The Handed World 150 Years of Chiral Molecules














- Slides: 23

The Handed World: 150 Years of Chiral Molecules Royal Society of Chemistry Historical Group Thursday 19 th March 2020, Burlington House, London From d and l to (R) and (S): discovery of absolute configuration Henry Rzepa, Imperial College London rzepa@ic. ac. uk

The actors and their times: • Jacobus van't Hoff and Joseph Le. Bel (1874), the number of stereoisomers of an organic compound containing no internal planes of symmetry is 2 n, where n represents the number of asymmetric carbon atoms. • Emil Fischer, (1891). A symbolic way of drawing asymmetric carbon atoms (the Fischer projection ) • M. A. Rosanoff (1905) • Sir William Thomson Lord Kelvin (1894). I call any geometrical figure, or group of points, 'chiral', and say that it has chirality if its image in a plane mirror, ideally realized, cannot be brought to coincide with itself. • Werner Kuhn (1936, DOI: dntb). The absolute configuration of Lactic acid • John Kirkwood (1937, DOI: bmnfq 2). On the Theory of Optical Rotatory Power. • Derek Barton (1947), Conformational analysis • Johannes Bijvoet (1951). Determination of the Absolute Configuration of Optically Active Compounds by Means of X-Rays • Linus Pauling (1951). The structure of proteins: Two hydrogen-bonded helical configurations of the polypeptide chain • James Watson and Francis Crick (1953). Molecular Structure of Nucleic Acids: A Structure for Deoxyribose Nucleic Acid • R. S. Cahn, C. Ingold and Vladimir Prelog (1951 -56), The specification of asymmetric configuration in organic chemistry

Hermann Emil Fischer (Nobel prize, 1902) During 1875 – 1918 he stablished the molecular structural relationships between a host of natural products of biological importance, effectively founding the area of Biochemistry. Memorial lecture (1920), DOI: dnqfv 7

The Fischer Projection (1891, 10. 1002/Fcber. 189102402322) An important tool Fischer used was optical rotation (by a solution) of the plane of linearly polarized light using a Polarimeter (an instrument known since ~1809 -1815) Positive rotations were initially annotated as dextro-rotatory or d, but are now annotated as (+).

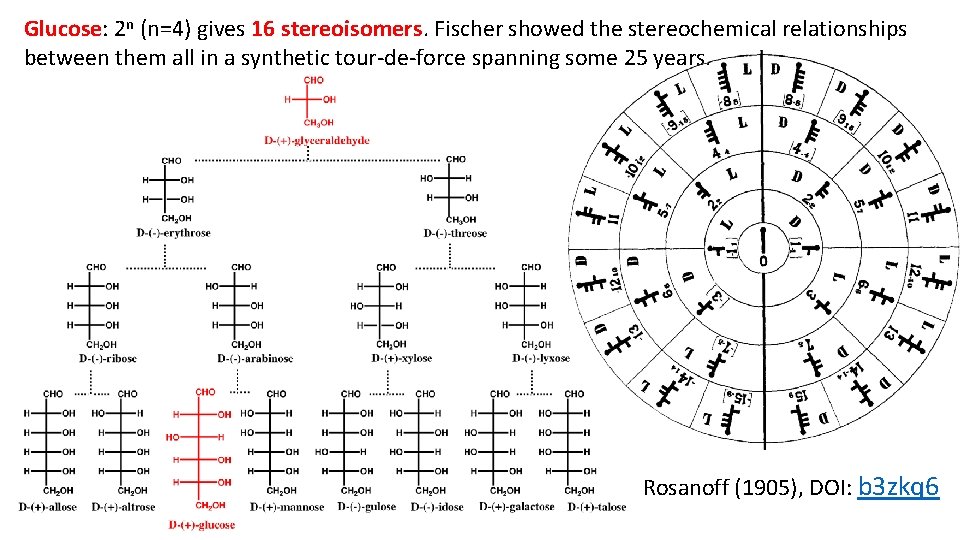
Glucose: 2 n (n=4) gives 16 stereoisomers. Fischer showed the stereochemical relationships between them all in a synthetic tour-de-force spanning some 25 years. Rosanoff (1905), DOI: b 3 zkq 6

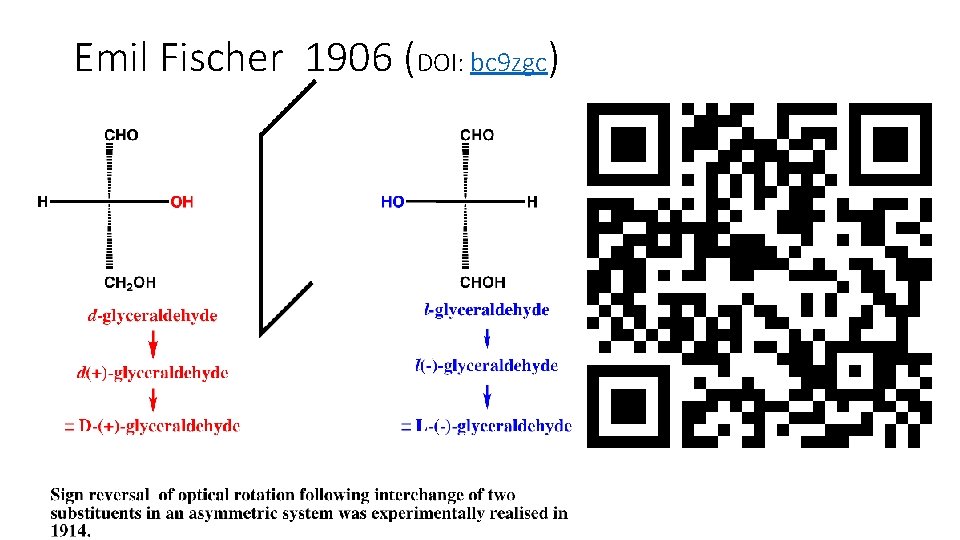
Emil Fischer 1906 (DOI: bc 9 zgc)

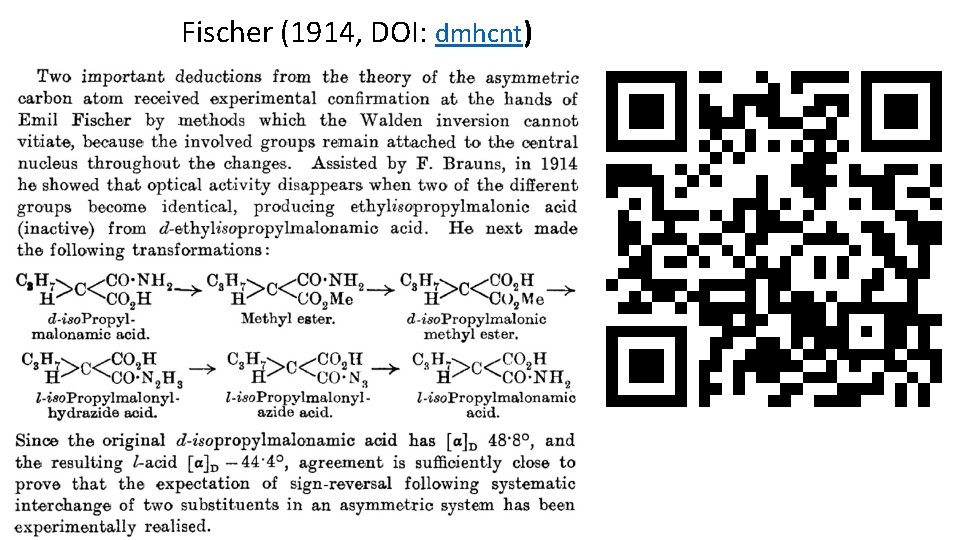
Fischer (1914, DOI: dmhcnt)

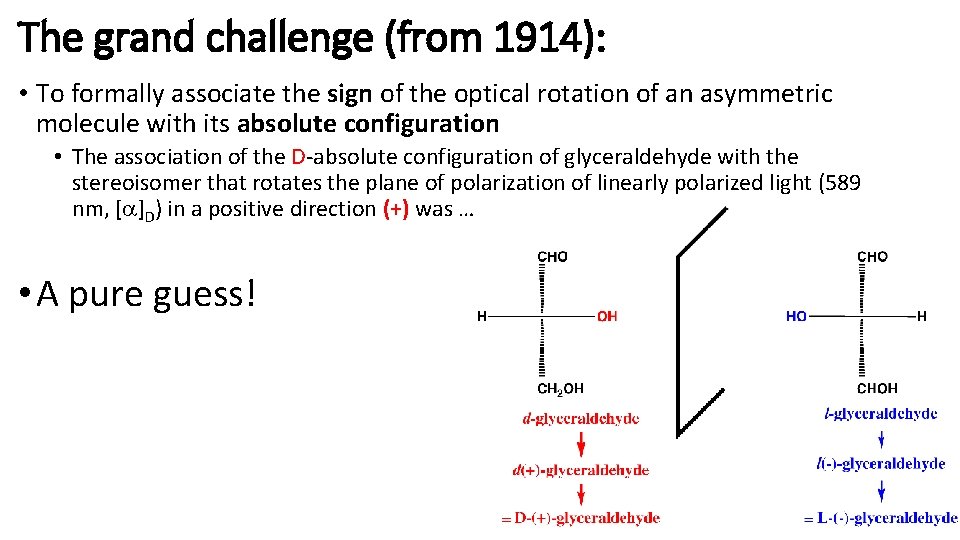
The grand challenge (from 1914): • To formally associate the sign of the optical rotation of an asymmetric molecule with its absolute configuration • The association of the D-absolute configuration of glyceraldehyde with the stereoisomer that rotates the plane of polarization of linearly polarized light (589 nm, [a]D) in a positive direction (+) was … • A pure guess!

Need a theory of optical rotation in which the sign can be computed, given only a knowledge of the absolute 3 D coordinates and nature of all the atoms. Einstein (1915): The use of theory to rationalize the anomalous precession of the perihelion of mercury Einstein (1915): The use of theory to rationalize the bending of light in gravitational fields Dirac (1928 -31): The use of quantum mechanical theory to predict the existence of antimatter

Werner Kuhn (1936, DOI: dntb): (-)-Lactic acid has a small (-5°) negative rotation at 589 nm and according to the Fischer hypothesis, this isomer is assigned to the D -absolute configuration. Note: d(-) in Fischer’s original notation is now D(-).

John Kirkwood (1937, DOI: bmnfq 2): On the Theory of Optical Rotatory Power • Takes issue with Kuhn’s theory and presents his own more rigorous method.

Kirkwood’s caveats:

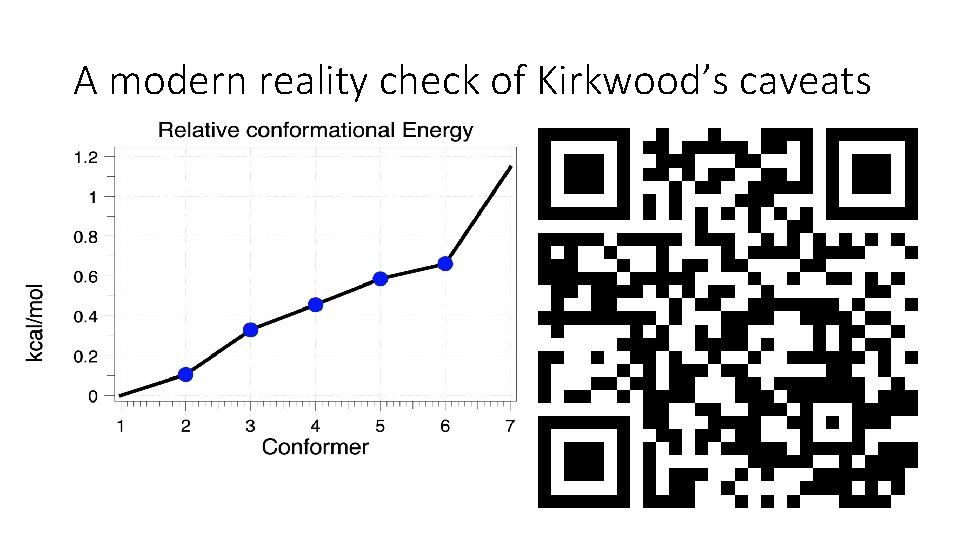
A modern reality check of Kirkwood’s caveats

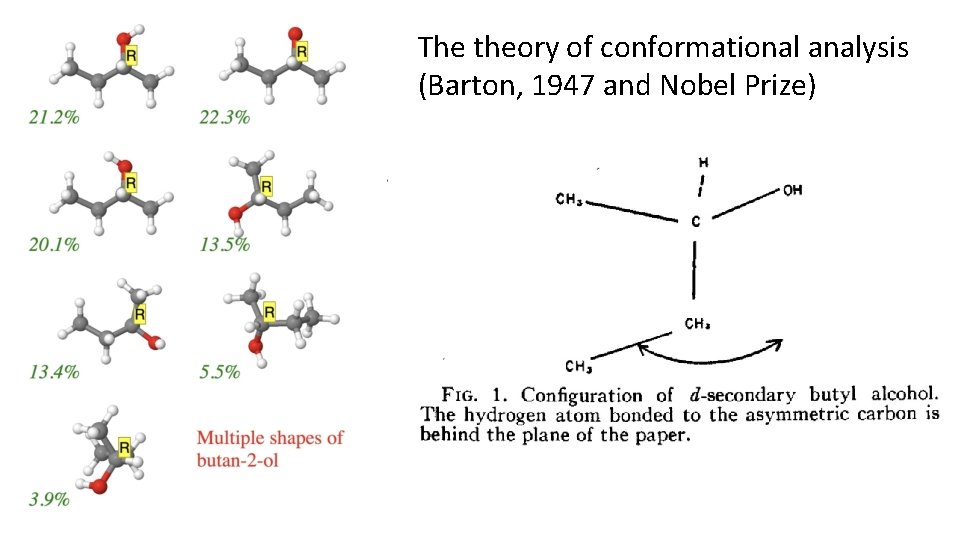
The theory of conformational analysis (Barton, 1947 and Nobel Prize)

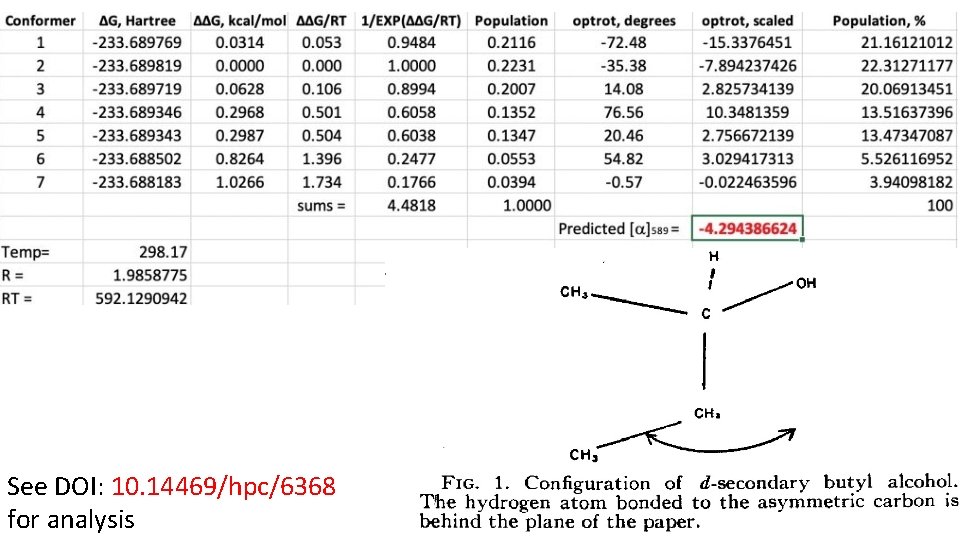
See DOI: 10. 14469/hpc/6368 for analysis

Lessons learned: • Kirkwood’s intuition that the shape of a molecule was important was correct • However, he chose to analyze a very flexible molecule with SEVEN populated shapes (conformations) • And he made a bad choice about the most populated one. • But this mistake led to better theories of “conformational analysis” in 1947, and ultimately the Nobel prize for these theories. • Did Barton know of Kirkwood’s mistake when presenting his own theory of conformational analysis?

Kirkwood (1952, DOI: bvth 8 c): learning from his mistake! • Choses a rigid molecule: 2, 3 -epoxybutane, which can be chemically related to D(+)-Glyceraldehyde. • Kirkwood calculates using his theory [α]D = +43° • Measured [α]D = +59° • Fischer’s hypothesis for D(+)-Glyceraldehyde was indeed (almost) correct! Too late! Johannes Bijvoet (1951, DOI: df 35 xj) using a new experimental technique based on anomalous scattering of X-rays on a solid specimen (of a rubidium tartrate salt) had shown a few months earlier that Fischer’s hypothesis for the absolute configuration of glyceraldehyde was indeed correct.

There is also the question of “signs”

A modern postscript: The (+) in D-(+)-glyceraldehyde means it has a positive optical rotation? Wrong! • D-(+)-glyceraldehyde actually has a negative rotation of -147 °(DOI: f 7 tdn 7) • The species that results in a (+) rotation is the hydrated diol, which constitutes 97% of what is present in solutions used for measurement. • Warning: Optical rotations in solution are VERY sensitive to conformation, solvation and hydration. • Another warning: Solutions and the crystalline state can be very different!

Cahn-Ingold-Prelog Stereochemical notation (1951 -56) • Cahn, R. S. ; Ingold, C. K. ; Prelog, V. The specification of asymmetric configuration in organic chemistry, DOI: ft 5 j 4 q • For a 2018 update (DOI: 10. 1021/acs. jcim. 8 b 00324 ), see Algorithmic Analysis of Cahn–Ingold–Prelog Rules of Stereochemistry: Proposals for Revised Rules and a Guide for Machine Implementation. • Tests: https: //cipvalidationsuite. github. io/Validation Suite/

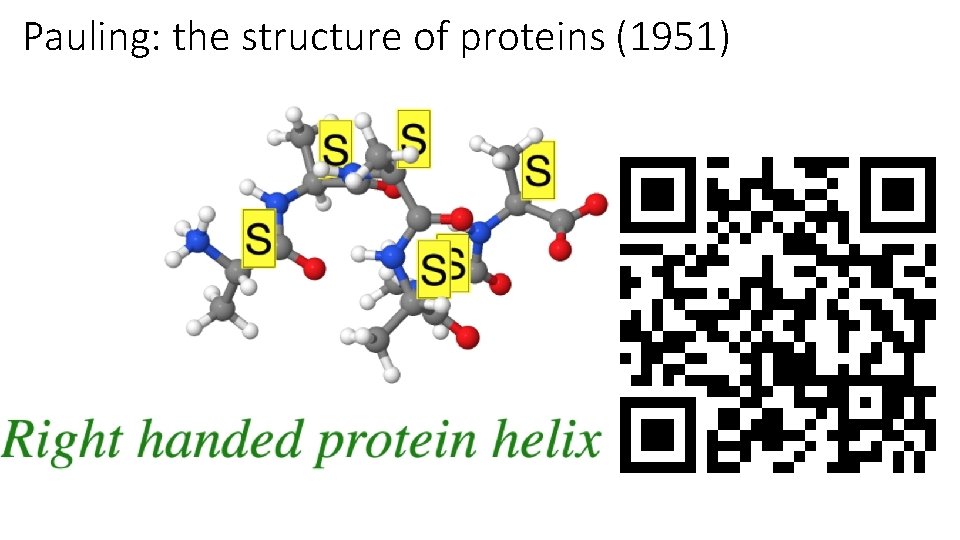
Pauling: the structure of proteins (1951)

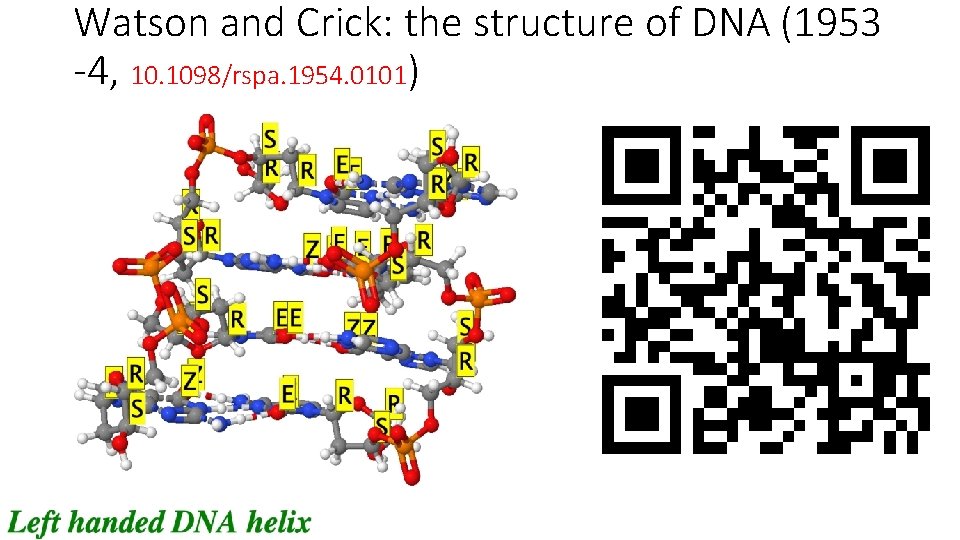
Watson and Crick: the structure of DNA (1953 -4, 10. 1098/rspa. 1954. 0101)

Some commentaries on the topics in this talk can be found at DOI: dns 9