The genetic basis for the development of cancer

The genetic basis for the development of cancer Molecular Biology of Cancer 1

u. Cancers arise through a multistage process in which inherited and somatic mutations of cellular genes lead to clonal selection of variant progeny with the most robust and aggressive growth properties. u. Two classes of genes are targets for the mutations: w Protooncogenes w tumor-suppressor genes Molecular Biology of Cancer 2

u. The vast majority of the mutations that contribute to the development and behavior of cancer cells w are somatic (ie, arising during tumor development) w present only in the neoplastic cells of the patient u. A small fraction of all mutations in cancer cells are constitutional w present in all somatic cells of affected individuals w such mutations not only predispose to cancer, but can also be passed on to future generations. Molecular Biology of Cancer 3

Tumor Suppressor Genes Molecular Biology of Cancer 4

A large number of tumor suppressor genes have been hypothesized to exist u. Thus far, approximately 20 tumorsuppressor genes have been identified and definitively implicated in cancer development. u. The cellular functions of the tumorsuppressor genes appear to be diverse Molecular Biology of Cancer 5

Cancer-inducing genes, specifically viral oncogenes, act in a dominant fashion u. Viral oncogenes dictate cellular behavior in spite of the continued presence and expression of opposing cellular genes within the virusinfected cell that usually functioned to ensure normal cell proliferation. w The viral genes were able to induce a dominant phenotype—they were bringing about a cell transformation. u. Most human cancers do not seem to arise as consequences of tumor virus infections Molecular Biology of Cancer 6
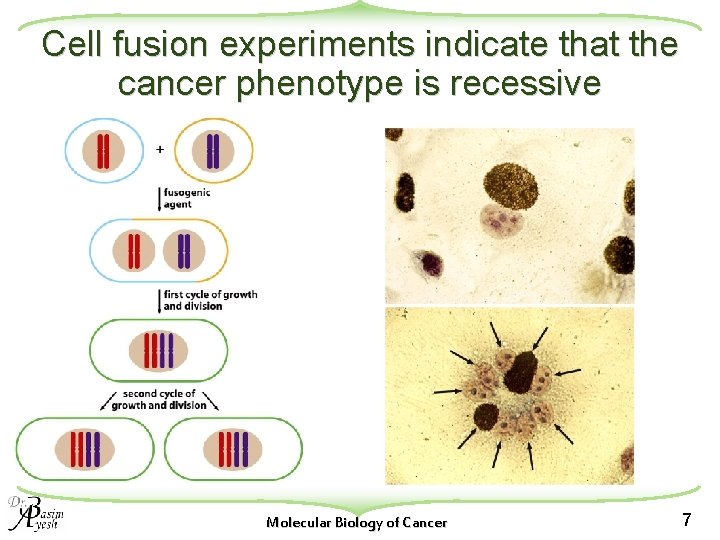
Cell fusion experiments indicate that the cancer phenotype is recessive Molecular Biology of Cancer 7
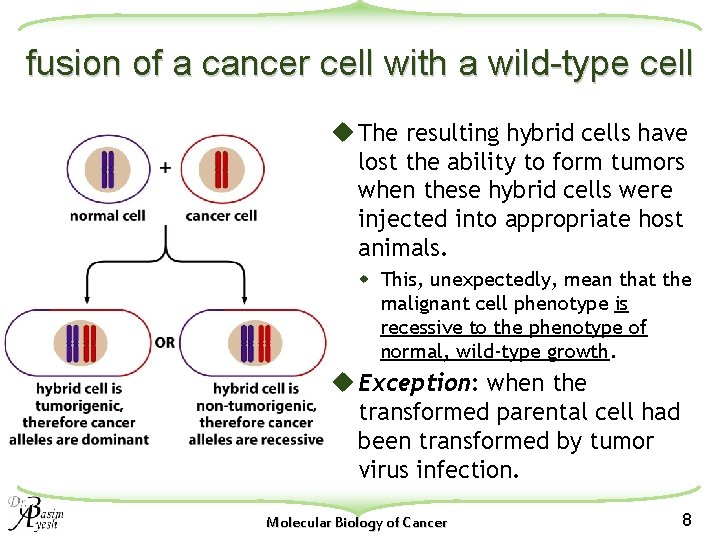
fusion of a cancer cell with a wild-type cell u The resulting hybrid cells have lost the ability to form tumors when these hybrid cells were injected into appropriate host animals. w This, unexpectedly, mean that the malignant cell phenotype is recessive to the phenotype of normal, wild-type growth. u Exception: when the transformed parental cell had been transformed by tumor virus infection. Molecular Biology of Cancer 8

Tumor suppressor genes (TSGs) Hypothesis u. Normal cells carry genes that constrain or suppress their proliferation. w During the development of a tumor, the evolving cancer cells inactivate one or more of these genes. w Once these growth-suppressing genes are lost, the proliferation of the cancer cells accelerates. u. As long as the cancer cell lacks these genes, it continues to proliferate in a malignant fashion. w When wild-type, intact versions of these genes operate once again within the cancer cell (by cell fusion) it will loose its ability to proliferate or to form tumors. Molecular Biology of Cancer 9
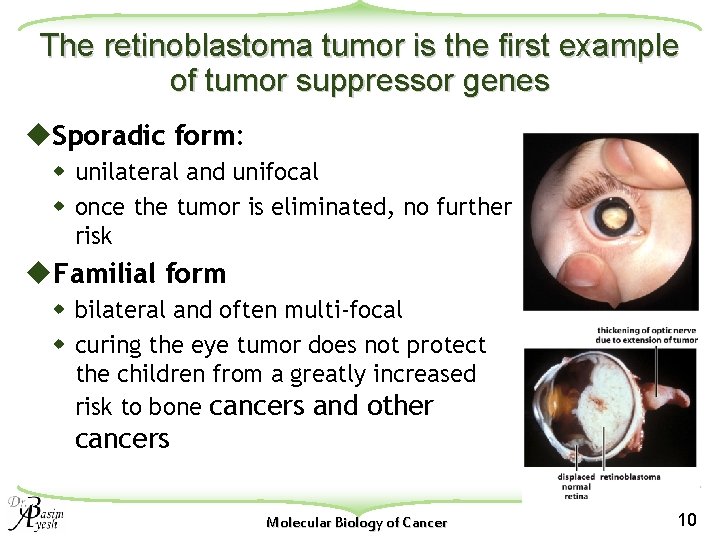
The retinoblastoma tumor is the first example of tumor suppressor genes u. Sporadic form: w unilateral and unifocal w once the tumor is eliminated, no further risk u. Familial form w bilateral and often multi-focal w curing the eye tumor does not protect the children from a greatly increased risk to bone cancers and other cancers Molecular Biology of Cancer 10
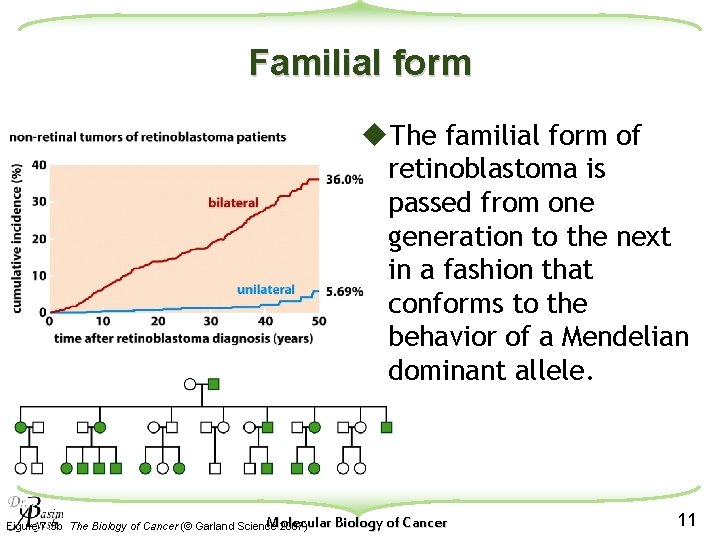
Familial form u. The familial form of retinoblastoma is passed from one generation to the next in a fashion that conforms to the behavior of a Mendelian dominant allele. Molecular Biology of Cancer Figure 7. 5 b The Biology of Cancer (© Garland Science 2007) 11
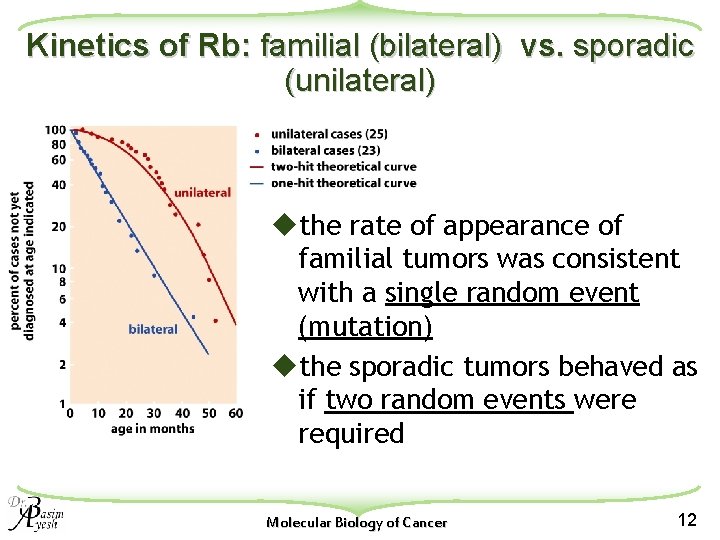
Kinetics of Rb: familial (bilateral) vs. sporadic (unilateral) uthe rate of appearance of familial tumors was consistent with a single random event (mutation) uthe sporadic tumors behaved as if two random events were required Molecular Biology of Cancer 12

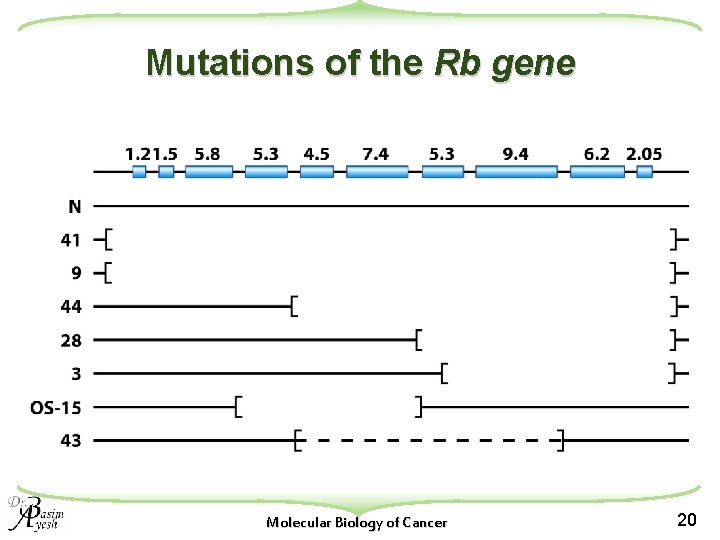
Two-hit hypothesis u. Two “hits” or mutagenic events were necessary for retinoblastoma development u. In an individual with the inherited form: w the first hit is present in the germ line, and thus in all cells of the body. w a second somatic mutation was hypothesized to be necessary for promoting tumor formation. } The second mutation explain the behavior of a Mendelian dominant allele. u. In the nonhereditary form: w both mutations were proposed to arise somatically within the same cell. Molecular Biology of Cancer 13
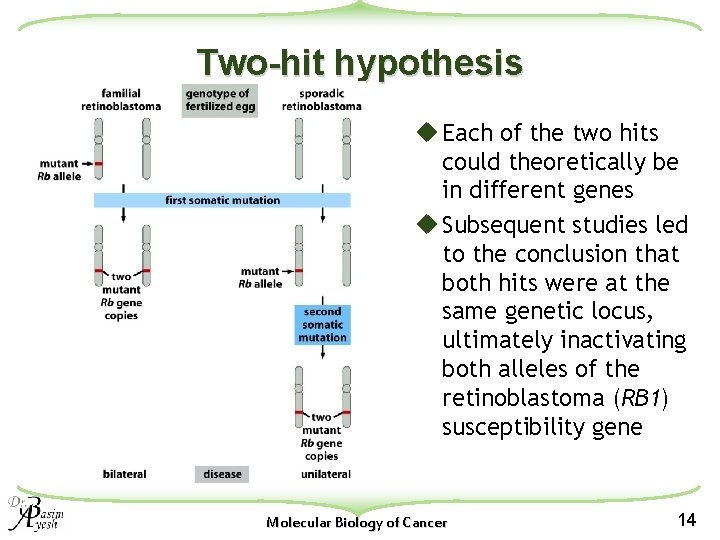
Two-hit hypothesis u Each of the two hits could theoretically be in different genes u Subsequent studies led to the conclusion that both hits were at the same genetic locus, ultimately inactivating both alleles of the retinoblastoma (RB 1) susceptibility gene Molecular Biology of Cancer 14

Loss of Rb heterozygosoty (LOH) Mitotic recombination: a possible mechanism to eliminate the wild-type copy of Rb gene u. The probability of inactivating a single gene copy by mutation is on the order of 10 -6 per cell generation u. The probability of silencing both copies is on the order of 10 -12 per cell generation. u. It seems highly unlikely that both copies of the Rb gene could be eliminated through two recessive mutational event in the relatively small target cell populations in the developing retina (about 106 cells). Molecular Biology of Cancer 15
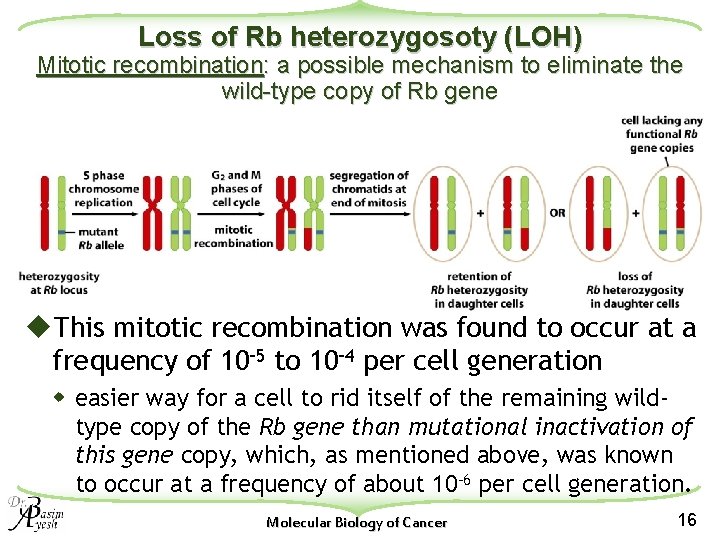
Loss of Rb heterozygosoty (LOH) Mitotic recombination: a possible mechanism to eliminate the wild-type copy of Rb gene u. This mitotic recombination was found to occur at a frequency of 10– 5 to 10– 4 per cell generation w easier way for a cell to rid itself of the remaining wildtype copy of the Rb gene than mutational inactivation of this gene copy, which, as mentioned above, was known to occur at a frequency of about 10– 6 per cell generation. Molecular Biology of Cancer 16
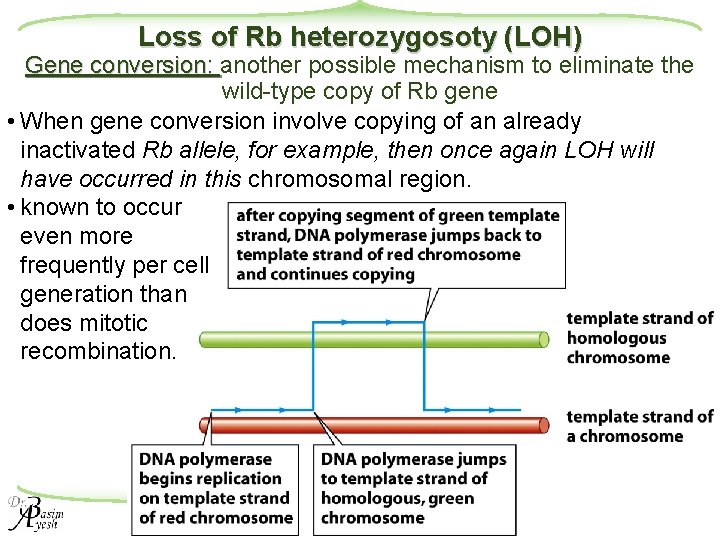
Loss of Rb heterozygosoty (LOH) Gene conversion: another possible mechanism to eliminate the wild-type copy of Rb gene • When gene conversion involve copying of an already inactivated Rb allele, for example, then once again LOH will have occurred in this chromosomal region. • known to occur even more frequently per cell generation than does mitotic recombination. Molecular Biology of Cancer 17
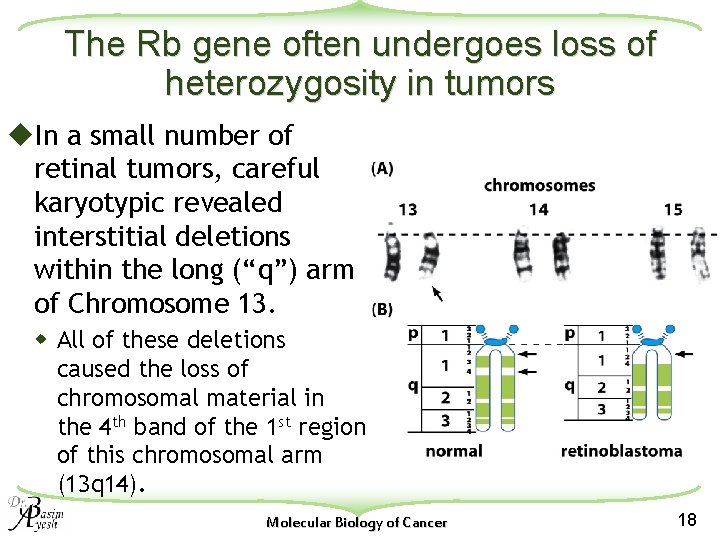
The Rb gene often undergoes loss of heterozygosity in tumors u. In a small number of retinal tumors, careful karyotypic revealed interstitial deletions within the long (“q”) arm of Chromosome 13. w All of these deletions caused the loss of chromosomal material in the 4 th band of the 1 st region of this chromosomal arm (13 q 14). Molecular Biology of Cancer 18

u. A number of genes (including Rb) in this region had been lost simultaneously by the developing retinal tumor cells. u. This is precisely the outcome predicted by the tumor suppressor gene theory. u. LOH achieved by simply breaking off and discarding an entire chromosomal region without replacing it with a copy duplicated from the other, homologous chromosome is called hemizygosity Molecular Biology of Cancer 19
Mutations of the Rb gene Molecular Biology of Cancer 20
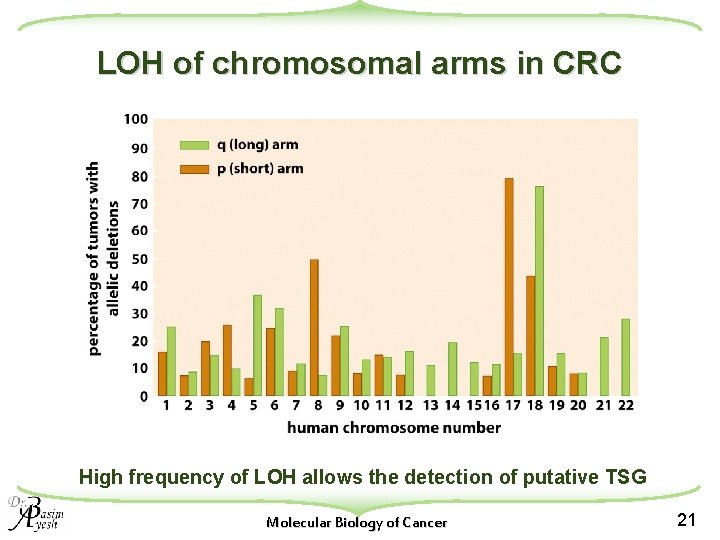
LOH of chromosomal arms in CRC High frequency of LOH allows the detection of putative TSG Molecular Biology of Cancer 21
Molecular Biology of Cancer 22
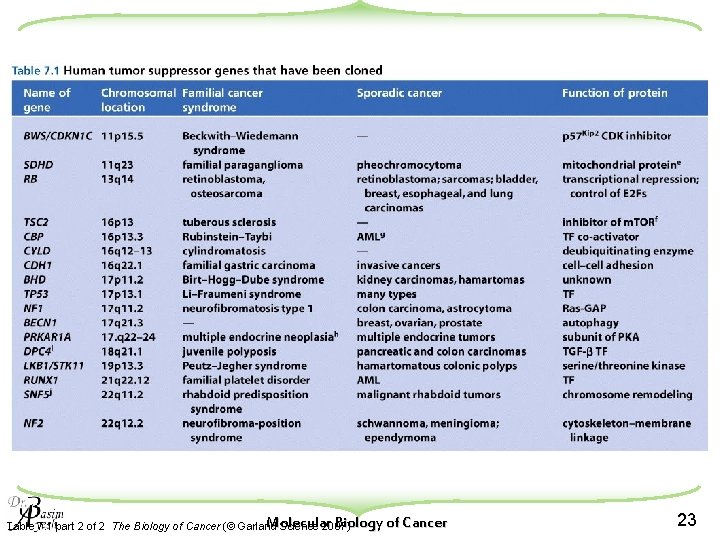
Molecular Biology of Cancer Table 7. 1 part 2 of 2 The Biology of Cancer (© Garland Science 2007) 23

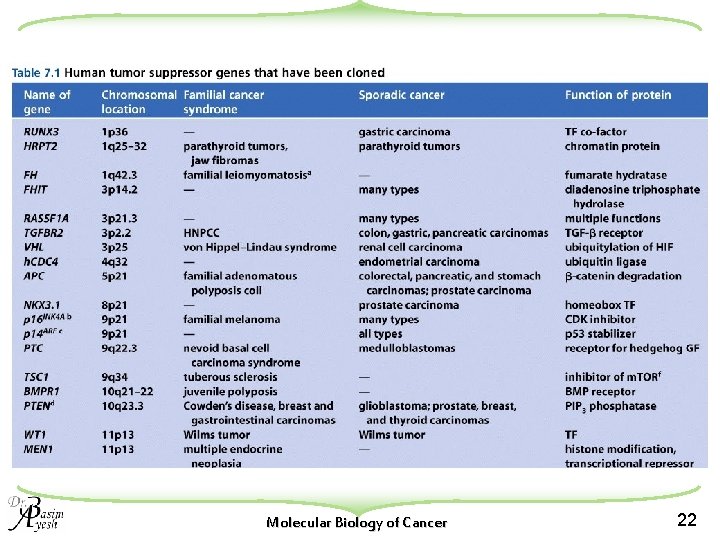
Many familial cancers can be explained by inheritance of mutant tumor suppressor genes u. These genes specify a diverse array of proteins that operate in many different intracellular sites to reduce the risk of cancer. u. An anti-cancer function is the only property that is shared by these otherwise unrelated genes. u. Many familial cancers can be explained by inheritance of mutant TSGs. u. Inheritance of defective copies of most TSGs creates an enormously increased risk for cancer w Often a type of relatively rare tumors Molecular Biology of Cancer 24

There are two distinct classes of familial cancer genes u. Gatekeepers: w Tumor suppressor genes that function to directly control the biology of cells (proliferation, differentiation, or apoptosis) u. Caretakers: w The DNA maintenance genes affect cell biology only indirectly by controlling the rate at which cells accumulate mutant genes Molecular Biology of Cancer 25

Promoter methylation represents an important mechanism for inactivating tumor suppressor genes u. DNA molecules can be altered covalently by the attachment of methyl groups to cytosine bases. w This modification of the genomic DNA is as important as mutation in shutting down tumor suppressor genes. u. In mammalian cells, this methylation is found only when these bases are located in a position that is 5’ to guanosines, that is, in the sequence Cp. G. u. Such methylation can affect the functioning of the DNA in this region of the chromosome. u. When Cp. G methylation occurs in the vicinity of a gene promoter, it can cause repression of transcription of the associated gene. Molecular Biology of Cancer 26
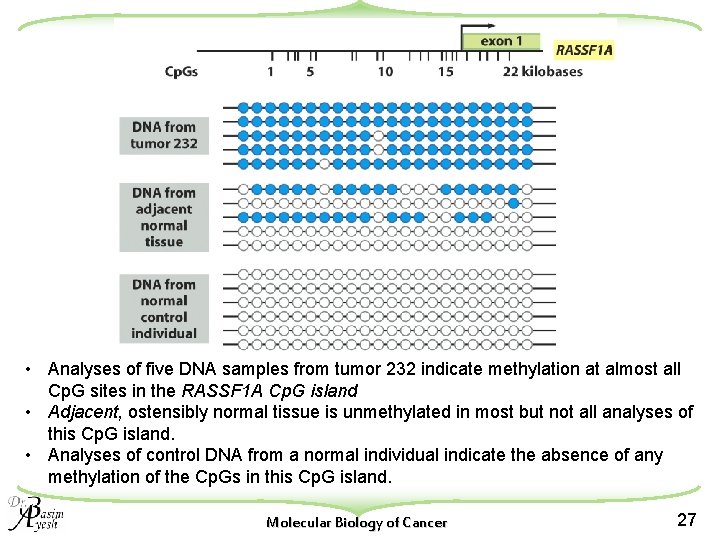
• Analyses of five DNA samples from tumor 232 indicate methylation at almost all Cp. G sites in the RASSF 1 A Cp. G island • Adjacent, ostensibly normal tissue is unmethylated in most but not all analyses of this Cp. G island. • Analyses of control DNA from a normal individual indicate the absence of any methylation of the Cp. Gs in this Cp. G island. Molecular Biology of Cancer 27

u. More than half of the tumor suppressor genes that are involved in familial cancer syndromes because of germ-line mutation have been found to be silenced in sporadic cancers by promoter methylation. w Ex. Rb germ line mutations familial retinoblastoma. w Rb somatic mutations or promoter methylation sporadic retinoblastomas. Molecular Biology of Cancer 28
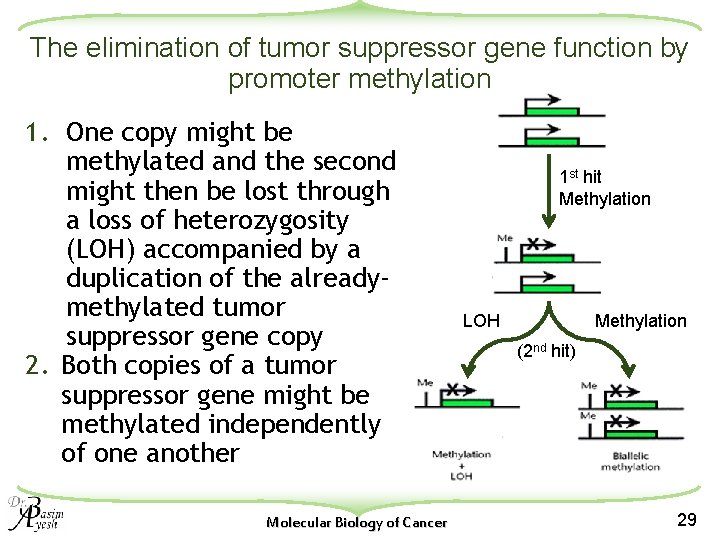
The elimination of tumor suppressor gene function by promoter methylation 1. One copy might be methylated and the second might then be lost through a loss of heterozygosity (LOH) accompanied by a duplication of the alreadymethylated tumor suppressor gene copy 2. Both copies of a tumor suppressor gene might be methylated independently of one another Molecular Biology of Cancer 1 st hit Methylation LOH Methylation (2 nd hit) 29

Example: p 16 INK 4 A tumor suppressor gene u. In a study of the normal bronchial (large airway) epithelia of the lungs: w p 16 INK 4 A methylation: } in 44% of (ostensibly normal) bronchial epithelial cells cultured from current and former smokers } not at all in the comparable cells prepared from those who had never smoked. w LOH in this chromosomal region: } in 71 to 73% of the two smoking populations } in 1. 5 to 1. 7% of never-smokers. u. Conclusion: methylation of critical growthcontrolling genes often occurs early in the complex, multi-step process of tumor formation, long before histological changes are apparent in a tissue Molecular Biology of Cancer 30
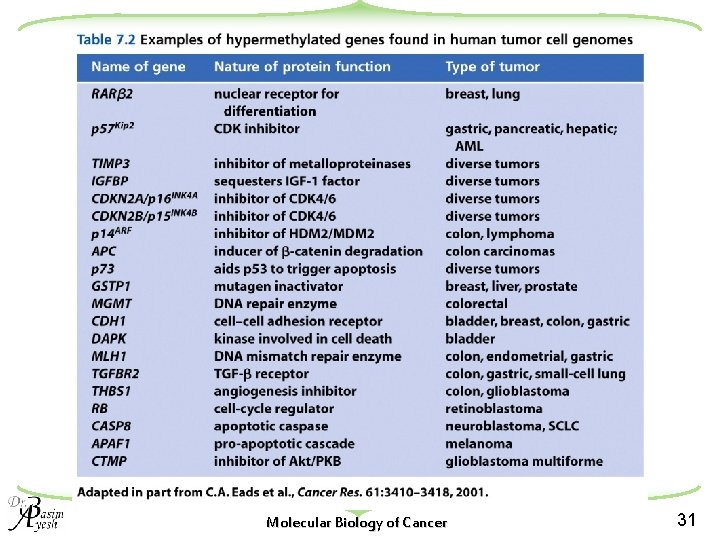
Molecular Biology of Cancer 31

Silencing of genes through promoter methylation can also involve the “caretaker” genes u. Example: the BRCA 1 gene: w Its product maintaining the chromosomal DNA in ways that are still poorly understood. w Inheritance of mutant alleles of BRCA 1 a high lifetime risk of familial breast and, to a lesser extent, ovarian cancer syndrome. w 10 to 15% of sporadic breast carcinomas carry inactive BRCA 1 gene copies that have been silenced through promoter methylation. Molecular Biology of Cancer 32
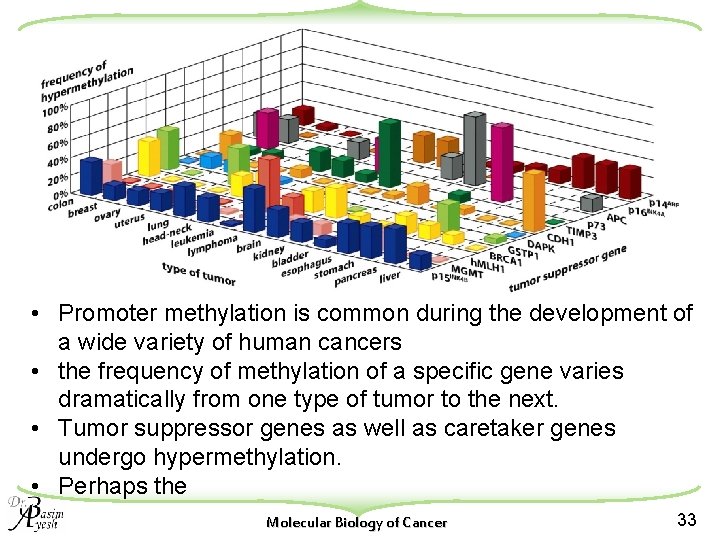
• Promoter methylation is common during the development of a wide variety of human cancers • the frequency of methylation of a specific gene varies dramatically from one type of tumor to the next. • Tumor suppressor genes as well as caretaker genes undergo hypermethylation. • Perhaps the Molecular Biology of Cancer 33
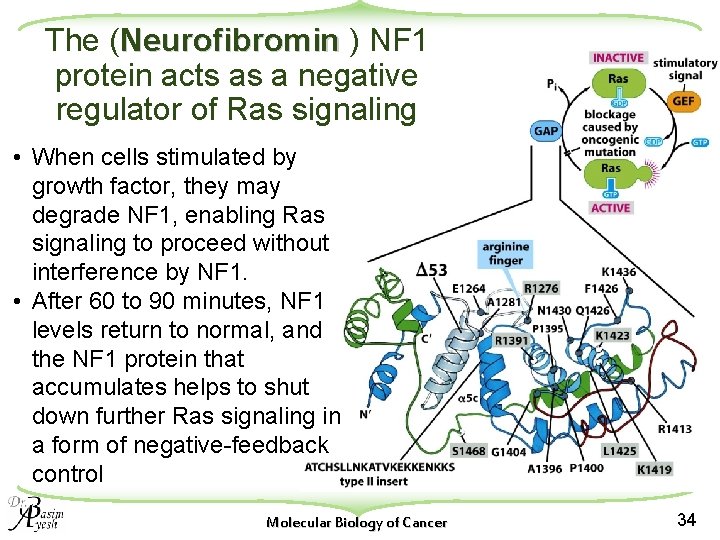
The (Neurofibromin ) NF 1 protein acts as a negative regulator of Ras signaling • When cells stimulated by growth factor, they may degrade NF 1, enabling Ras signaling to proceed without interference by NF 1. • After 60 to 90 minutes, NF 1 levels return to normal, and the NF 1 protein that accumulates helps to shut down further Ras signaling in a form of negative-feedback control Molecular Biology of Cancer 34

u. Neurofibromatosis type 1 is a relatively common familial cancer syndrome, with 1 in 3500 individuals affected on average worldwide. u. The primary feature of this disease is the development of benign neurofibromas of the cell sheaths around nerves in the peripheral nervous system u. On occasion, a subclass of these neurofibromas, progress to malignant tumors termed neurofibrosarcomas. Molecular Biology of Cancer 35
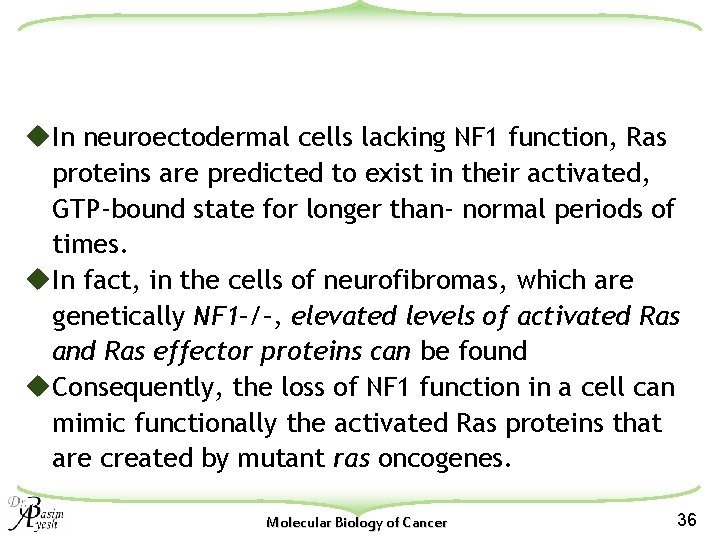
u. In neuroectodermal cells lacking NF 1 function, Ras proteins are predicted to exist in their activated, GTP-bound state for longer than- normal periods of times. u. In fact, in the cells of neurofibromas, which are genetically NF 1–/–, elevated levels of activated Ras and Ras effector proteins can be found u. Consequently, the loss of NF 1 function in a cell can mimic functionally the activated Ras proteins that are created by mutant ras oncogenes. Molecular Biology of Cancer 36
- Slides: 36