bohr Microscope-continued

(a)Limitation in determing the position of electron =half angle subtended by the objective at the object i. e. electron

(b)Limitation in determining the momentum of the electron If photon is scattered along OQ, Then

If photon is scattered along OP, Then


Diffraction of Electron by a slit

Electron Diffraction -Continued

Electron Diffraction -Continued

Applications of Heisenberg uncertainty Principle-Non existence of electron in the nucleus Size of Nucleus =10 -14 m If electron is present in the nucleus uncertainty in the position of electron is =10 -14 m

The minimum momentum of the electron must be at least equal to uncertainty in momentum

Zero point Energy or minimum energy of a particle in the box The minimum energy of a system at 0 K is called zero point energy. Let a particle of mass m 0 is moving in a one dimensional box of length L So uncertainty of the position of the particle in the box Δx=L Uncertainty in the momentum Δp h/Δx = h/L

Minimum energy of a particle in the box Minimum momentum of the particle is at least equal to uncertainty in momentum p=Δp=ℏ/L K. E. of the particle is =p 2 /2 m 0 =(Δp)2 /2 m 0 K. E. =ℏ/ 2 m 0 L 2 This is the energy of the particle. Because the energy of the system is minimum at 0 K.

Minimum energy of a particle in the box Since the Δp 0 at 0 K, So the particle will have some energy even at 0 K. This minimum energy is called end point energy. So a particle confined to a region of space cannot have zero energy.

Binding energy of an electron Electron-revolving around the nucleous in an orbit of radius r So uncrtainity in the position of the electron is equal to the radius of the atom Δx=r Uncertainty in the momentum Δp h/Δx = h/r

Binding energy of an electron Minimum momentum of the particle is at least equal to uncertainty in momentum p=Δp=ℏ/r K. E. of the particle is =p 2 /2 m 0 =(Δp)2 /2 m 0 K. E. =ℏ/ 2 m 0 r 2

Binding energy of an electron The potetial energy of the electron in the field of the nucleus of atomic no Z is
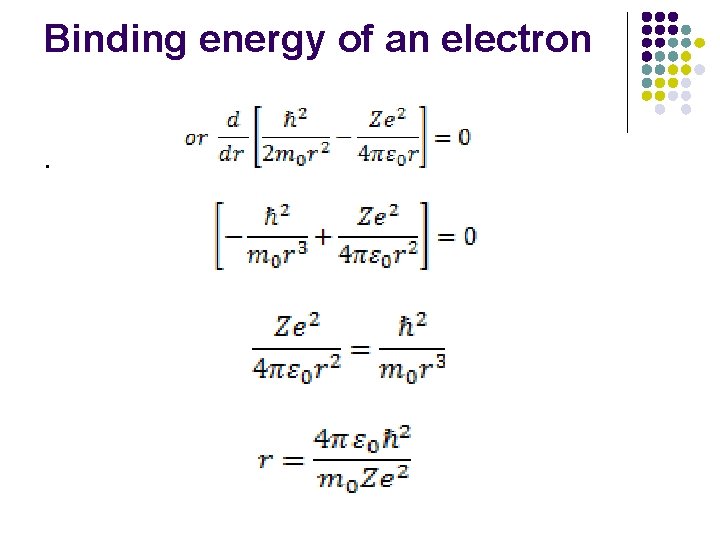
Binding energy of an electron.

Binding energy of an electron.

Binding energy of an electron.

Minimum Energy of the Hermonic Oscillator Consider a mechanical oscillator –spring attached at one end and mass attached at the other end. Classical mechanics-when at rest-energy of the oscillator is zero Quantum mechanics—Energy of the oscillator can never be zero. Let Δx is the displacement of the mass. Uncertainity in position= Δx

Harmonic Oscillator-Continued Uncertainty in the momentum Δp h/Δx Minimum momentum of the particle is at least equal to uncertainty in momentum p=Δp=ℏ/ Δx Energy of the Hermonic oscillator= PE+KE

Harmonic Oscillator-Continued.

Harmonic Oscillator-Continued.
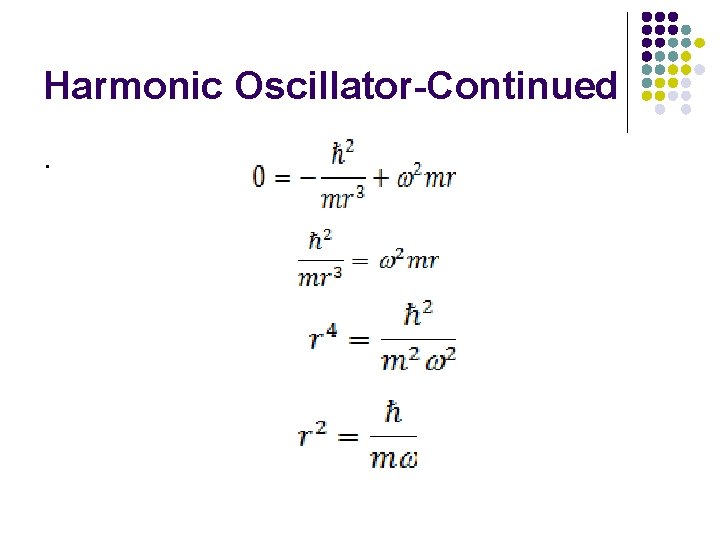
Harmonic Oscillator-Continued.

Harmonic Oscillator-Continued

THANKS
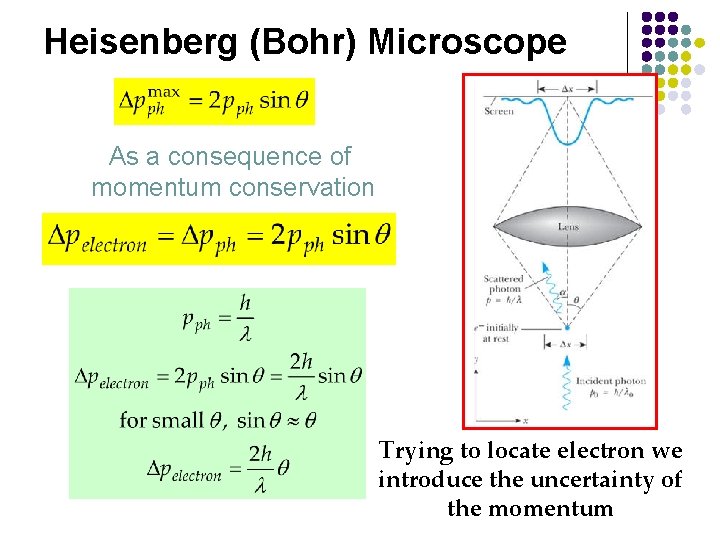
Heisenberg (Bohr) Microscope As a consequence of momentum conservation Trying to locate electron we introduce the uncertainty of the momentum
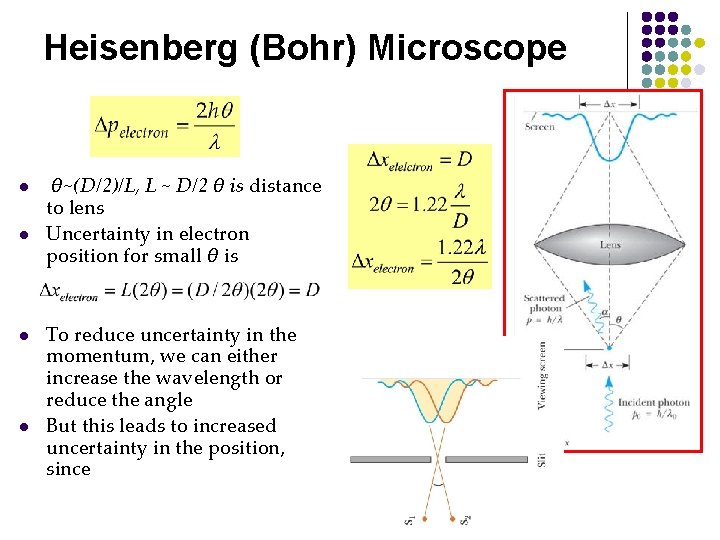
Heisenberg (Bohr) Microscope l l θ~(D/2)/L, L ~ D/2 θ is distance to lens Uncertainty in electron position for small θ is To reduce uncertainty in the momentum, we can either increase the wavelength or reduce the angle But this leads to increased uncertainty in the position, since
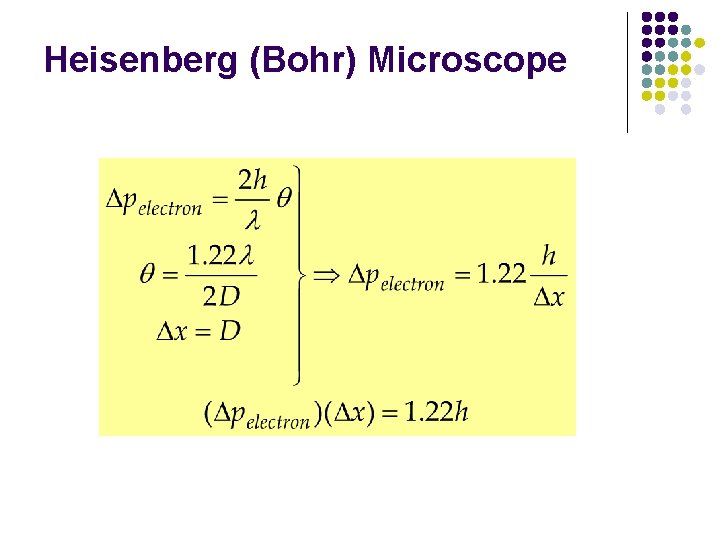
Heisenberg (Bohr) Microscope

PROBLEM 3 An electron is moving along x axis with the speed of 2× 106 m/s (known with a precision of 0. 50%). What is the minimum uncertainty with which we can simultaneously measure the position of the electron along the x axis? Given the mass of an electron 9. 1× 10 -31 kg SOLUTION From the uncertainty principle: if we want to have the minimum uncertainty: We evaluate the momentum: The uncertainty of the momentum is:

PROBLEM 4 In an experiment, an electron is determined to be within 0. 1 mm of a particular point. If we try to measure the electron’s velocity, what will be the minimum uncertainty? SOLUTION Observation: We can predict the velocity of the electron to within 1. 2 m/s. Locating the electron at one position affects our ability to know where it will be at later times

PROBLEM 5 A grain of sand with the mass of 1. 00 mg appears to be at rest on a smooth surface. We locate its position to within 0. 01 mm. What velocity limit is implied by our measurement of its position? SOLUTION Observation: The uncertainty of velocity of the grain is so small that we do not observe it: The grain of sand may still be considered at rest, as our experience says it should

PROBLEM 6 An electron is confined within a region of width 1. 0 10 - 10 m. (a) Estimate the minimum uncertainty in the x-component of the electron's momentum. (b) If the electron has momentum with magnitude equal to the uncertainty found in part (a), what is its kinetic energy? Express the result in jou 1 es and in electron volts. �� (a) (b) SOLUTION

PROBLEM 7 A sodium atom is in one of the states labeled ''Lowest excited levels". It remains in that state for an average time of 1. 6 10 -8 s before it makes a transition back to a ground state, emitting a photon with wavelength 589. 0 nm and energy 2. 105 e. V. What is the uncertainty in energy of that excited state? What is the wavelength spread of the corresponding spectrum line? SOLUTION The fractional uncertainty of the photon energy is

The Heisenberg Uncertainty Principle The Heisenberg uncertainty principle states that it is impossible to know both the momentum and the position of a particle at the same time. l This limitation is critical when dealing with small particles such as electrons. l But it does not matter for ordinary-sized objects such as cars or airplanes.

The Heisenberg Uncertainty Principle l l l To locate an electron, you might strike it with a photon. The electron has such a small mass that striking it with a photon affects its motion in a way that cannot be predicted accurately. The very act of measuring the position of the electron changes its momentum, making its momentum uncertain.
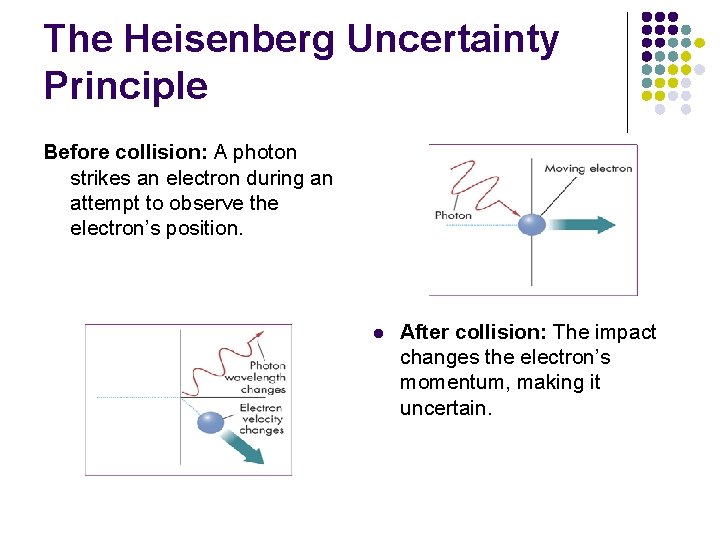
The Heisenberg Uncertainty Principle Before collision: A photon strikes an electron during an attempt to observe the electron’s position. l After collision: The impact changes the electron’s momentum, making it uncertain.

The Heisenberg Uncertainty Principle l l If we want accuracy in position, we must use short wavelength photons because the best resolution we can get is about the wavelength of the radiation used. Short wavelength radiation implies high frequency, high energy photons. When these collide with the electrons, they transfer more momentum to the target. If we use longer wavelength, i. e less energetic photons, we compromise resolution and position.
- Slides: 58