The growing role of CDK 4 6 inhibitors

The growing role of CDK 4 -6 inhibitors in therapeutic strategy of metastatic breast cancer: how to choose? Dott. Katia Cannita UOC Oncologia Medica Università degli Studi L’Aquila

• Estrogen is involved in the pathogenesis of breast carcinoma • Estrogen sustains the growth of breast cancer cells that express the receptor for this hormone • 75% of Breast cancer (BC) patients are ER+ and/or Pg. R +

Estrogen receptor (ER) and progesterone receptor (PR) expression are currently the best predictors of response to endocrine therapy in the clinical setting. However, 25% of ER+/PR+ tumors, 66% of ER+/PR– tumors, 55% of ER–/PR+ tumors fail to respond to tamoxifen or develop early resistance to tamoxifen for reasons that are unclear ( Moy and Goss, Clin canc res’ 06).
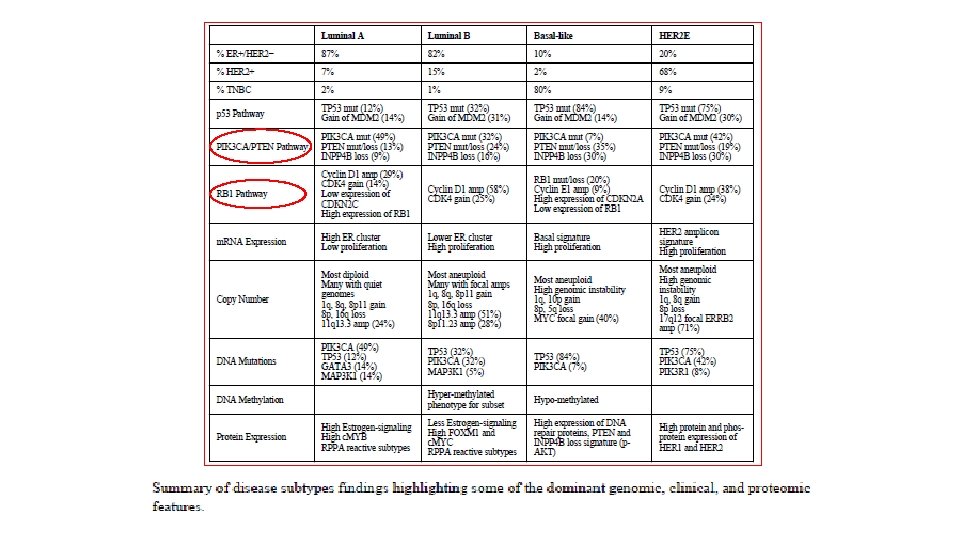

The role of CDK 4/6 in Breast Cancer I. A. /Fulvestrant ER/Pg. R • Cyclin D–CDK 4/6 complexes ciclib NF-k. B Pl 3 K/m. TOR p 5 3 p 21 MAPKs STATs Wnt/β-catenin p 16 Rb G 1 E 2 F M G 0 Restriction Spoint E 2 F • • promote cell proliferation through Rb protein phosphorylation 1 Increased CDK 4/6 activity is frequently observed in HR+ breast cancer 1, 2 Activation of the cyclin D–CDK 4/6– INK 4–Rb pathway has been associated with poor response and resistance to endocrine therapy 1, 2 G 2 1. Hosford S, Miller T. Pharmgenomics Pers Med 2014; 7: 203– 215; 2. Thangavel C, et al. Endocr Relat Cancer 2011; 18: 333– 345; 3. Kim S, et al. Mol Cancer Ther 2013; 12(Suppl 11): abst PR 02 (oral). CDK, cyclin-dependent kinase; HR+, hormone receptor-positive; INK 4, inhibitor of CDK 4; Rb, retinoblastoma.

Treatment for HR+, HER 2– Advanced Breast Cancer Recent Advances and Upcoming Therapies

HR+, HER 2─ Advanced Breast Cancer: Where Are We Now? • Investigations into CDK 4/6 inhibition in combination with endocrine therapy have significantly altered the treatment landscape for patients with HR+, HER 2─ ABC 1 ─ In the first-line setting, the addition of CDK 4/6 inhibitors to endocrine therapy significantly improves PFS, ORR, and CBR versus endocrine therapy alone 2 ABC, advanced breast cancer; ABE, abemaciclib; CBR, clinical benefit rate; CDK, cyclin-dependent kinase; ET, endocrine therapy; FUL, fulvestrant; HER 2–, human epidermal growth factor receptor-2–negative; HR, hormone receptor; LET, letrozole; NSAI, nonsteroidal aromatase inhibitor; OFS, ovarian function suppression; PAL, palbociclib; PBO, placebo; PFS, progression-free survival; RIBO, ribociclib. 1. Ramos-Esquivel A, et al. Breast Cancer. 2018. doi: 10. 1007/s 12282 -018 -0848 -6 [Epub ahead of print]; 2. clincialtrials. gov.

How and When to incorporate CDK 4/6 Inh in the managemenet of HR+ MBC Remain one the main unmeet clinical need in this setting ? ? ? ET naive Non visceral disease Long DFI CIRS, old patient with no good caregiver

Ciclibs: NOT if but when Tamoxifen 105€ year Letrozole 880€ year Fulvestrant Ciclibs 6. 000€ year 25. 000€ year . . . it is clinically necessary, scientifically wrong, ethically acceptable and financially sustainable to prescribe everything to everyone. . . Davis C et al. BMJ 2017; 359: j 4530; Cancerworld Spring 2018; 4 -11; Zhang B et al. Breast Cancer Res Treat 2019; epub ahead of print

LG AIOM 2019 Palbo o Ribo o Abema+ NSAI Fulvestrant I. A. Palbo o Abema + Fulvestrant Everolimus + exemestane Fulvestrant Ripresa di malattia tardiva > 12 mesi dal termine adiuvante Precedente esposizione a Terapia endocrina in adiuvante SI Ripresa di malattia precoce < 12 mesi dal termine dell’adiuvante o in corso di adiuvante I line II line

HOW TO CHOOSE?


Conclusions: CDK 4/6 produced significant improvement in PFS, CB, ORR in all pts The results are indipendent of age, race, PS, Disease site, prior Ct or ET Indipendent of menopausal status, DFI, CDK 4/6 Inhibitor, and Pg. R expression Improvement of PFS in favor of CDK 4/6 inh even in pts with bone only disease or in non visceral disease

CDK 4/6 Inh Trials In First Line Setting • PALOMA 2 • MONALEESA 2 • MONARCH 3 HOW TO CHOSE ? Premenopausal status • MONALEESA 7
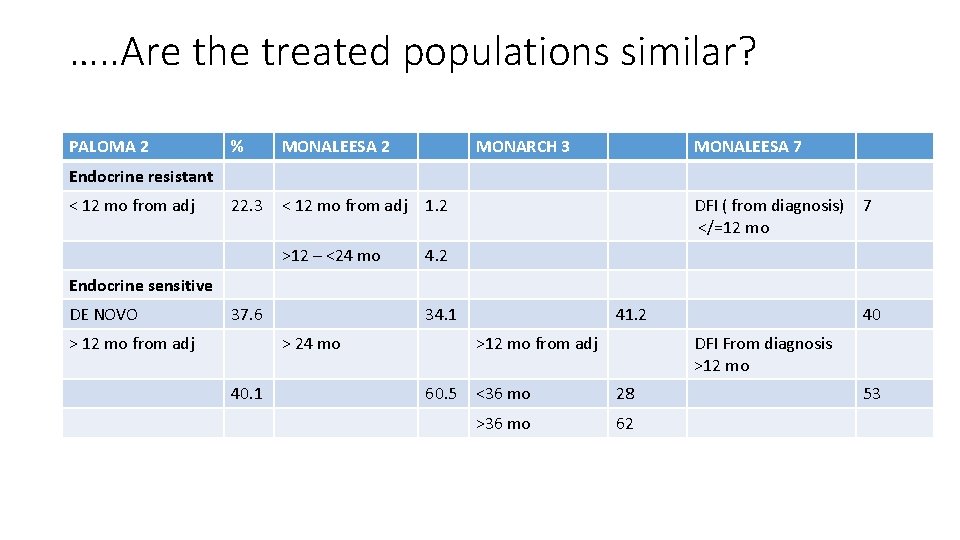
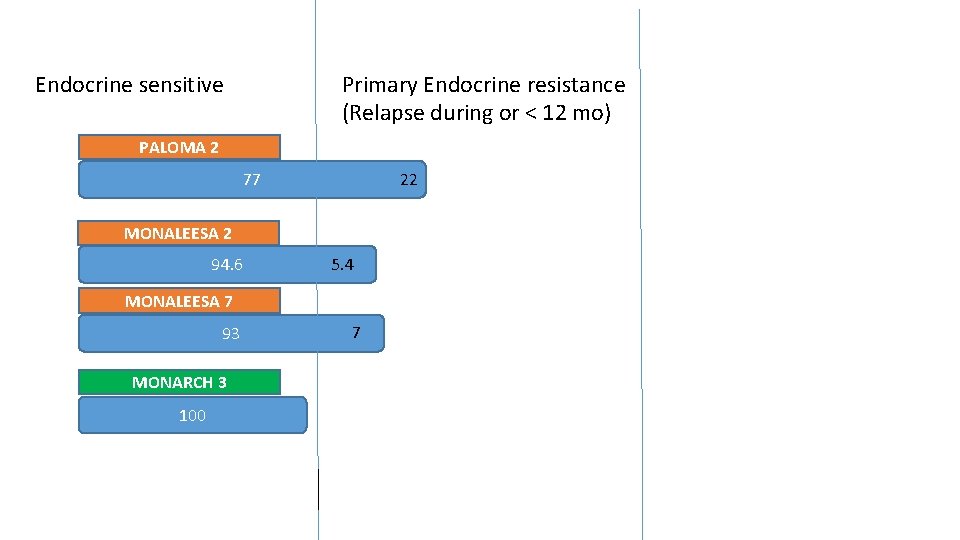
…. . Are the treated populations similar? PALOMA 2 % MONALEESA 2 MONARCH 3 22. 3 < 12 mo from adj 1. 2 >12 – <24 mo 4. 2 MONALEESA 7 Endocrine resistant < 12 mo from adj DFI ( from diagnosis) </=12 mo 7 Endocrine sensitive DE NOVO 37. 6 > 12 mo from adj 34. 1 > 24 mo 40. 1 41. 2 >12 mo from adj 60. 5 40 DFI From diagnosis >12 mo <36 mo 28 >36 mo 62 53
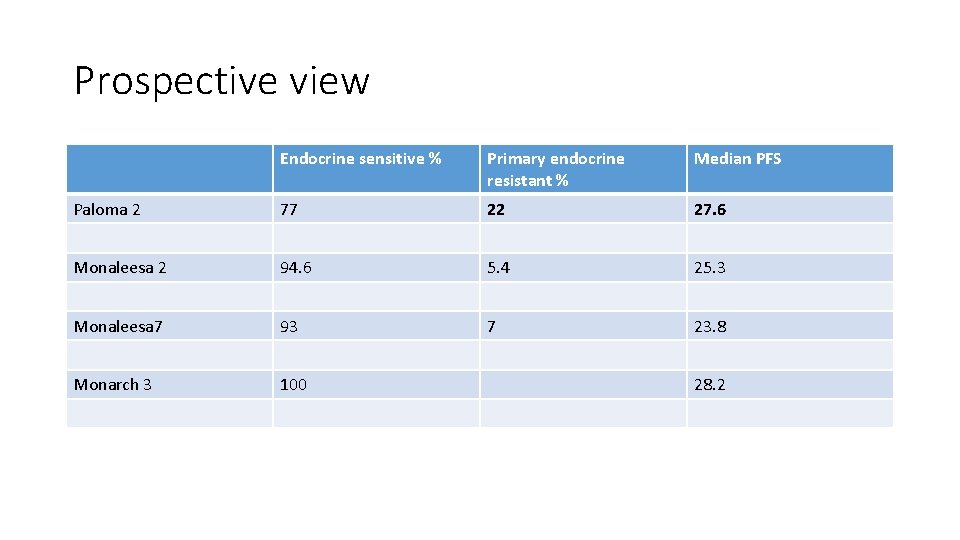
Prospective view Endocrine sensitive % Primary endocrine resistant % Median PFS Paloma 2 77 22 27. 6 Monaleesa 2 94. 6 5. 4 25. 3 Monaleesa 7 93 7 23. 8 Monarch 3 100 28. 2
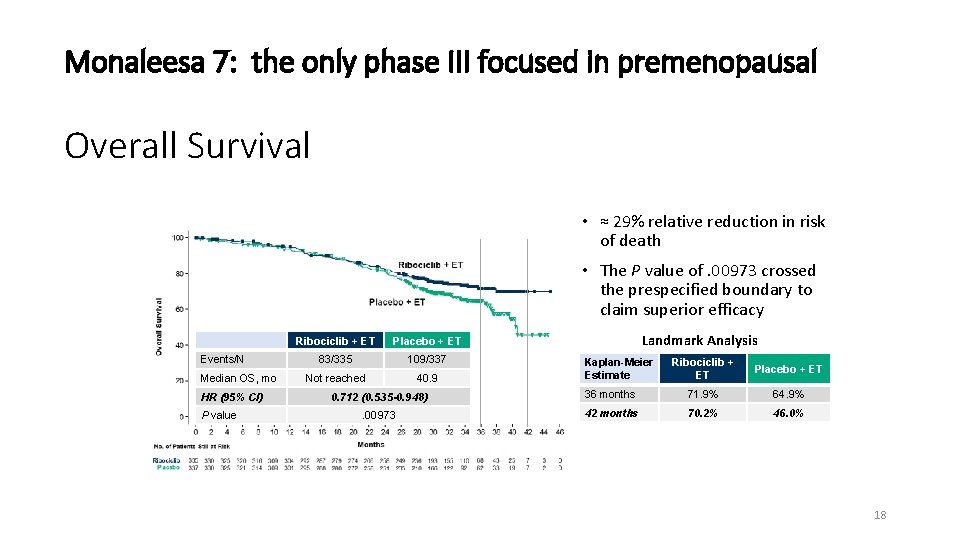
Monaleesa 7: the only phase III focused in premenopausal Overall Survival • ≈ 29% relative reduction in risk of death • The P value of. 00973 crossed the prespecified boundary to claim superior efficacy Events/N Median OS, mo HR (95% CI) P value Ribociclib + ET Placebo + ET 83/335 109/337 Not reached 40. 9 Landmark Analysis Kaplan-Meier Estimate Ribociclib + ET Placebo + ET 0. 712 (0. 535 -0. 948) 36 months 71. 9% 64. 9% . 00973 42 months 70. 2% 46. 0% Dr Sara Hurvitz 18
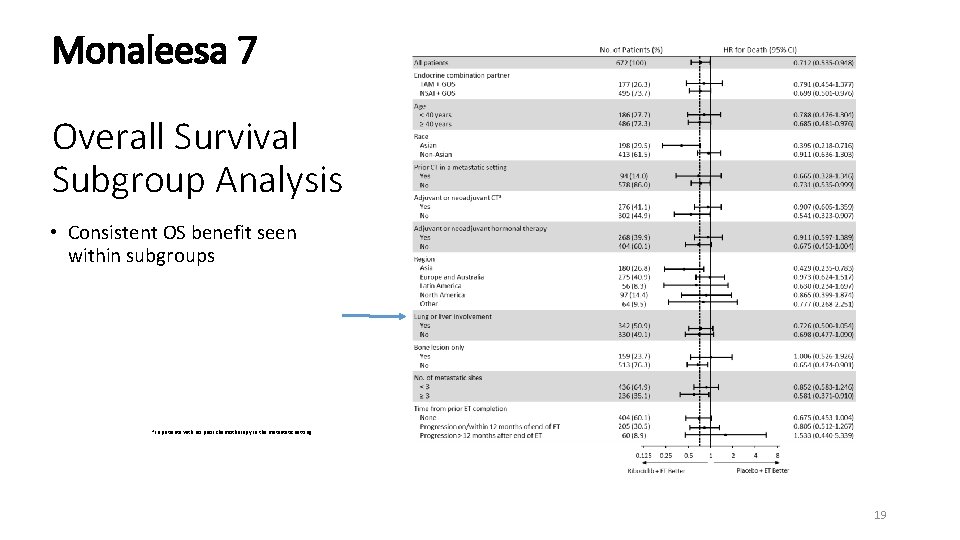
Monaleesa 7 Overall Survival Subgroup Analysis • Consistent OS benefit seen within subgroups a In patients with no prior chemotherapy in the metastatic setting. 19

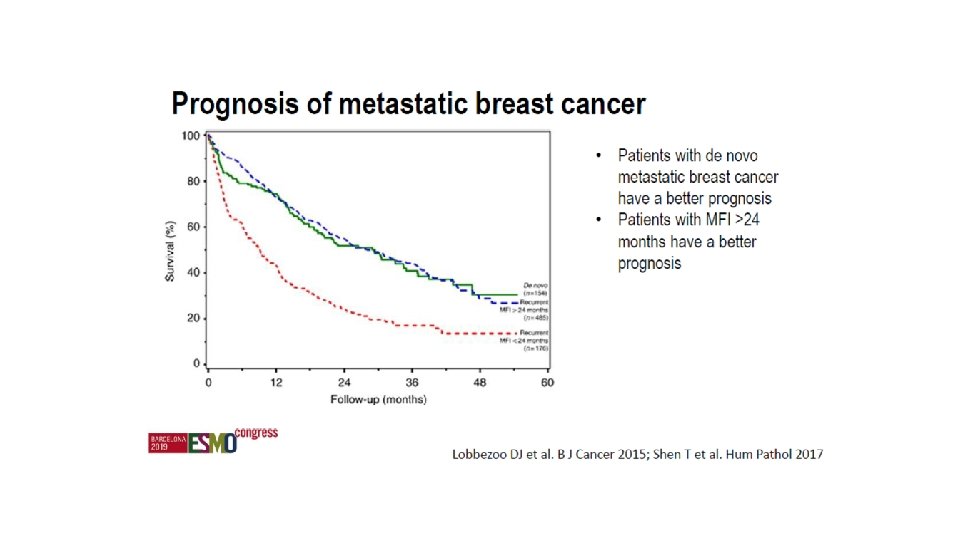
Patient populations in trials and OS



Results for Sensitive vs Non sensitive pts


HOW to CHOSE Molecular mechanism: Abema inhibitor of CDK 2/ciclin E/A (resistance in palbo) Activity in p. RB deficient Profile toxicities

Registrative trials CDK 4/6 inhibitors Main toxicities % G 3 -G 4 neutropenia % Diarrhea % G 3 -G 4 AST and/or ALT elevation % G 1 -G 4 creatinine elevation % QTc. F interval increase (>60 msec) % Thromboembolic events Palbociclib* Ribociclib** Abemaciclib*** 66 59 21 1(G 3) 36(G 2=27/G 3=9) 3 9 7 NR NR 19 (G 3/G 4=2/0) <1 3 NR 1 3 5 *Finn RS et al New Engl J Med 2016; **Hortobagyi G et al, New Engl J Med 2016; ***Goetz MP t al, J Clin Oncol 2017

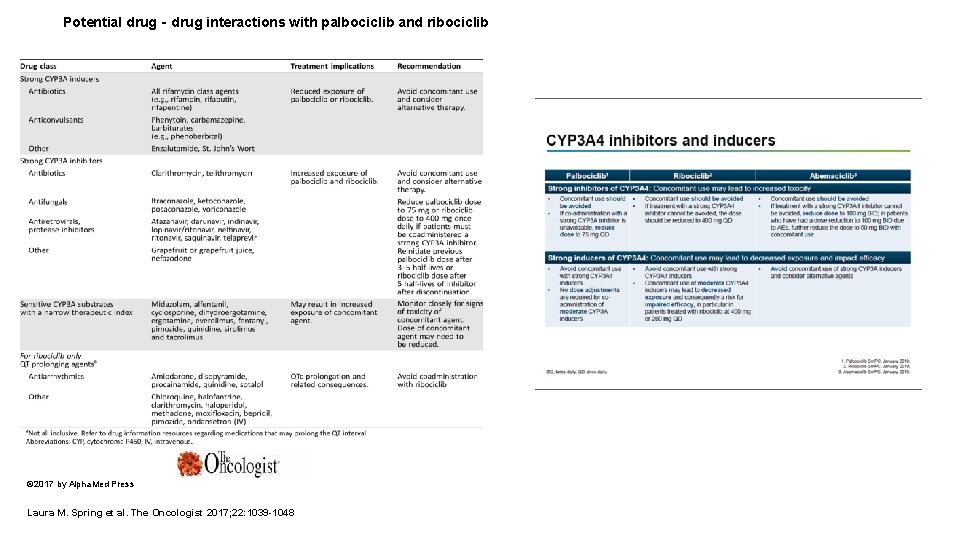
Potential drug‐drug interactions with palbociclib and ribociclib © 2017 by Alpha. Med Press Laura M. Spring et al. The Oncologist 2017; 22: 1039 -1048
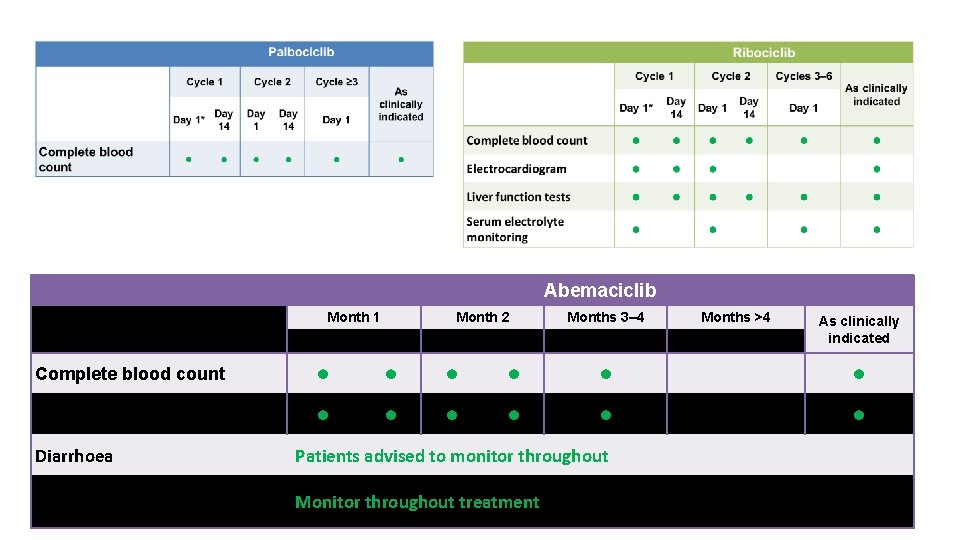
Abemaciclib Month 1 Complete blood count Liver function tests Diarrhoea Month 2 Months 3– 4 Months >4 Day 1 Baseline Day 14 Day 1 • • • Patients advised to monitor throughout Venous thromboembolism and Monitor throughout treatment pulmonary embolism As clinically indicated • •

Conclusions HOW CHOSE? Trials cannot be compared • Different population • Different subgoup Profile of toxicities Drug interaction Patients preferences are probably the only parameter for decision making process

Conclusions • Endocrine sensitive + bone only : Palbociclib and Ribociclib ? • Endocrine resistant and visceral disease: Abemaciclib? • Premenopausal: Ribocilcib? Combine all CDK 4/6 clinical trial data (in 1 st and 2 nd line) and perform a meta-analysis to reveal any potential differences in subgroups

Grazie !


Endocrine sensitive Primary Endocrine resistance (Relapse during or < 12 mo) PALOMA 2 22 77 MONALEESA 2 94. 6 5. 4 MONALEESA 7 7 93 MONARCH 3 100 26

- Slides: 36