The Haber Process The Haber Process The Haber

The Haber Process

The Haber Process • The Haber process is used to gain ammonia • Ammonia is used for: – Plant Fertilliser – Hair Dye
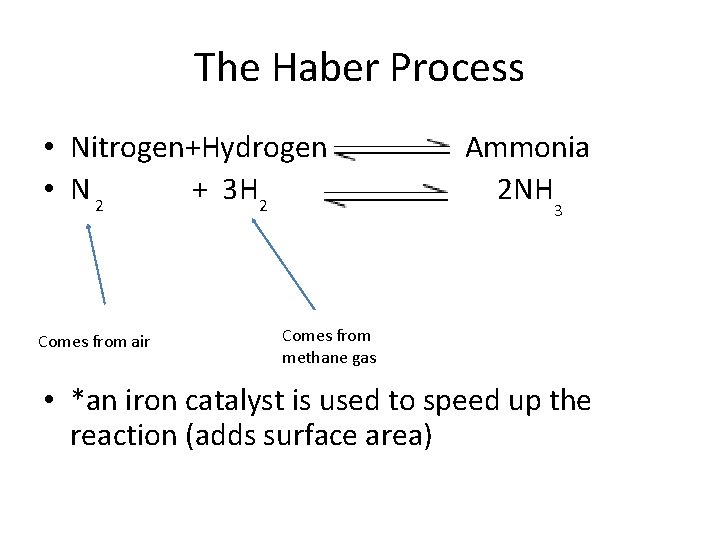
The Haber Process • Nitrogen+Hydrogen • N 2 + 3 H 2 Comes from air Ammonia 2 NH 3 Comes from methane gas • *an iron catalyst is used to speed up the reaction (adds surface area)
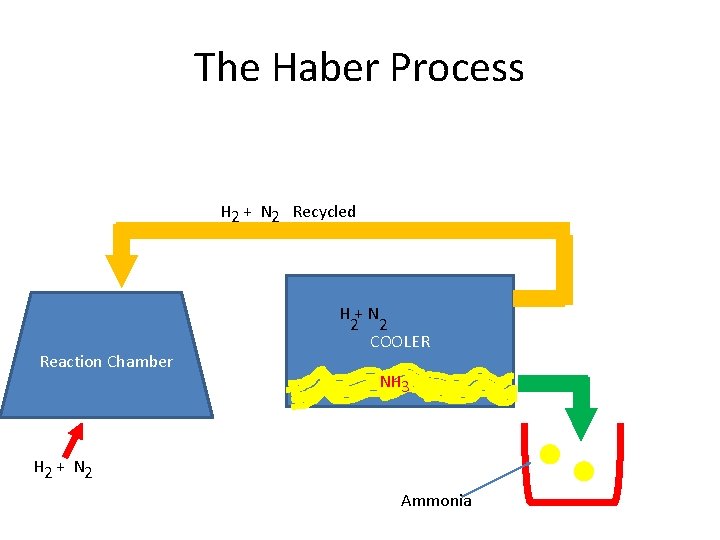
The Haber Process H 2 + N 2 Recycled Reaction Chamber H+N 2 2 COOLER NH 3 H 2 + N 2 Ammonia

What are the Ideal conditions to create Ammonia? • Highest yield (amount of product) is theoretically produced by using: – Low temperature – High Pressure • However these are not used. Why not?

Why are ideal conditions not used? • Low temperature – Slows the rate of reaction • High Pressure – Too Costly • So a compromise is reached. . .

The Compromise • Higher temp. used to increase rate of reaction (450 C) • Medium pressure used (200 atm)

Complete this paragraph using the words in the box • Ammonia is an important chemical used for making fertillisers. The raw materials are ______ from. Nitrogen the _____ and Air hydrogen Gasare reacted at about_____C and 450 from _____. Natural These 200 atm using an ______ Ammonia _____ is removed from Iron catalyst. _____ the reaction mixture before it can break down into the reactants again by. Liquefying ______ the gas. Nitrogen, 200, air, 450, liquefying, iron, Ammonia, Natural Gas

Task 2 • Create a flow chart NOT a diagram showing the Haber Process
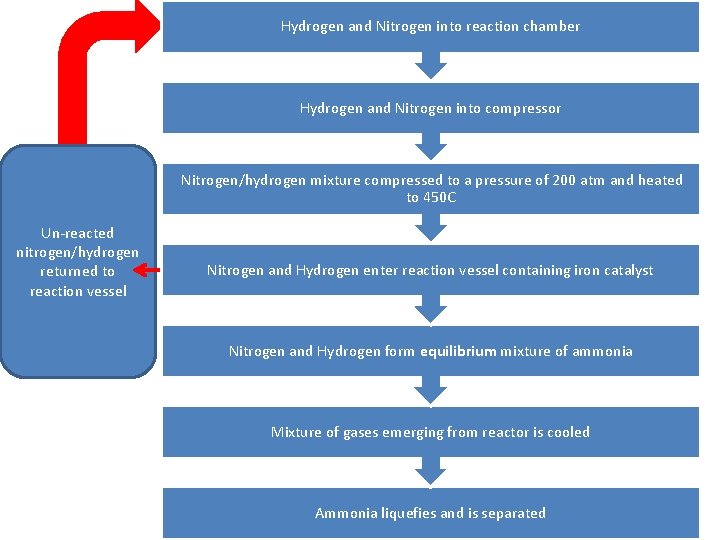
Hydrogen and Nitrogen into reaction chamber Hydrogen and Nitrogen into compressor Nitrogen/hydrogen mixture compressed to a pressure of 200 atm and heated to 450 C Un-reacted nitrogen/hydrogen returned to reaction vessel Nitrogen and Hydrogen enter reaction vessel containing iron catalyst Nitrogen and Hydrogen form equilibrium mixture of ammonia Mixture of gases emerging from reactor is cooled Ammonia liquefies and is separated
- Slides: 10