The Greenhouse Effect Terrestrial Emissions Terrestrial emissions have









- Slides: 15

The Greenhouse Effect

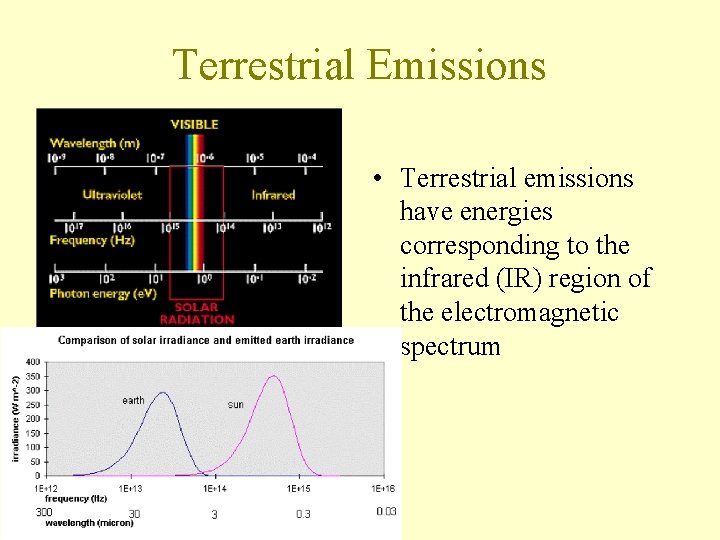
Terrestrial Emissions • Terrestrial emissions have energies corresponding to the infrared (IR) region of the electromagnetic spectrum

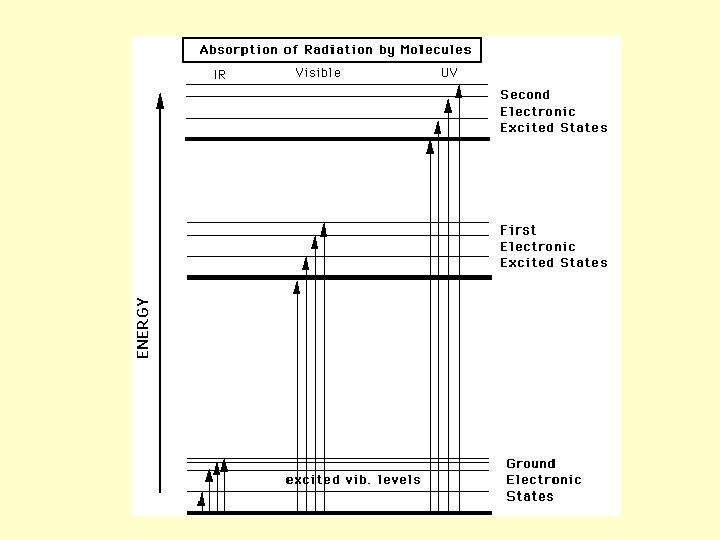
Visible & UV Radiation • Certain energies cause electrons to be excited into higher energy orbitals • Very energetic photons (uv to x-ray) may cause electrons to be ejected from molecule (ionization)

IR Radiation • Lower in energy than visible radiation • Does not possess enough energy to eject electrons • Absorbed IR radiation can excite vibrations in molecules


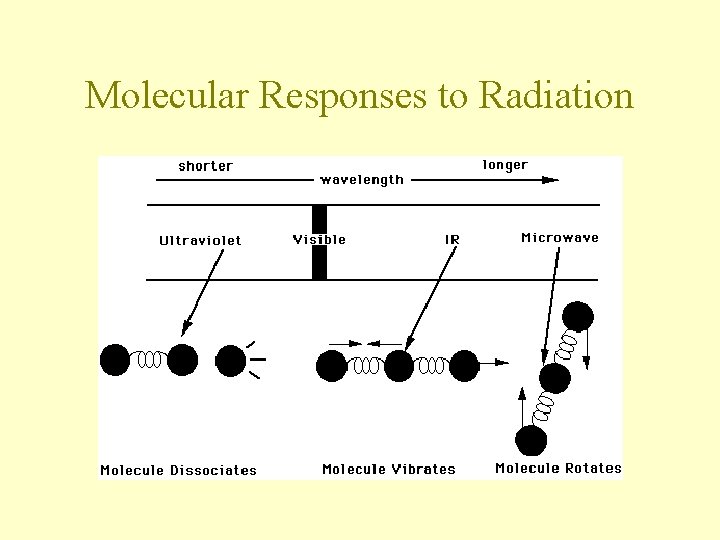
Molecular Responses to Radiation

What Are Molecular Vibrations? • Consider a diatomic molecule as being 2 balls connected by a spring • When the molecule vibrates, the balls move towards and away from each other at a certain frequency • The energy of the system is related to the amount the spring stretches or compresses

• The frequency of the vibration is proportional to the square root of the ratio of the spring force constant to the masses on the string. – the lighter the masses on the spring, the higher the vibrational frequency – the tighter the spring, the higher the vibrational frequency • Vibrational frequencies for stretching bonds in molecules are related to: – bond strength – masses of atoms

• Molecules differ from balls & springs because their vibrational frequencies are quantized • Only certain energies are allowed • only photons of certain energies will excite molecular vibrations

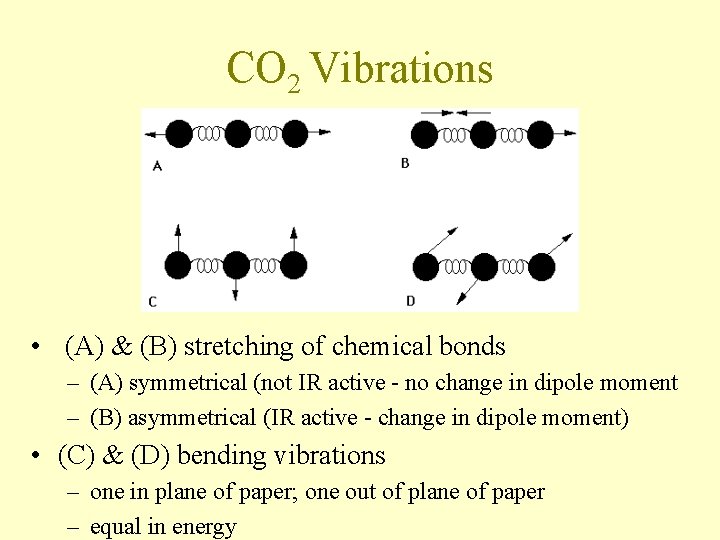
Number of Vibrational Modes • # different types of vibrations in an N atom molecule – linear molecule • 3 N - 5 – nonlinear molecule • 3 N - 6 • Diatomic molecule (3 x 2) - 5 = 1 (stretching of bond) • CO 2 – (3 x 3) - 6 = 4

CO 2 Vibrations • (A) & (B) stretching of chemical bonds – (A) symmetrical (not IR active - no change in dipole moment – (B) asymmetrical (IR active - change in dipole moment) • (C) & (D) bending vibrations – one in plane of paper; one out of plane of paper – equal in energy

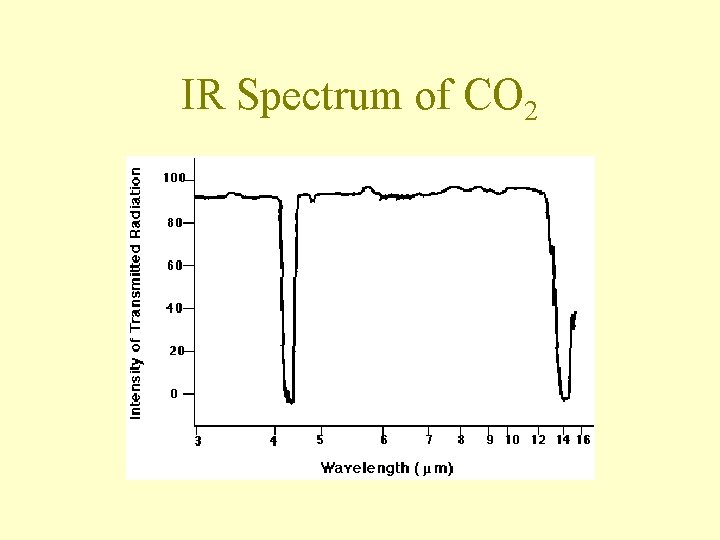
IR Spectrum of CO 2

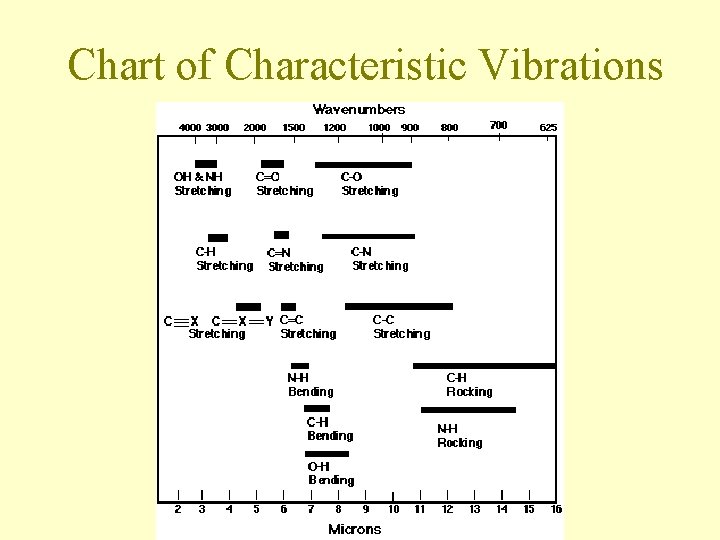
General Trends • The stronger the bond, the more energy required to excite the stretching vibration – frequency decreases in the order triple bond > double bond > single bond • The heavier an atom, the lower the frequencies of vibrations involving that atom

General Trends • Polar molecules and molecules with polar bonds capture IR photons more efficiently than nonpolar molecules • All molecules with three or more atoms absorb IR radiation because they all have some vibrations that change the polarity of the molecul

Chart of Characteristic Vibrations