The Goldstein Canal Ray Tube This tube demonstrates
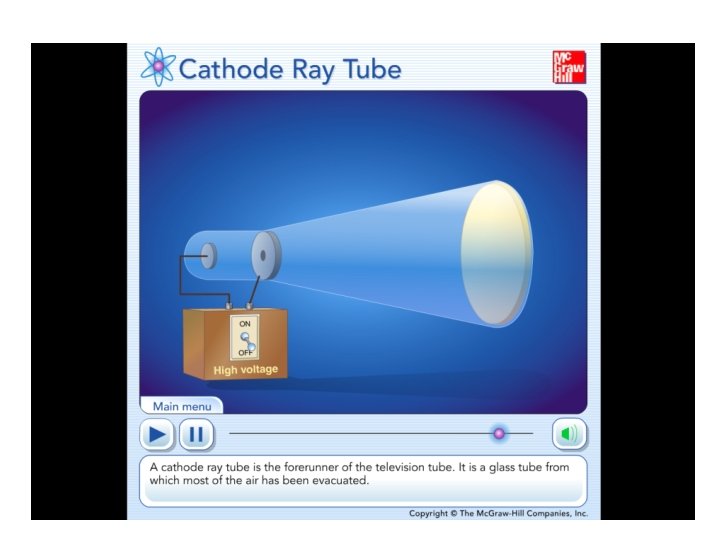


The Goldstein Canal Ray Tube • This tube demonstrates that besides the cathode rays there is another stream that travels in the opposite direction as the electron flow. • Discovered in 1886 by Eugen Goldstein (1850 - 1931) who named this"canal rays". In fact these are positively charged protons, producing a reddish light in the upper part of the tube while in the lower part the usual green emission of electrons can be seen when they hit the glass wall. • The electrons in the lower part of the tube can be deflected bya magnetic field but the canal rays almost not. Goldstein could not explainthis phenomenon, it took 12 years before Goldstein's paper was published.
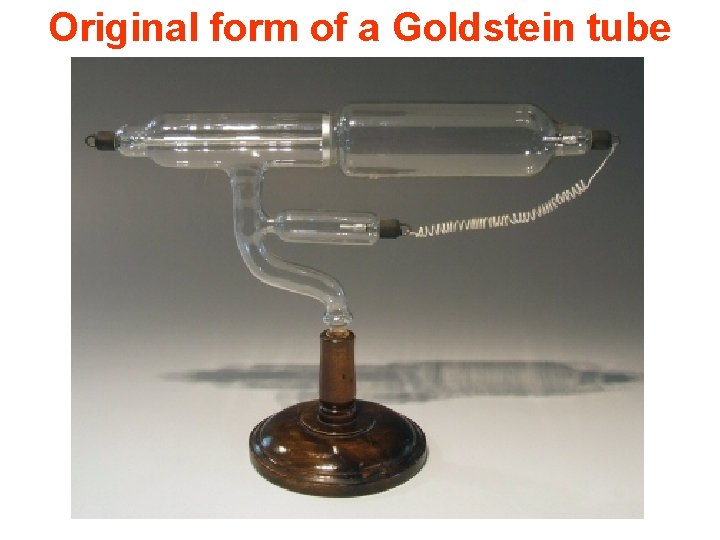
Original form of a Goldstein tube
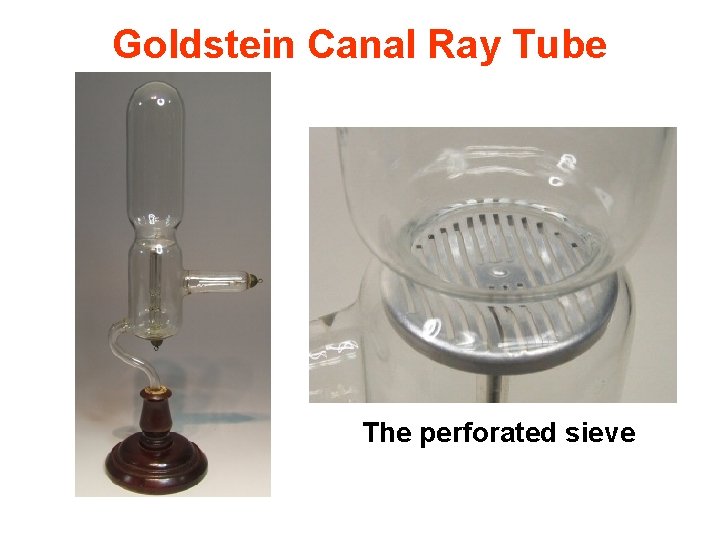
Goldstein Canal Ray Tube The perforated sieve
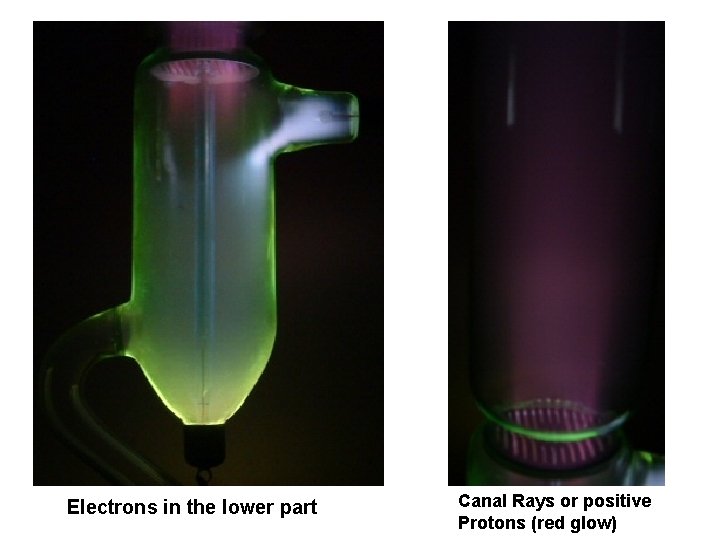
Electrons in the lower part Canal Rays or positive Protons (red glow)

Canal Rays • When the cathode of a cathode-ray tube was perforated, Goldstein observed rays he called "canal rays, " which passed through the holes, or channels, in the cathode to strike the glass walls of the tube at the end near the cathode. • Since these canal rays travel in the opposite direction from the cathode rays, they must carry the opposite charge.

Canal Rays • In 1886 Eugen Goldstein noted that cathode-ray tubes with a perforated cathode emit a glow from the end of the tube near the cathode. • Goldstein concluded that in addition to the electrons, or cathode rays, that travel from the negatively charged cathode toward the positively charged anode, there is another ray that travels in the opposite direction, from the anode toward the cathode. • Because these rays pass through the holes, or channels, in the cathode, Goldstein called them canal rays.

DISCOVERY OF PROTON POSITIVE RAYS OR CANAL RAYS • Atoms are electrically neutral. • Hence after the discovery of the negatively charged constituent (electron) of an atom, attempts were made to discover the positively charged counterpart of electrons. • By using a discharge tube containing a perforated cathode. Goldstein (1886) found that some rays passed through these holes in a direction opposite to that of the cathode rays.



Properties of Canal Rays

Properties of Canal Rays

Properties of Canal Rays


Wien's Canal Ray tube • Wien's Canal Ray tube is named after it's inventor Wilhelm Wien did several experiments from 1897 -1912, his experiments were similar to JJ. Thomson almost the same time. • This tube has some extra electrodes compared to the Goldstein Canal Ray tube. • To connect the tube in different way's to a galvanometer the positive or negative charged particles can be measured by experiment.

The sieve with tiny holes. Wien's Canal Ray tube

Canal rays can be seen in the left of the picture.

References • http: //www. crtsite. com/index. html • http: //kr. cs. ait. ac. th/~radok/physics/k 10. htm • http: //www. askiitians. com/iit_jee. Atomic_Structure/Discovery_of_Proton
- Slides: 19